Summary of Recommendations and Evidence
The U.S. Preventive Services Task Force (USPSTF) recommends biennial screening mammography for women 50 to 74 years of age. B recommendation.
The decision to start regular, biennial screening mammography before 50 years of age should be an individual one and take patient context into account, including the patient's values regarding specific benefits and harms. C recommendation.
The USPSTF concludes that the current evidence is insufficient to assess the additional benefits and harms of screening mammography in women 75 years or older. I statement.
The USPSTF recommends against teaching breast self-examination (BSE). D recommendation.
The USPSTF concludes that the current evidence is insufficient to assess the additional benefits and harms of clinical breast examination (CBE) beyond screening mammography in women 40 years or older. I statement.
The USPSTF concludes that the current evidence is insufficient to assess the additional benefits and harms of digital mammography or magnetic resonance imaging (MRI) instead of film mammography as screening modalities for breast cancer. I statement.
Rationale
Importance.Breast cancer is the secondleading cause of cancer death among women in the United States. Widespread use of screening, with treatment advances in recent years, have been credited with significant reductions in breast cancer mortality.
Detection.Mammography, as well as physical examination of the breasts (CBE and BSE), can detect presymptomatic breast cancer (Tables 1 and 2). Because of its demonstrated effectiveness in randomized controlled trials of screening, film mammography is the standard for detecting breast cancer; in 2002, the USPSTF found convincing evidence of its adequate sensitivity and specificity.
Table 1. Screening for Breast Cancer Using Film Mammography: Clinical Summary of the USPSTF Recommendation
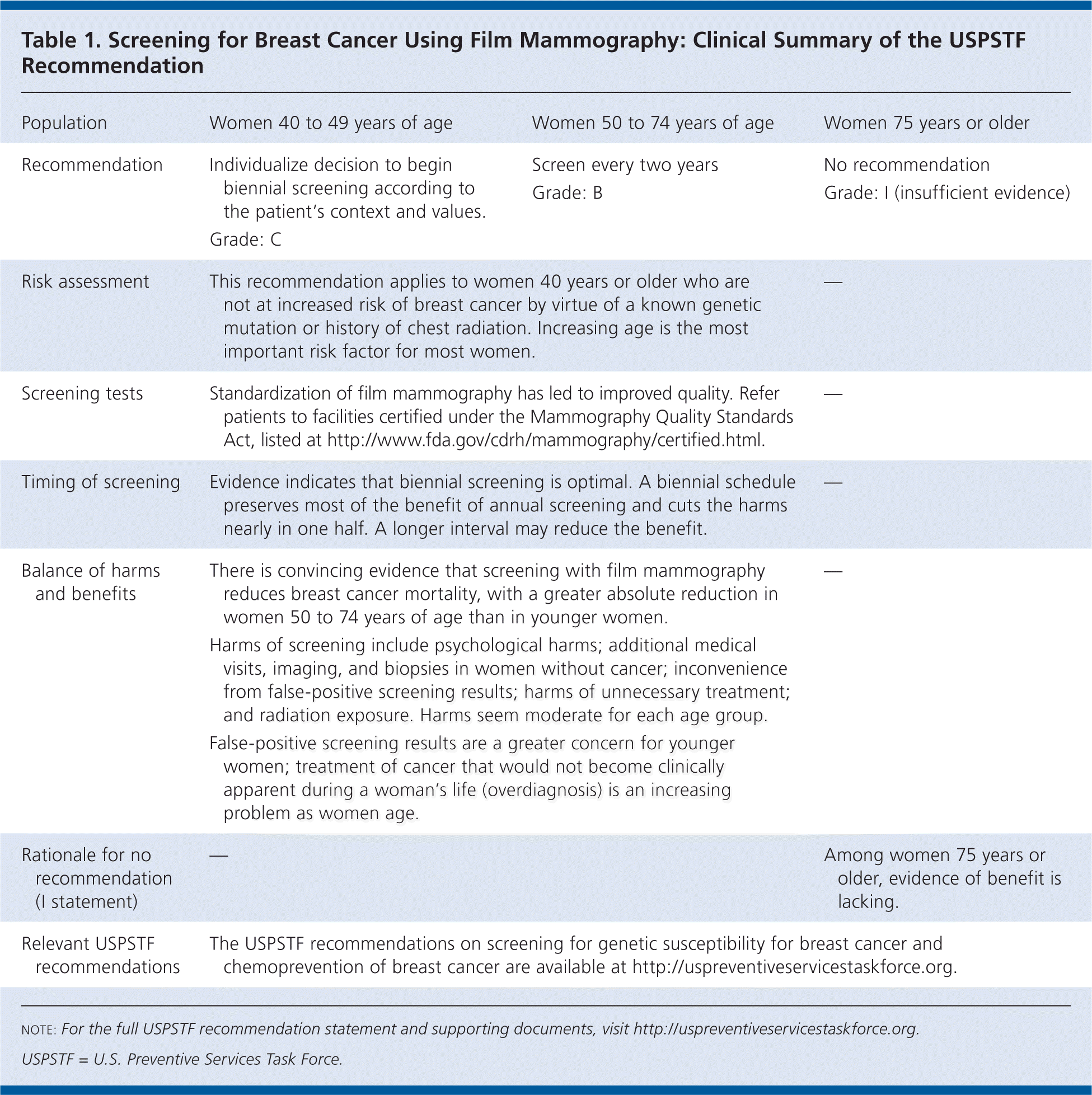
| Population | Women 40 to 49 years of age | Women 50 to 74 years of age | Women 75 years or older |
