Summary of Recommendations and Evidence
The U.S. Preventive Services Task Force (USPSTF) recommends screening adults for depression when staff-assisted depression care supports are in place to assure accurate diagnosis, effective treatment, and follow-up (Table 1). B recommendation.
Table 1. Screening for Depression in Adults: Clinical Summary of the USPSTF Recommendation
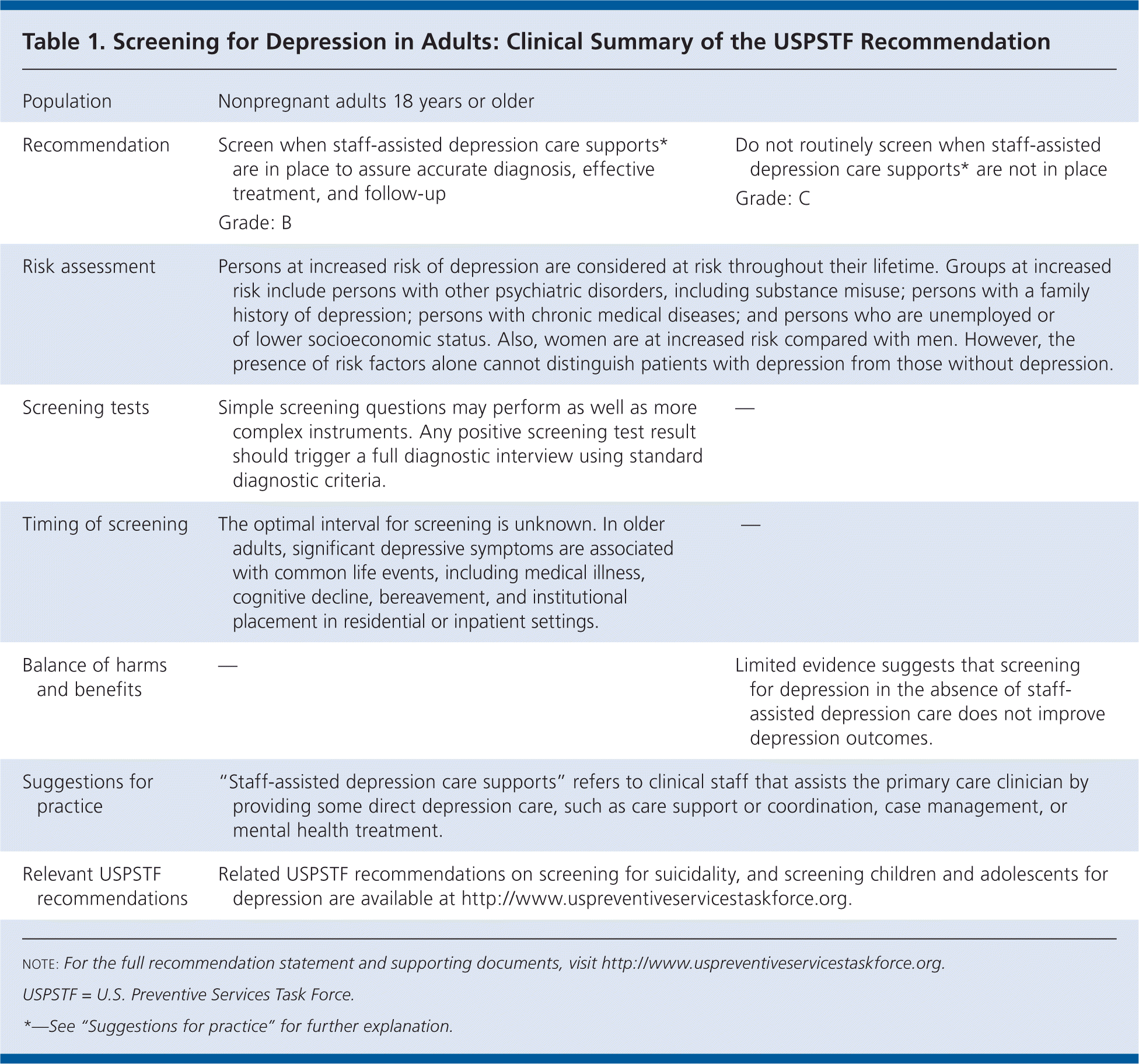
| Population | Nonpregnant adults 18 years or older | |
| Recommendation | Screen when staff-assisted depression care supports* are in place to assure accurate diagnosis, treatment, and follow-up Grade: B | Do not routinely screen when staff-assisted effective depression care supports* are not in place Grade: C |
| Risk assessment | Persons at increased risk of depression are considered at risk throughout their lifetime. Groups at increased risk include persons with other psychiatric disorders, including substance misuse; persons with a family history of depression; persons with chronic medical diseases; and persons who are unemployed or of lower socioeconomic status. Also, women are at increased risk compared with men. However, the presence of risk factors alone cannot distinguish patients with depression from those without depression. | |
| Screening tests | Simple screening questions may perform as well as more complex instruments. Any positive screening test result should trigger a full diagnostic interview using standard diagnostic criteria. | — |
| Timing of screening | The optimal interval for screening is unknown. In older adults, significant depressive symptoms are associated with common life events, including medical illness, cognitive decline, bereavement, and institutional placement in residential or inpatient settings. | — |
| Balance of harms and benefits | — | Limited evidence suggests that screening for depression in the absence of staff-assisted depression care does not improve depression outcomes. |
| Suggestions for practice | “Staff-assisted depression care supports” refers to clinical staff that assists the primary care clinician by providing some direct depression care, such as care support or coordination, case management, or mental health treatment. | |
| Relevant USPSTF recommendations | Related USPSTF recommendations on screening for suicidality, and screening children and adolescents for depression are available at http://www.uspreventiveservicestaskforce.org. | |
note:For the full recommendation statement and supporting documents, visit http://www.uspreventiveservicestaskforce.org.
