Guideline source: Institute for Clinical Systems Improvement (ICSI)
Evidence rating system used? Yes
Literature search described? No
Attention-deficit/hyperactivity disorder (ADHD) is a common condition marked by inattention, hyperactivity, and impulsivity. Diagnosis can be straightforward or complex; however, many patients who present with learning or behavioral problems and in whom ADHD is suspected can be evaluated and treated in the primary care setting.
Diagnosis and Evaluation
The National Health and Nutrition Examination Survey found that approximately 8.7 percent of children eight to 15 years of age meet criteria for ADHD. The condition is classified into three subtypes, depending on the prevalence of specific behaviors: predominantly inattentive, predominantly hyperactive/impulsive, and combined types.
A comprehensive interview with parents and caregivers is key to the diagnosis of ADHD. It should include questions about current symptoms and the patient's medical, developmental, educational, family, and psychosocial histories. The diagnosis is based on a clinical picture of early onset and significant duration and pervasiveness of symptoms, and functional impairment. These symptoms can be elicited by use of a semistructured interview or questionnaire, with behavior rating scales completed by the parents, other caregivers, and school personnel.
As with many conditions, ADHD is rarely a singular diagnosis. In addition to evaluating for primary symptoms of ADHD as described in the Diagnostic and Statistical Manual of Mental Disorders (4th ed. or primary care manual), physicians should screen for other primary conditions and comorbidities (e.g., vision, hearing, or speech problems; learning disabilities; other psychiatric conditions; Table 1). Subspecialty consultation should be obtained if necessary.
Table 1. Differential Diagnosis and Assessment of Comorbidity in Children with Attention-Deficit/Hyperactivity Disorder

| Academic factors |
| Cognitive impairment |
| Giftedness |
| Learning disability |
| Other learning style variations and dysfunctions (e.g., memory or auditory discrimination problems) |
| Biomedical problems |
| Chromosomal abnormalities (e.g., fragile X syndrome) |
| Chronic illness |
| Iron deficiency |
| Metabolic/endocrine conditions (e.g., hypothyroidism) |
| Neurologic conditions (e.g., Tourette syndrome, seizure disorder) |
| Perinatal complications |
| Sensory impairment |
| Sleep disorder |
| Toxins or medications |
| Family and psychosocial problems |
| Abuse or neglect |
| Cultural factors |
| Disruptive home environment |
| Family stresses |
| Mismatch of behavioral style and environmental expectations |
| Parental psychopathology or chemical dependency |
| Social skills deficits |
| Psychiatric problems |
| Adjustment disorder |
| Anxiety disorder |
| Childhood mania or juvenile bipolar disorder |
| Depression or dysthymia |
| Developmentally normal variation |
| Oppositional defiant disorder or conduct disorder |
| Pervasive developmental disorders (e.g., autism) |
| Psychosis |
| Substance abuse |
| Speech and language problems |
| Apraxia |
| Central auditory processing disorder |
| Dysfluency |
| Expressive/receptive language disorder |
| Phonologic disorder |
Copyright 2010 by Institute for Clinical Systems Improvement. Adapted with permission.
If comorbid issues are not identified and addressed, they may complicate the patient's level of functional impairment and lead to higher morbidity with a poor prognosis. One way to evaluate for comorbidities is to use standardized screening instruments, such as the Child Behavior Checklist. However, it should be noted that this instrument is for screening purposes only, and should not be used to diagnose any specific condition.
Once ADHD has been diagnosed, the physician must determine whether it is the primary or secondary diagnosis. If an alternative primary diagnosis (e.g., anxiety disorder, oppositional defiant disorder, depression) is identified, the patient should be treated or referred as appropriate. If ADHD is the likely primary diagnosis but a comorbid condition is suspected, physicians may choose to begin treatment for ADHD while concurrently evaluating for the suspected comorbidity.
Management
Once ADHD is confirmed, physicians should counsel the child and the parents about the diagnosis. For the child, a developmentally appropriate explanation of ADHD may be helpful. Parents should be given information about neurologic mechanisms, common features of ADHD and how they relate to the child's previous and current problems, and expectations of the clinical course and intervention strategies. The importance of individual teacher selection each year should be emphasized. It is also important to provide specific teacher-focused information for the parents to share with school personnel.
