
Am Fam Physician. 2011;83(5):585-590
Author disclosure: Nothing to disclose.
Surgical site infections are the most common nosocomial infections in surgical patients, accounting for approximately 500,000 infections annually. Surgical site infections also account for nearly 4 million excess hospital days annually, and nearly $2 billion in increased health care costs. To reduce the burden of these infections, a partnership of national organizations, including the Centers for Medicare and Medicaid Services and the Centers for Disease Control and Prevention, created the Surgical Care Improvement Project and developed six infection prevention measures. Of these, three core measures contain recommendations regarding selection of prophylactic antibiotic, timing of administration, and duration of therapy. For most patients undergoing clean-contaminated surgeries (e.g., cardiothoracic, gastrointestinal, orthopedic, vascular, gynecologic), a cephalosporin is the recommended prophylactic antibiotic. Hospital compliance with infection prevention measures is publicly reported. Because primary care physicians participate in the pre- and postoperative care of patients, they should be familiar with the Surgical Care Improvement Project recommendations.
A surgical site infection is defined as an infection that occurs at or near a surgical incision within 30 days of the procedure or within one year if an implant is left in place.1,2 The Centers for Disease Control and Prevention (CDC) estimates that approximately 500,000 surgical site infections occur annually in the United States.3 They are the leading cause of nosocomial infections after surgery, accounting for nearly 40 percent of nosocomial infections in surgical patients.2 The cost of care for patients with surgical site infections is nearly threefold higher than that for surgical patients without the infections during the first eight weeks after hospital discharge.3 These infections reduce patients' quality of life4 and account for 3.7 million excess hospital days and more than $1.6 billion in excess costs annually.5 Furthermore, patients who develop surgical site infections are five times more likely to be readmitted to the hospital, 60 percent more likely to spend time in the intensive care unit, and twice as likely to die compared with surgical patients without the infections.6
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| To help prevent surgical site infection, the perioperative antibiotic should be infused within one hour before incision. | A | 7, 9, 11, 12, 18 |
| Perioperative antibiotic prophylaxis should be consistent with published guidelines. | C | 7, 8 |
| Perioperative antibiotic prophylaxis should generally be discontinued within 24 hours after surgery completion. | C | 7, 8 |
In 2002, the Centers for Medicare and Medicaid Services (CMS), collaborating with the CDC, developed and implemented the Surgical Infection Prevention Project. Its goal was to provide evidence-based performance measures for the appropriate selection, administration, and termination of prophylactic antibiotics for patients undergoing clean-contaminated surgeries. In 2003, the CDC, CMS, and 10 additional national organizations developed the Surgical Care Improvement Project (SCIP).7 The Surgical Infection Prevention Project measures were subsequently incorporated into the SCIP, which has been widely disseminated.7–9
Publicly reported SCIP performance measures targeted at reducing postoperative surgical site infections include the following (the first three comprise the core infection prevention measures)10:
Prophylactic antibiotics should be initiated within one hour before surgical incision, or within two hours if the patient is receiving vancomycin or fluoroquinolones.
Patients should receive prophylactic antibiotics appropriate for their specific procedure.
Prophylactic antibiotics should be discontinued within 24 hours of surgery completion (within 48 hours for cardiothoracic surgery).
Postoperative 6 a.m. blood glucose levels should be controlled (200 mg per dL [11.10 mmol per L] or less) in patients undergoing cardiac surgery.
Surgical site hair removal should be appropriate for the location and procedure (e.g., clippers, depilation, no hair removal).
Patients undergoing colorectal surgery should be normothermic (96.8°F [36°C] or greater) within the first 15 minutes after leaving the operating room.
Despite demonstrated reductions in the incidence of surgical site infections following implementation of these measures,11–14 a recent survey of U.S. hospitals found that the recommendations are not routinely followed. Only 55.7 percent of surgical patients received prophylactic antibiotics within one hour of incision, and the antibiotic was discontinued within the 24 hours after surgery in only 40.7 percent of patients.15 Other studies show that approximately 80 to 90 percent of surgical patients received antibiotic prophylaxis, but the choice of regimen, timing of administration, or duration of prophylaxis were inappropriate in approximately 25 to 50 percent of patients.14 As an incentive to reduce rates of surgical site infections, CMS reduced reimbursement to hospitals for some of these infections.16
Because primary care physicians are involved in pre- and postoperative care and some perform or assist in surgical procedures,17 they have the opportunity to impact the incidence of surgical site infections by understanding which surgeries call for prophylactic antibiotic administration, which antibiotic is appropriate, and when the antibiotic should be administered and discontinued.
