Purpose
In AFP Journal Club, three presenters review an interesting journal article in a conversational manner. These articles involve “hot topics” that affect family physicians or “bust” commonly held medical myths. The presenters give their opinions about the clinical value of the individual study discussed. The opinions reflect the views of the presenters, not those of AFP or the AAFP.
Article
Cortunum S, Sørensen P, Jørgensen J. Determining the sensitivity of computed tomography scanning in early detection of subarachnoid hemorrhage. Neurosurgery. 2010;66(5):900–902.
Is CT sensitive enough to rule out SAH?
Bob: Emergency medicine dogma has stated that computed tomography (CT) is not sensitive enough to detect subtle subarachnoid hemorrhage (SAH), and that negative head CT must be followed by a lumbar puncture. As recently as 2008, researchers noted that four-slice CT had a 93 percent sensitivity for detecting SAH.1 Because 7 percent of SAH cases (one in 14) could be missed by CT, the mantra has been “head CT followed by lumbar puncture” when SAH is suspected.
This article attempts to make the argument that with the development of advanced generation CT machines, head CT is sensitive enough to detect all cases of SAH, and follow-up lumbar puncture is not required. Unfortunately, I believe this conclusion is wrong and dangerous.
What does this article say?
Bob: This retrospective study was performed in Denmark at a neurosurgical unit where the authors reviewed the charts of all patients with suspected SAH who were referred to the unit over five years. CT was obtained in all referred patients. If head CT was read as “normal,” a lumbar puncture was performed.
CT identified SAH in 295 of the 499 patients evaluated. Only one patient with negative CT results ultimately had a lumbar puncture indicating the presence of SAH. This patient had been experiencing a headache for more than four days. The remaining 203 patients had negative findings on CT and on lumbar puncture, resulting in a 99.7 percent sensitivity for CT (95% confidence interval, 98.1 to 99.9 percent). The authors conclude that CT is excellent for identifying SAH, and that follow-up lumbar puncture is not required for patients with symptoms lasting three days or less.
Should we believe this study?
Bob: No—retrospective studies are inherently problematic. First, they rely on accurate and complete medical record documentation (and we all know how good we are at that). Second, there must be unambiguous and unbiased extraction of the data from the medical records. Gilbert and colleagues laid out eight concise steps necessary for a retrospective review (Table 1).2 Unfortunately, this study misses many of these standard measures. The data abstractors were not identified, other than “two experienced members of the neurosurgical staff.” The study authors did not comment on the abstractors' training or whether they were blinded to the study hypothesis or monitored/tested for interrater agreement. How the data were abstracted also was not addressed. Even the basic question of which type of CT machine was used in the study was not clearly documented.
Table 1. Strategies to Improve Accuracy and Minimize Inconsistencies in Medical Chart Review

| Training |
| Train chart abstractors to perform their jobs. Describe the qualifications and training of the chart abstractors. Ideally, train abstractors before the study starts, using a set of “practice” medical charts. |
| Case selection |
| Use explicit protocols and describe the criteria for case selection or exclusion. |
| Definition of variables |
| Define important variables precisely. |
| Abstraction forms |
| Use standardized abstraction forms to guide data collection. Ensure uniform handling of data that are conflicting, ambiguous, missing, or unknown. |
| Meetings |
| Hold periodic meetings with chart abstractors and study coordinators to resolve disputes and review coding rules. |
| Monitoring |
| Monitor the performance of the chart abstractors. |
| Blinding |
| Blind chart reviewers to the etiologic relation being studied or the hypotheses being tested. If groups of patients are to be compared, the abstractor should be blinded to the patient's group assignment. |
| Testing of interrater agreement |
| A second reviewer should reabstract a sample of charts, blinded to the information obtained by the first correlation reviewer. Report a kappa statistic, intraclass coefficient, or other measure of agreement to assess interrater reliability of the data. |
Adapted with permission from Gilbert EH, Lowenstein SR, Kozol-McLain J, et al. Chart reviews in emergency medicine research: where are the methods? Ann Emerg Med. 1996;27(3):306.
However, this study's bigger problem is the incorporation of a type of selection bias known as spectrum bias. This phenomenon acknowledges that a diagnostic test's performance changes with different patient populations in different clinical settings. If we look at the results reported from this neurosurgical referral center, 59 percent of the patients evaluated had an SAH. Clearly, this referral population is far different from the population that arrives in most community hospitals. This study does not tell us if CT is sensitive enough in the typical community hospital setting, where the presentation may be less dramatic.