The decision to treat ADHD with medication should be made with the parents after a discussion of the expected benefits and potential risks. Factors such as the child's age, severity of symptoms, and comorbidities should also be considered and may influence the choice of medication. Stimulant and nonstimulant medications are approved by the U.S. Food and Drug Administration for use in children with ADHD (Table 2). The use of stimulants should be avoided in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious cardiac problems that could put them at increased risk of sympathomimetic effects. A family and personal cardiovascular history should be obtained for all patients before initiating stimulant therapy. Findings from the history or physical examination that suggest cardiac disease may require evaluation by a cardiologist. The American Academy of Pediatrics recommends against performing routine electrocardiography or routine subspecialty cardiology evaluations before initiating stimulant therapy in children with ADHD.
Table 2. FDA-Approved Medications for Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents
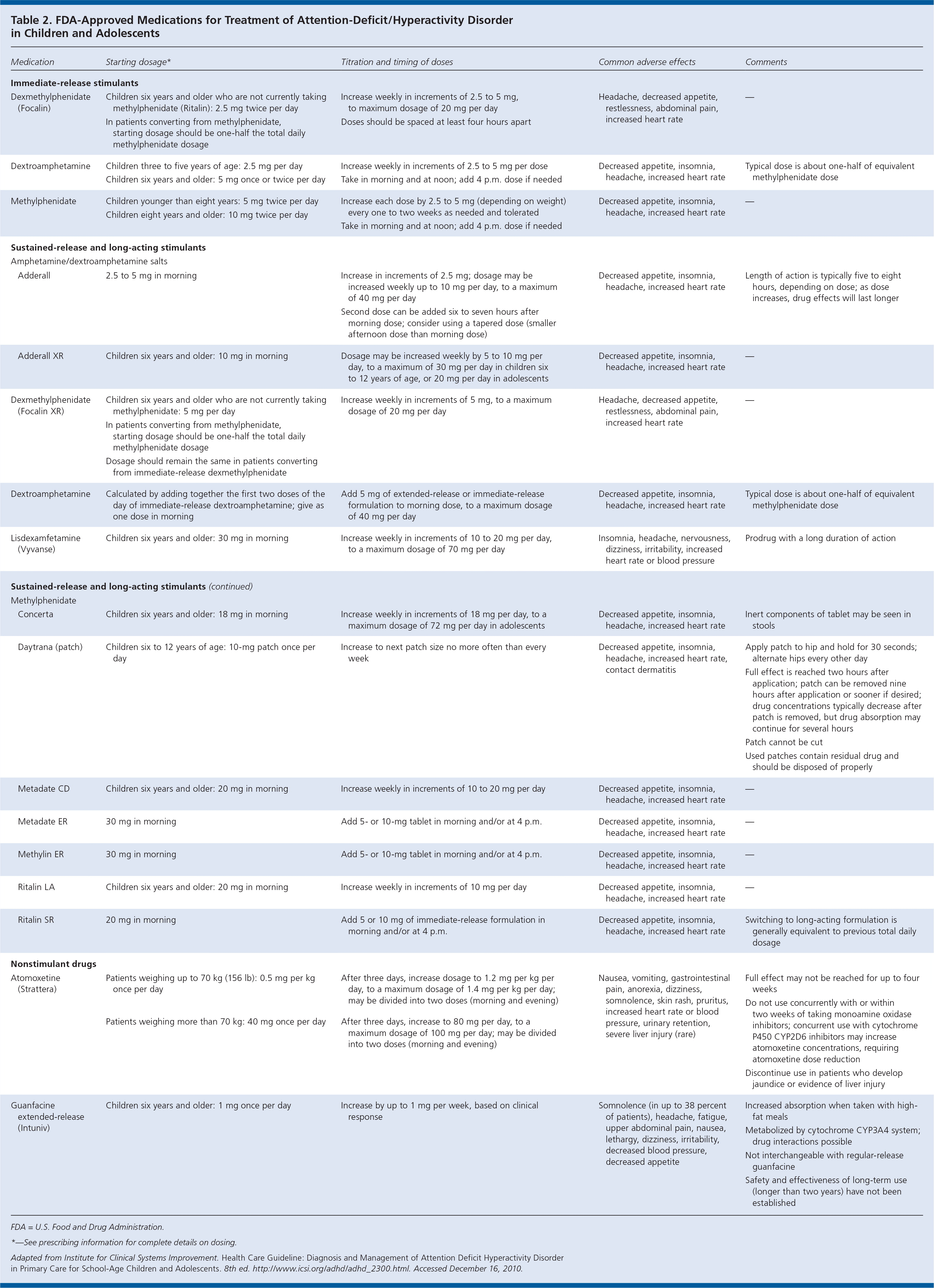
| Medication | Starting dosage* | Titration and timing of doses | Common adverse effects | Comments | |
|---|---|---|---|---|---|
| Immediate-release stimulants | |||||
| Dexmethylphenidate(Focalin) | Children six years and older who are not currently taking methylphenidate (Ritalin): 2.5 mg twice per day |
| Headache, decreased appetite, restlessness, abdominal pain, increased heart rate |
| |
| In patients converting from methylphenidate, starting dosage should be one-half the total daily methylphenidate dosage | |||||
| Dextroamphetamine | Children three to five years of age: 2.5 mg per day |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Children six years and older: 5 mg once or twice per day | |||||
| Methylphenidate | Children younger than eight years: 5 mg twice per day |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Children eight years and older: 10 mg twice per day | |||||
| Sustained-release and long-acting stimulants | |||||
| Amphetamine/dextroamphetamine salts | |||||
| Adderall | 2.5 to 5 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Adderall XR | Children six years and older: 10 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Dexmethylphenidate (Focalin XR) | Children six years and older who are not currently taking methylphenidate: 5 mg per day |