Core Infection Prevention Measures: Evidence Summary
Prophylactic antibiotic administration should be initiated within one hour before the surgical incision, or within two hours if the patient is receiving vancomycin or fluoroquinolones.
The goal of antibiotic prophylaxis is to ensure effective serum and tissue levels of the drug for the duration of the surgery. Analysis of data from 2,847 patients undergoing clean or clean-contaminated surgical procedures showed that those receiving antibiotic prophylaxis within two hours before incision had a surgical site infection rate of 0.6 percent. In contrast, patients receiving prophylactic antibiotics more than three hours after surgical incision had a twofold increase in surgical site infection, and those receiving antibiotics more than two hours before incision had approximately a sixfold increase in risk.11
Another study demonstrated that the risk of surgical site infection following total hip arthroplasty was lowest when the appropriate antibiotic was administered within one hour before incision.12 A recent multicenter study of 29 hospitals in the United States also supported administration within one hour before incision and showed that administration within 30 minutes before incision may reduce the risk even further.13 A meta-analysis of randomized controlled trials showed that antibiotic administration just before or at the time of anesthesia resulted in significantly lower infection rates in patients undergoing spinal surgery.18
When Is Repeat Antibiotic Infusion Considered? Most authorities and study results support a single dose of antibiotic given within one hour before incision.8,9,19,20 However, the antibiotic infusion may be repeated intraoperatively for procedures lasting more than four hours and when substantial blood loss (more than 1,500 mL) occurs.9,21 The antibiotic should be redosed at one to two times the half-life of the drug.9 A retrospective analysis of patients who underwent cardiac surgery showed that in patients with surgeries lasting more than four hours, the risk of surgical site infection was reduced from 16 to 7.7 percent with repeat intra-operative dosing of cefazolin.22 Another study of patients undergoing colorectal surgery found that low gentamicin levels at wound closure were associated with an increased risk of surgical site infection.23
Antibiotic prophylaxis should be appropriate for the specific procedure and consistent with SCIP guidelines.