USPSTF = U.S. Preventive Services Task Force.
*—See “Suggestions for practice” for further explanation.
The USPTF recommends against routinely screening adults for depression when staff-assisted depression care supports are not in place. There may be considerations that support screening for depression in an individual patient. C recommendation.
Rationale
Importance. Depression is among the leading causes of disability in persons 15 years or older. It affects individuals, families, businesses, and society. It is common in primary care patients.
Detection. The USPSTF found good evidence that screening improves the accurate identification of patients with depression in primary care settings.
Benefits of detection and early intervention. The USPSTF found good evidence that treatment with antidepressants, psychotherapy, or both decreases clinical morbidity in adults and older adults with depression identified through screening in primary care settings.
The USPSTF found good evidence that programs combining depression screening and feedback with staff-assisted depression care supports improve clinical outcomes in adults and older adults.
The USPSTF found fair evidence that screening and feedback alone without staff-assisted care supports does not improve clinical outcomes in adults and older adults.
Harms of detection and early intervention. The USPSTF found no evidence of harms of screening for depression in adults or older adults.
The USPSTF found at least fair-quality evidence that second-generation antidepressants (mostly selective serotonin reuptake inhibitors [SSRIs]) increase suicidal behaviors in adults 18 to 29 years of age, especially those with major depressive disorder and those who receive paroxetine. The USPSTF found at least fair-quality evidence that SSRI use is associated with an increased risk of upper gastrointestinal bleeding in adults older than 70 years, and the risk increases with age.
USPSTF assessment. The USPSTF concludes that for adults who receive care in clinical practices that have staff-assisted depression care supports in place, there is at least moderate certainty that the net benefit of screening for depression is at least moderate.
The USPSTF concludes that for adults who receive care in clinical practices without staff-assisted depression care supports in place, there is at least moderate certainty that the net benefit of screening for depression is small.
Clinical Considerations
Patient population. This recommendation applies to nonpregnant adults, including older adults. It does not apply to children and adolescents, who are considered a separate population.
Assessment of risk. Persons at increased risk of depression are considered at risk throughout their lifetime. Groups at increased risk include persons with other psychiatric disorders, including substance misuse; persons with a family history of depression; persons with chronic medical diseases; and persons who are unemployed or of lower socioeconomic status. Also, women are at increased risk compared with men. Significant depressive symptoms are associated with common life events in older adults, including medical illness, cognitive decline, bereavement, and institutional placement in residential or inpatient settings. However, the presence of risk factors alone cannot distinguish patients with depression from those without depression.
Screening tests. In 2002, the USPSTF reviewed evidence about the accuracy of screening instruments in identifying depression in adults.1 Many formal screening tools are available, including instruments designed specifically for older adults. Asking two simple questions about mood and anhedonia (“Over the past two weeks, have you felt down, depressed, or hopeless?” and “Over the past two weeks, have you felt little interest or pleasure in doing things?”) may be as effective as using more formal instruments.2 There is little evidence to recommend one screening method over another; therefore, clinicians may choose the method most consistent with their personal preference, the patient population being served, and the practice setting.
All positive screening tests should trigger full diagnostic interviews that use standard diagnostic criteria (i.e., those from the updated Diagnostic and Statistical Manual of Mental Disorders, 4th ed.) to determine the presence or absence of specific depressive disorders, such as major depressive disorder or dysthymia. The severity of depression and comorbid psychological problems (e.g., anxiety, panic attacks, substance abuse) should be addressed.
Treatment. The reviews of evidence on which this recommendation is based cover treatment of adults with antidepressants or psychotherapy 1 and updated evidence on the efficacy of depression treatment in older adults.3 Treatment may include antidepressants or specific psychotherapeutic approaches (e.g., cognitive behavior therapy, brief psychosocial counseling) alone or in combination. Both are effective in treating adults and older adults.