| Recommendation | Individualize decision to begin biennial screening according to the patient's context and values. Grade: C | Screen every two years Grade: B | No recommendation Grade: I (insufficient evidence) |
| Risk assessment | This recommendation applies to women 40 years or older who are not at increased risk of breast cancer by virtue of a known genetic mutation or history of chest radiation. Increasing age is the most important risk factor for most women. | — | |
| Screening tests | Standardization of film mammography has led to improved quality. Refer patients to facilities certified under the Mammography Quality Standards Act, listed at http://www.fda.gov/cdrh/mammography/certified.html. | — | |
| Timing of screening | Evidence indicates that biennial screening is optimal. A biennial schedule preserves most of the benefit of annual screening and cuts the harms nearly in one half. A longer interval may reduce the benefit. | — | |
| Balance of harms and benefits | There is convincing evidence that screening with film mammography reduces breast cancer mortality, with a greater absolute reduction in women 50 to 74 years of age than in younger women. | — | |
| Harms of screening include psychological harms; additional medical visits, imaging, and biopsies in women without cancer; inconvenience from false-positive screening results; harms of unnecessary treatment; and radiation exposure. Harms seem moderate for each age group. | |||
| False-positive screening results are a greater concern for younger women; treatment of cancer that would not become clinically apparent during a woman's life (overdiagnosis) is an increasing problem as women age. | |||
| Rationale for no recommendation (I statement) | — | Among women 75 years or older, evidence of benefit is lacking. | |
| Relevant USPSTF recommendations | The USPSTF recommendations on screening for genetic susceptibility for breast cancer and chemoprevention of breast cancer are available at http://www.uspreventiveservicestaskforce.org. | ||
note: For the full USPSTF recommendation statement and supporting documents, visit http://www.uspreventiveservicestaskforce.org.
USPSTF = U.S. Preventive Services Task Force.