The prophylactic regimen in patients undergoing surgery should include an agent effective against the most likely infecting organisms, but need not eradicate every potential pathogen.8 The choice of antibiotic should be based on the local antibiogram. Antibiotic prophylaxis should be used in all clean-contaminated procedures and in some clean procedures in which a surgical site infection would have devastating consequences for the patient (e.g., placement of a prosthetic joint).8,9 Patients undergoing dirty or contaminated procedures (e.g., repair of a perforated colon) generally do not require antimicrobial prophylaxis because they already are receiving specific antibiotic treatment for an established infection.8 Cephalosporins are recommended for most surgical procedures because they are active against the common skin pathogens Staphylococcus aureus and Streptococcal species. For some gynecologic or gastrointestinal surgeries, antibiotic combinations are recommended.8
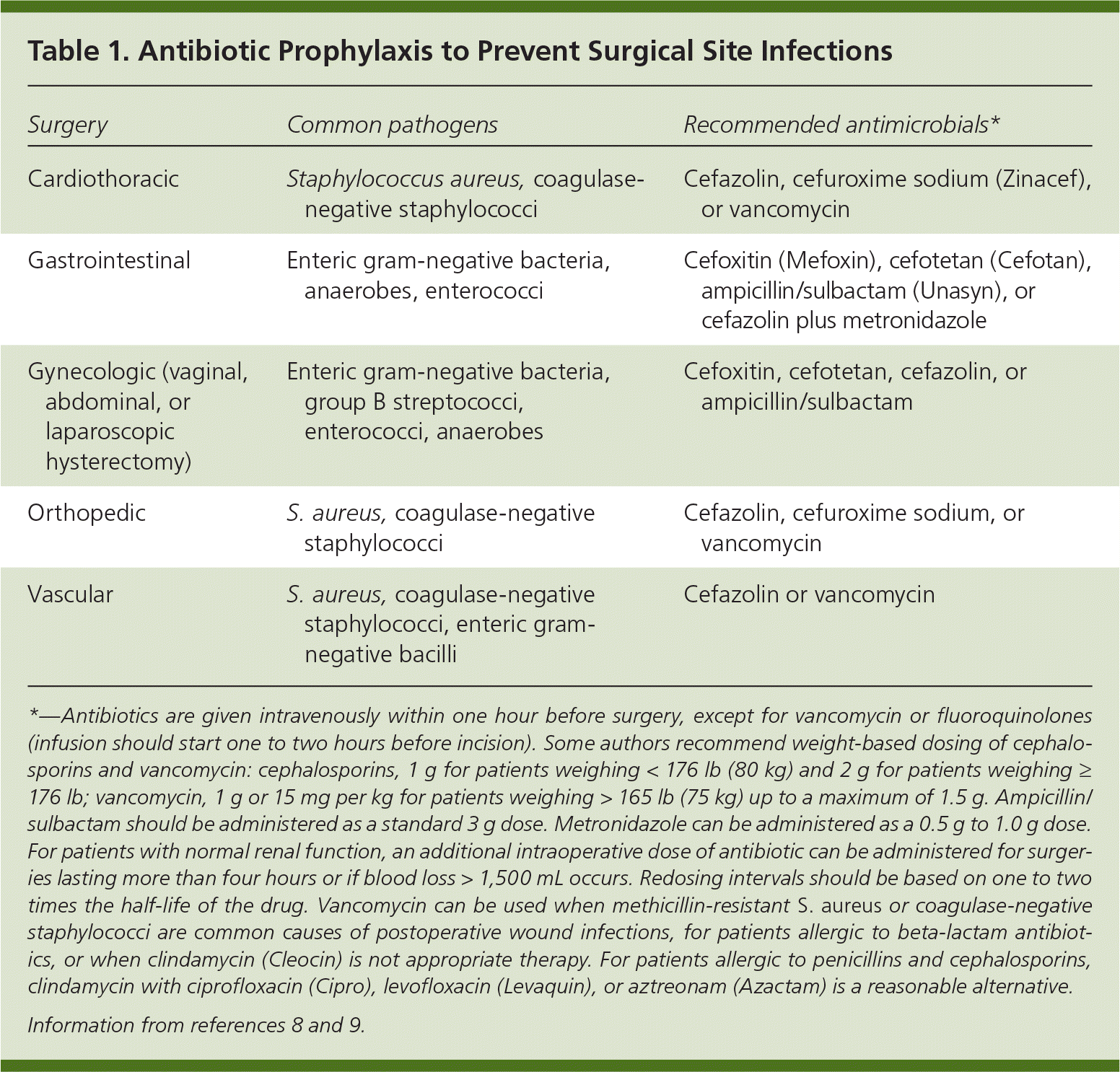
| Surgery | Common pathogens | Recommended antimicrobials* |
|---|---|---|
| Cardiothoracic | Staphylococcus aureus, coagulase-negative staphylococci | Cefazolin, cefuroxime sodium (Zinacef), or vancomycin |
| Gastrointestinal | Enteric gram-negative bacteria, anaerobes, enterococci | Cefoxitin (Mefoxin), cefotetan (Cefotan), ampicillin/sulbactam (Unasyn), or cefazolin plus metronidazole |
| Gynecologic (vaginal, abdominal, or laparoscopic hysterectomy) | Enteric gram-negative bacteria, group B streptococci, enterococci, anaerobes | Cefoxitin, cefotetan, cefazolin, or ampicillin/sulbactam |
| Orthopedic | S. aureus, coagulase-negative staphylococci | Cefazolin, cefuroxime sodium, or vancomycin |
| Vascular | S. aureus, coagulase-negative staphylococci, enteric gram-negative bacilli | Cefazolin or vancomycin |
When Is Vancomycin Considered the Agent of Choice for Prophylaxis? Routine use of vancomycin for surgical site infection prophylaxis is not recommended for any type of surgery.2,8 However, vancomycin may be considered for patients with a beta-lactam allergy and in institutions with high rates of methicillin-resistant S. aureus (MRSA) or an increase in surgical site infections caused by MRSA or coagulase-negative staphylococci, particularly following prosthetic joint or cardiothoracic surgeries.2,8 Available data do not clearly define a threshold constituting a high rate of MRSA infection that can be applied to all institutional situations.24 Furthermore, studies from institutions with perceived high rates of MRSA have not provided consistent evidence that vancomycin is superior to cefazolin for reducing surgical site infections.25–27 In one study, patients who received vancomycin prophylaxis were more likely to develop surgical site infections from methicillin-sensitive S. aureus compared with those who received cefazolin.26 The decision to routinely use vancomycin for high-risk surgeries should consider local rates of MRSA infection and include consultation among surgeons, infectious disease physicians, and infection prevention personnel.