| Headache, decreased appetite, restlessness, abdominal pain, increased heart rate |
| |
| In patients converting from methylphenidate, starting dosage should be one-half the total daily methylphenidate dosage | |||||
| Dosage should remain the same in patients converting from immediate-release dexmethylphenidate | |||||
| Dextroamphetamine | Calculated by adding together the first two doses of the day of immediate-release dextroamphetamine; give as one dose in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Lisdexamfetamine (Vyvanse) | Children six years and older: 30 mg in morning |
| Insomnia, headache, nervousness, dizziness, irritability, increased heart rate or blood pressure |
| |
| Methylphenidate | |||||
| Concerta | Children six years and older: 18 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Daytrana (patch) | Children six to 12 years of age: 10-mg patch once per day |
| Decreased appetite, insomnia, headache, increased heart rate, contact dermatitis |
| |
| Metadate CD | Children six years and older: 20 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Metadate ER | 30 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Methylin ER | 30 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Ritalin LA | Children six years and older: 20 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Ritalin SR | 20 mg in morning |
| Decreased appetite, insomnia, headache, increased heart rate |
| |
| Nonstimulant drugs | |||||
| Atomoxetine (Strattera) | Patients weighing up to 70 kg (156 lb): 0.5 mg per kg once per day |
| Nausea, vomiting, gastrointestinal pain, anorexia, dizziness, somnolence, skin rash, pruritus, increased heart rate or blood pressure, urinary retention, severe liver injury (rare) |
| |
| Patients weighing more than 70 kg: 40 mg once per day |
| ||||
| Guanfacine extended-release (Intuniv) | Children six years and older: 1 mg once per day |
| Somnolence (in up to 38 percent of patients), headache, fatigue, upper abdominal pain, nausea, lethargy, dizziness, irritability, decreased blood pressure, decreased appetite |
| |
FDA = U.S. Food and Drug Administration.
*—See prescribing information for complete details on dosing.
Adapted from Institute for Clinical Systems Improvement. Health Care Guideline: Diagnosis and Management of Attention Deficit Hyperactivity Disorder in Primary Care for School-Age Children and Adolescents. 8th ed. http://www.icsi.org/adhd/adhd_2300.html. Accessed December 16, 2010.