Table 2. Screening for Breast Cancer Using Methods Other than Film Mammography: Clinical Summary of the USPSTF Recommendation
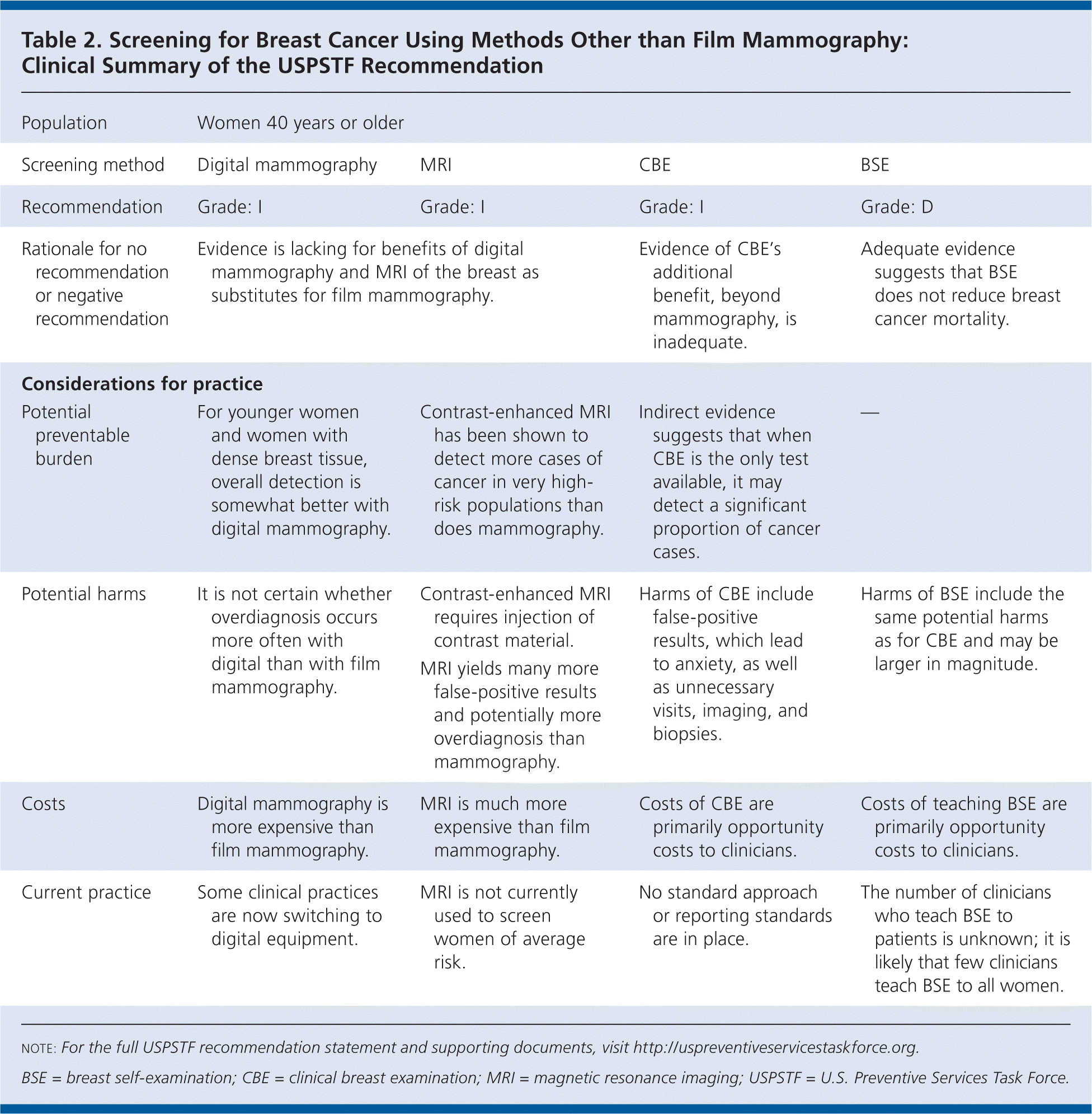
| Population | Women 40 years or older | |||
| Screening method | Digital mammography | MRI | CBE | BSE |
| Recommendation | Grade: I | Grade: I | Grade: I | Grade: D |
| Rationale for no recommendation or negative recommendation | Evidence is lacking for benefits of digital mammography and MRI of the breast as substitutes for film mammography. | Evidence of CBE's additional benefit, beyond mammography, is inadequate. | Adequate evidence suggests that BSE does not reduce breast cancer mortality. | |
| Considerations for practice | ||||
| Potential preventable burden | For younger women and women with dense breast tissue, overall detection is somewhat better with digital mammography. | Contrast-enhanced MRI has been shown to detect more cases of cancer in very high-risk populations than does mammography. | Indirect evidence suggests that when CBE is the only test available, it may detect a significant proportion of cancer cases. | — |
| Potential harms | It is not certain whether overdiagnosis occurs more often with digital than with film mammography. | Contrast-enhanced MRI requires injection of contrast material. | Harms of CBE include false-positive results, which lead to anxiety, as well as unnecessary visits, imaging, and biopsies. | Harms of BSE include the same potential harms as for CBE and may be larger in magnitude. |
| MRI yields many more false-positive results and potentially more overdiagnosis than mammography. | ||||
| Costs | Digital mammography is more expensive than film mammography. | MRI is much more expensive than film mammography. | Costs of CBE are primarily opportunity costs to clinicians. | Costs of teaching BSE are primarily opportunity costs to clinicians. |
| Current practice | Some clinical practices are now switching to digital equipment. | MRI is not currently used to screen women of average risk. | No standard approach or reporting standards are in place. | The number of clinicians who teach BSE to patients is unknown; it is likely that few clinicians teach BSE to all women. |
note: For the full USPSTF recommendation statement and supporting documents, visit http://www.uspreventiveservicestaskforce.org.