Andrea: I absolutely agree that you cannot extrapolate these results to the community hospital setting. And when we talk about the level of sensitivity we are willing to accept, we must remember what disease process we are talking about. For example, if a test is 93 percent sensitive for a self-limiting disease like influenza or pharyngitis, we would all find that acceptable. However, 93 percent sensitivity for identifying a life-threatening problem, such as SAH or myocardial infarction, is not acceptable. Because SAH often presents with a herald bleed before a large, fatal rupture, this is the point when the physician can truly save a patient's life—each bleed has approximately a 50 percent mortality rate. However, a herald bleed is often of small volume and is not detected by CT.
Mark: I am not sure why there is a movement to try to avoid the lumbar puncture. Yes, there is transient discomfort, and a small percentage of patients develop postdural puncture headaches, but these headaches can be managed with intravenous fluids, intravenous caffeine, or a blood patch.3 Are you willing to risk a patient's life because of the fear of a postprocedure headache?
Another strategy that has been recently advocated, but one I cannot support, is to follow the negative head CT with CT angiography.4 First, you are talking about a lot of radiation and the risk of contrast media–induced reactions with this strategy. Second, this recommendation is based on a mathematical model and has never been tested. Third, we know that 2 percent of a typical middle-aged population harbors asymptomatic intracranial aneurysms.5 If one were to follow this proposed strategy of negative head CT followed by CT angiography and an aneurysm is discovered, a lumbar puncture would have to be performed anyway to determine whether it is symptomatic. If the aneurysm is not bleeding and was found incidentally, you have another problem—what are you going to do with an asymptomatic aneurysm? The overwhelming number of these small aneurysms (smaller than 5 mm) will never rupture, but will likely result in significant patient anxiety and neurosurgical consultation. If the neurosurgeon elects to intervene, you can expect a 2 percent mortality rate and 11 percent morbidity.5
And a last note: when all the patients in this study with negative head CT underwent lumbar puncture, four of the remaining 204 patients were diagnosed with viral meningitis. The take-home message here is that the lumbar puncture gives you the answers you need in a patient with new, unexplained headache.
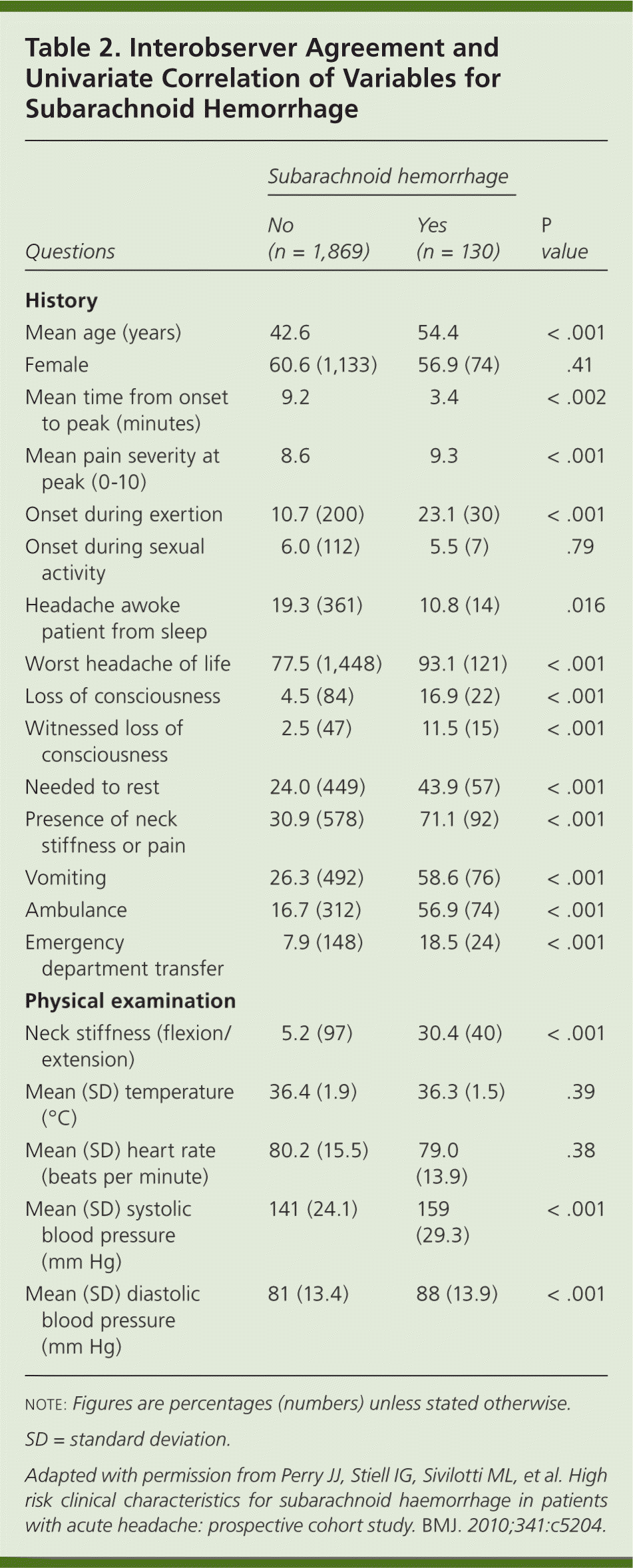