Should Patients Be Screened for MRSA to Determine if Vancomycin Should Be Used? The effect of preoperative identification and treatment of MRSA carriers on the incidence of surgical site infections is controversial.28–30 The Medical Letter consultants acknowledge the controversy, but do not offer a recommendation.8 Guidelines from the Society of Thoracic Surgeons recommend routine administration of topical mupirocin (Bactroban) for all patients undergoing cardiovascular procedures in the absence of documented tests negative for MRSA.31 The American Academy of Orthopaedic Surgeons advises that patients at risk of colonization by methicillin-resistant or methicillin-sensitive S. aureus be screened and decolonized preoperatively.32 Populations at risk of MRSA colonization may include patients recently discharged from a hospital or long-term care facility, patients with previous MRSA colonization or infection, patients with chronic hemodialysis, and intravenous drug users.33
Prophylactic antibiotics should be discontinued within 24 hours of surgery completion (48 hours for cardiothoracic surgery).
Current guidelines recommend that prophylactic antibiotics end within 24 hours of surgery completion.8 There is no documented benefit of antibiotics after wound closure in the reduction of surgical site infections.19,20,34 However, guidelines from the Society of Thoracic Surgeons recommend that antibiotic prophylaxis be continued for 48 hours after the completion of cardiothoracic surgery due to the effects of cardiopulmonary bypass on immune function and antibiotic pharmacokinetics.35,36 There is no evidence to support using prophylactic antibiotics for longer than 48 hours.35,37 Antibiotics given for implantation of a pacemaker or defibrillator should be discontinued within 24 hours of surgery.8,38
Should the Duration of Prophylactic Antibiotics Be Extended When Drainage Tubes Are Still in Place? Surveys of academic orthopedic surgeons in the United States 39 and cardiac surgeons in the United Kingdom40 show that 45 and 28 percent, respectively, continue antibiotics while drainage tubes are in place. However, there is no evidence to support antibiotic administration until drains are removed.39,34 The practice is not recommended by the American Academy of Orthopaedic Surgeons,21 Society of Thoracic Surgeons,35 or Medical Letter consultants.8
Further Research
Although evidence shows that adherence to SCIP infection prevention measures reduces the incidence of surgical site infections,11,12,18 it is uncertain which combination of measures and other factors have the greatest impact on reducing infections. An analysis of data from 398 hospitals in the United States showed that adherence to the core infection prevention measures may not reduce surgical site infections to the same degree as adherence to all of the measures.10 Research is needed to clarify how other factors, such as the patient's preexisting conditions, operating room traffic, and postoperative wound management, influence the incidence of surgical site infections. Determining the benefit of preoperative screening for MRSA and the appropriate use of vancomycin are also important areas for study.
Data Sources: Studies evaluating the use of antibiotic prophylaxis to prevent surgical site infections were identified by searching Medline for English-language literature using the following keywords: surgical antibiotic prophylaxis, perioperative antibiotics, surgical site infections, surgical wound infections, surgical care improvement project, or surgical infection prophylaxis. Other sources searched were the Agency for Healthcare Research and Quality Evidence Reports, Cochrane Database of Systematic Reviews, National Guideline Clearinghouse, and Institute for Clinical Systems Improvement. All searches were performed electronically, initially conducted in January 2010, and repeated in July 2010. The bibliographies of pertinent articles were searched to identify additional references. Included articles consisted of randomized controlled studies, evidence-based guidelines, systematic reviews, and meta-analyses.