BSE = breast self-examination; CBE = clinical breast examination; MRI = magnetic resonance imaging; USPSTF = U.S. Preventive Services Task Force.
Benefits of detection and early intervention.There is convincing evidence that screening with film mammography reduces breast cancer mortality, with a greater absolute reduction in women 50 to 74 years of age than in women 40 to 49 years of age. The strongest evidence for the greatest benefit is in women 60 to 69 years of age.
In women 75 years or older, evidence of benefits of mammography is lacking.
Adequate evidence suggests that teaching BSE does not reduce breast cancer mortality.
The evidence for additional effects of CBE beyond mammography on breast cancer mortality is inadequate.
The evidence for benefits of digital mammography and MRI of the breast, as a substitute for film mammography, is also lacking.
Harms of detection and early intervention.The harms resulting from screening for breast cancer include psychological harms, unnecessary imaging tests and biopsies in women without cancer, and inconvenience from false-positive screening results. Furthermore, one must also consider the harms associated with treatment of cancer that would not become clinically apparent during a woman's lifetime (overdiagnosis), as well as the harms of unnecessary earlier treatment of breast cancer that would have become clinically apparent but would not have shortened a woman's life. Radiation exposure (from radiologic tests), although a minor concern, is also a consideration.
Adequate evidence suggests that the overall harms associated with mammography are moderate for every age group considered, but the main components of the harms shift over time. Although false-positive test results, overdiagnosis, and unnecessary earlier treatment are problems for all age groups, false-positive test results are more common in women 40 to 49 years of age, whereas overdiagnosis is a greater concern for women in the older age groups.
There is adequate evidence that teaching BSE is associated with harms that are at least small. There is inadequate evidence concerning harms of CBE.
USPSTF assessment.The U SPSTF h as reached the following conclusions:
For biennial screening mammography in women 40 to 49 years of age, there is moderate certainty that the net benefit is small. Although the USPSTF recognizes that the benefit of screening seems equivalent for women 40 to 49 years of age and women 50 to 59 years of age, the incidence of breast cancer and the consequences differ. The USPSTF emphasizes the adverse consequences for most women—who will not develop breast cancer—and therefore uses the number needed to screen to save one life as its metric. By this metric, the USPSTF concludes that there is moderate evidence that the net benefit is small in women 40 to 49 years of age.
For biennial screening mammography in women 50 to 74 years of age, there is moderate certainty that the net benefit is moderate.
For screening mammography in women 75 years or older, evidence is lacking and the balance of benefits and harms cannot be determined.
For the teaching of BSE, there is moderate certainty that the harms outweigh the benefits.
For CBE as a supplement to mammography, evidence is lacking and the balance of benefits and harms cannot be determined.
For digital mammography and MRI as a replacement for mammography, the evidence is lacking and the balance of benefits and harms cannot be determined.
Clinical Considerations
PATIENT POPULATION
This recommendation statement applies to women 40 years or older who are not at increased risk of breast cancer by virtue of a known underlying genetic mutation or a history of chest radiation.
ASSESSMENT OF RISK
Increasing age is the most important risk factor for breast cancer for most women. Women without known deleterious genetic mutations (such as BRCA1 or BRCA2) may still have other demographic, physical, or historical risk factors for breast cancer, but none convey a clinically important absolute increased risk of cancer.
SCREENING TESTS
In recent decades, the early detection of breast cancer has been accomplished by physical examination by a clinician (CBE), by a woman herself (BSE), or by mammography. Standardization of mammography practices enacted by the Mammography Quality Standards Act has led to improved mammography quality. Clinicians should refer patients to Mammography Quality Standards Act–certified facilities; the list is available at http://www.fda.gov/cdrh/mammography/certified.html.
SCREENING INTERVALS
In trials that demonstrated the effectiveness of mammography in decreasing breast cancer mortality, screening was performed every 12 to 33 months. The evidence reviewed by the USPSTF indicates that a large proportion of the benefit of screening mammography is maintained by biennial screening, and changing from annual to biennial screening is likely to reduce the harms of mammography screening by nearly one half. At the same time, benefit may be reduced when extending the interval beyond 24 months; therefore, the USPSTF recommends biennial screening.