When adequate trials of stimulants and nonstimulants are unsuccessful because of poor response or adverse effects, or if a comorbidity is present, alternative medications can be considered (Table 3, p. 768 ). Adverse effects of these medications may be more common and more serious than those associated with stimulants. In addition, there are fewer studies documenting their benefit and safety in children and adolescents. Occasionally a comorbid condition may warrant the use of alternative medications. In these cases, the primary symptoms should influence the medication decision.
Table 3. Alternative Medications for Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents
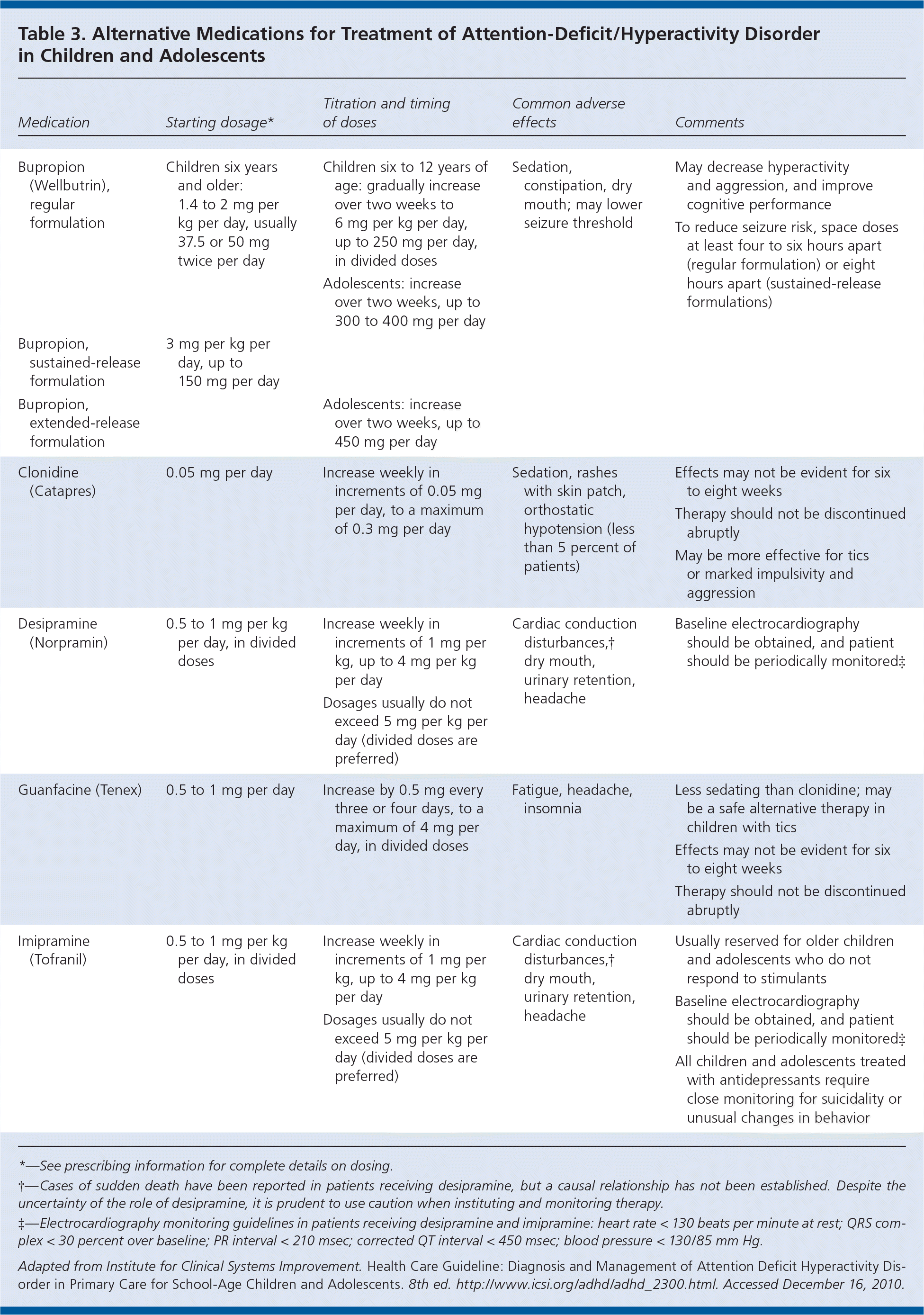
| Medication | Starting dosage* | Titration and timing of doses | Common adverse effects | Comments |
|---|---|---|---|---|
| Bupropion (Wellbutrin), regular formulation | Children six years and older: 1.4 to 2 mg per kg per day, usually 37.5 or 50 mg twice per day | Children six to 12 years of age: gradually increase over two weeks to 6 mg per kg per day, up to 250 mg per day, in divided doses | Sedation, constipation, dry mouth; may lower seizure threshold |
|
| Adolescents: increase over two weeks, up to 300 to 400 mg per day | ||||
| Bupropion, sustained-release formulation | 3 mg per kg per day, up to 150 mg per day | |||
| Bupropion, extended-release formulation | Adolescents: increase over two weeks, up to 450 mg per day | |||
| Clonidine (Catapres) | 0.05 mg per day | Increase weekly in increments of 0.05 mg per day, to a maximum of 0.3 mg per day | Sedation, rashes with skin patch, orthostatic hypotension (less than 5 percent of patients) |
|
| Desipramine (Norpramin) | 0.5 to 1 mg per kg per day, in divided doses | Increase weekly in increments of 1 mg per kg, up to 4 mg per kg per day | Cardiac conduction disturbances,† dry mouth, urinary retention, headache |
|
| Dosages usually do not exceed 5 mg per kg per day (divided doses are preferred) | ||||
| Guanfacine (Tenex) | 0.5 to 1 mg per day | Increase by 0.5 mg every three or four days, to a maximum of 4 mg per day, in divided doses | Fatigue, headache, insomnia |
|
| Imipramine (Tofranil) | 0.5 to 1 mg per kg per day, in divided doses | Increase weekly in increments of 1 mg per kg, up to 4 mg per kg per day | Cardiac conduction disturbances,† dry mouth, urinary retention, headache |
|
| Dosages usually do not exceed 5 mg per kg per day (divided doses are preferred) |
*—See prescribing information for complete details on dosing.