In treating patients 18 to 29 years of age, clinicians may want to select a psychotherapeutic approach or medications other than SSRIs because of the increased risk of suicidal behavior associated with SSRI use. Similarly, for adults 65 years or older, clinicians may want to select a psychotherapeutic approach or medications other than SSRIs because of the increased risk of upper gastrointestinal bleeding associated with the use of SSRIs. In addition, the concurrent use of SSRIs with a nonsteroidal anti-inflammatory drug or low-dose aspirin increases the risk of upper gastrointestinal bleeding in adults (40 to 79 years of age), although the increase in risk is less with aspirin. The risk of upper gastrointestinal bleeding is greater with medications that feature a moderate to high degree of serotonin reuptake inhibition.
Staff-assisted depression care supports. “Staff-assisted depression care supports” refers to clinical staff that assist the primary care clinician by providing some direct depression care, such as care support or coordination, case management, or mental health treatment.
In the available evidence, the lowest effective level of staff-assisted depression care supports consisted of a screening nurse who advised resident physicians of positive screening results and provided a protocol that facilitated referral to behavior treatment.4 At the highest level, staff-assisted depression care supports included screening; institutional monetary commitment; staff and clinician training (one- or two-day workshops); clinician manuals; monthly training lectures; academic detailing; many materials for clinicians, staff, and patients; an initial visit with a nurse specialist for assessment, education, and discussion of patient preferences and goals; a visit with a trained nurse specialist for follow-up assessment and ongoing support for adherence to medication for patients prescribed antidepressant medications; a visit with a trained therapist for cognitive behavior therapy; and a reduced copay for patients referred for psychotherapy.5,6
In a successful study designed for practices without ready access to mental health specialty care, office staff recruited, screened, and enrolled participants who screened positive for depression before a clinic visit.7 If the physician confirmed the depression diagnosis, the participant was scheduled for a return visit with the physician and to meet with the nurse specialist in one week. The nurse specialist reassessed the patient's level of depression, discussed treatment options and preferences, and asked the participant to complete a homework assignment. Participants completed up to eight additional sessions that followed the same pattern, by phone or in person.
Multidisciplinary team-based primary care that includes self-management support and care coordination has been shown to be effective in management of depression. These components of primary care are detailed in recent recommendations from the Task Force on Community Preventive Services.8 It recommends collaborative care for treatment of adults 18 years or older with major depression on the basis of strong evidence of effectiveness in improving short-term treatment outcomes. As defined, collaborative care and disease management of depressive disorders includes a systematic, multi-component, team-based approach that “strengthens and supports self-care ... while assuring that effective medical, preventive, and health maintenance interventions take place” to improve quality and outcome of patient care for treatment of major depressive disorders.9
Screening intervals. The optimal interval for screening for depression is unknown. Recurrent screening may be most productive in patients with a history of depression, unexplained somatic symptoms, comorbid psychological conditions (e.g., panic disorder, generalized anxiety), substance abuse, or chronic pain.
Other approaches to prevention. The Task Force on Community Preventive Services also has made several recommendations about depression care in older adults. It recommends clinic-based depression care management to reduce depression in older adults on the basis of sufficient evidence and home-based depression care management on the basis of strong evidence. The Task Force on Community Preventive Services found insufficient evidence to determine the effectiveness of community-based exercise interventions for reducing depression in older adults.
The Task Force on Community Preventive Services makes recommendations on population-based interventions appropriate for use by communities and health care systems to promote health and to prevent disease, injury, disability, and premature death. More information about the task force and its recommendations on depression interventions is available at http://www.thecommunityguide.org.
Useful resources. In 2009, the USPSTF updated its recommendation on screening for depression in children and adolescents. The USPSTF recommends screening adolescents (12 to 18 years of age) for major depressive disorder when systems are in place to assure accurate diagnosis, psychotherapy (cognitive behavior or interpersonal), and follow-up (B recommendation).
In addition, the USPSTF concluded that the current evidence is insufficient to assess the balance of benefits and harms of screening children (seven to 11 years of age) for major depressive disorder (I statement). In 2004, the USPSTF concluded that the evidence is insufficient to recommend for or against routine screening by primary care clinicians to detect suicide risk in the general population (I statement). At that time, the USPSTF found no evidence that screening for suicide risk reduces suicide attempts or mortality. The USPSTF also found limited evidence on the accuracy of screening tools to identify suicide risk in the primary care setting, including tools to identify those at high risk, and found no evidence that directly addressed the harms of screening and treatment of suicide risk. In addition, the USPSTF found insufficient evidence that treatment of persons at high risk reduces suicide attempts or mortality.
For the full recommendation statements and evidence reviews, visit the USPSTF Web site (http://www.uspreventiveservicestaskforce.org).