TREATMENT
Effective treatments, including radiation, chemotherapy (including hormonal treatment), and surgery, are available for invasive carcinoma. Although the standard treatments women receive for ductal carcinoma in situ include surgical approaches, as well as radiation and hormonal therapy, considerable debate exists about the optimal treatment strategy for this condition.
CONSIDERATIONS FOR PRACTICE REGARDING I STATEMENTS
Clinical Breast Examination
Potential preventable burden. The evidence for CBE, although indirect, suggests that CBE may detect a substantial proportion of cancer cases if it is the only screening test available. In parts of the world where mammography is not feasible or unavailable (such as India), CBE is being investigated in this way.
Potential harms. The potential harms of CBE are thought to be small but include false-positive test results, which lead to anxiety and breast cancer worry, as well as repeated visits and unwarranted imaging and biopsies.
Costs. The principal cost of CBE is the opportunity cost incurred by clinicians in the patient encounter.
Current practice. Surveys suggest that the CBE technique used in the United States lacks a standard approach and reporting standards.1 Clinicians who are committed to spending the time on CBE would benefit their patients by considering the evidence in favor of a structured, standardized examination.2
Digital Mammography
Potential preventable burden. Digital mammography detects some cases of cancer not identified by film mammography; film mammography detects some cases of cancer not identified by digital mammography. Overall detection is similar for many women. For women who are younger than 50 years or have dense breast tissue, overall detection is somewhat higher with digital mammography. It is not clear whether this additional detection would lead to reduced mortality from breast cancer.
Potential harms. The possibility of false-positive test results is similar for film and digital mammography. It is uncertain whether overdiagnosis occurs more often with digital mammography than with film mammography.
Costs. Digital mammography is more expensive than film mammography.
Current practice. Some clinical practices are switching their mammography equipment from film to digital. This may curtail the availability of film mammography in some areas.
Magnetic Resonance Imaging
Potential preventable burden. Studies of the use of contrast-enhanced MRI for breast cancer screening have been conducted only in very high-risk populations. In these studies, MRI detected more cases of cancer than did mammography. It is unknown whether detecting these additional cases of cancer would lead to reduced breast cancer mortality.
Potential harms. Contrast-enhanced MRI requires the injection of contrast material. Studies of MRI screening have shown that MRI yields many more false-positive results than does mammography. MRI has the potential to be associated with a greater degree of overdiagnosis than mammography.
Costs. MRI is much more expensive than film or digital mammography.
Current practice. MRI is not currently used for screening women at average risk of breast cancer.
Screening Mammography in Women 75 Years or Older
Potential preventable burden. No women 75 years or older have been included in the multiple randomized clinical trials of breast cancer screening. Breast cancer is a leading cause of death in older women, which might suggest that the benefits of screening could be important at this age. However, three facts suggest that benefits from screening would probably be smaller for this age group than for women 60 to 69 years of age and would probably decrease with increasing age: (1) the benefits of screening only occur several years after the actual screening test, whereas the percentage of women who survive long enough to benefit decreases with age; (2) a higher percentage of the type of breast cancer detected in this age group is the more easily treated estrogen receptor–positive type; and (3) women of this age are at much greater risk of dying of other conditions that would not be affected by breast cancer screening.
Potential harms. Screening detects not only cancer that could lead to a woman's death but also cancer that will not shorten a woman's life. Women cannot benefit from—but can be harmed by—the discovery and treatment of this second type of cancer, which includes cancer that might some day become clinically apparent, as well as cancer that never will. Detection of cancer that would never have become clinically apparent is called overdiagnosis, and it is usually followed by overtreatment. Because of a shortened life span among women 75 years or older, the probability of overdiagnosis and unnecessary earlier treatment increases dramatically after about 70 to 75 years of age. Overdiagnosis and unnecessary earlier treatment are important potential harms from screening women in this age group.
Current practice. Studies show that many women 75 years or older are currently being screened.
USEFUL RESOURCES
Other USPSTF recommendations on screening for genetic susceptibility for breast cancer and chemoprevention of breast cancer are available on the USPSTF Web site (http://www.uspreventiveservicestaskforce.org).