†—Cases of sudden death have been reported in patients receiving desipramine, but a causal relationship has not been established. Despite the uncertainty of the role of desipramine, it is prudent to use caution when instituting and monitoring therapy.
‡—Electrocardiography monitoring guidelines in patients receiving desipramine and imipramine: heart rate < 130 beats per minute at rest; QRS complex < 30 percent over baseline; PR interval < 210 msec; corrected QT interval < 450 msec; blood pressure < 130/85 mm Hg.
Adapted from Institute for Clinical Systems Improvement. Health Care Guideline: Diagnosis and Management of Attention Deficit Hyperactivity Disorder in Primary Care for School-Age Children and Adolescents. 8th ed. http://www.icsi.org/adhd/adhd_2300.html. Accessed December 16, 2010.
Although medication is the cornerstone of ADHD treatment, multimodal intervention may be needed for concomitant conditions and comorbidities. Primary care physicians are in a unique position to coordinate care.
Family-focused management strategies include ADHD support groups, advocacy groups, and parenting skills training. Parents should learn to provide a structured home environment, clear expectations, consistent responses, positive attention for appropriate behaviors, and appropriate consequences for maladaptive behaviors. These methods serve to give the child direction, goals, and limits in the hopes of improving compliance, increasing self-esteem, enhancing the parent-child relationship, and reducing tension within the home.
Children with ADHD may benefit from social skills training to improve peer relationships that are often negatively affected by ADHD symptoms (e.g., impulsivity). Cognitive behavior therapy also may be warranted. Study and organizational skills training should be offered in conjunction with curriculum intervention.
Neurofeedback, a form of biofeedback, has been promoted as an alternative therapy for ADHD. Neurofeedback uses electroencephalography biofeedback to teach children with ADHD how to self-regulate certain brain activity patterns and then to generalize these skills to daily life. Although early studies provided some evidence that neurofeedback may have some positive effects, these studies had significant methodologic problems that limit their usefulness. A recent randomized controlled trial showed that neurofeedback is superior to computerized attention skills training. Based on this evidence, neurofeedback may be a reasonable alternative to medication use, or may be used as part of a multimodal treatment program.