Andrea: When deciding whether to proceed with the head CT followed by lumbar puncture in a patient with a headache, you may ask yourself if you should even consider SAH in this patient's differential diagnosis. A recent study prospectively evaluated 1,999 patients with sudden severe headache peaking within one hour of onset. Twelve findings on history and three findings on physical examination were associated with SAH (Table 2).6 The authors suggest that if patients do not have any of these characteristics, the chance that the patient with a headache has an SAH is nil. A prospective study to assess the reliability of this suggestion is currently ongoing.
Table 2. Interobserver Agreement and Univariate Correlation of Variables for Subarachnoid Hemorrhage
| Questions | Subarachnoid hemorrhage | P value | |
|---|---|---|---|
| No (n = 1,869) | Yes (n = 130) | ||
| History | |||
| Mean age (years) | 42.6 | 54.4 | < .001 |
| Female | 60.6 (1,133) | 56.9 (74) | .41 |
| Mean time from onset to peak (minutes) | 9.2 | 3.4 | < .002 |
| Mean pain severity at peak (0–10) | 8.6 | 9.3 | < .001 |
| Onset during exertion | 10.7 (200) | 23.1 (30) | < .001 |
| Onset during sexual activity | 6.0 (112) | 5.5 (7) | .79 |
| Headache awoke patient from sleep | 19.3 (361) | 10.8 (14) | .016 |
| Worst headache of life | 77.5 (1,448) | 93.1 (121) | < .001 |
| Loss of consciousness | 4.5 (84) | 16.9 (22) | < .001 |
| Witnessed loss of consciousness | 2.5 (47) | 11.5 (15) | < .001 |
| Needed to rest | 24.0 (449) | 43.9 (57) | < .001 |
| Presence of neck stiffness or pain | 30.9 (578) | 71.1 (92) | < .001 |
| Vomiting | 26.3 (492) | 58.6 (76) | < .001 |
| Ambulance | 16.7 (312) | 56.9 (74) | < .001 |
| Emergency department transfer | 7.9 (148) | 18.5 (24) | < .001 |
| Physical examination | |||
| Neck stiffness (flexion/extension) | 5.2 (97) | 30.4 (40) | < .001 |
| Mean (SD) temperature (°C) | 36.4 (1.9) | 36.3 (1.5) | .39 |
| Mean (SD) heart rate (beats per minute) | 80.2 (15.5) | 79.0 (13.9) | .38 |
| Mean (SD) systolic blood pressure (mm Hg) | 141 (24.1) | 159 (29.3) | < .001 |
| Mean (SD) diastolic blood pressure (mm Hg) | 81 (13.4) | 88 (13.9) | < .001 |
note: Figures are percentages (numbers) unless stated otherwise.
SD = standard deviation.
Adapted with permission from Perry JJ, Stiell IG, Sivilotti ML, et al. High risk clinical characteristics for subarachnoid haemorrhage in patients with acute headache: prospective cohort study. BMJ . 2010;341:c5204.
Bob: The article Andrea is referring to brings home two points. One, the authors performed their study in six Canadian hospitals, and of the 1,999 patients with sudden severe headache, only 130 (6.5 percent) had SAH. This spectrum of patients better reflects what we see in community hospital settings. Two, of the 130 patients with SAH, the hemorrhage was identified on CT in 121 patients and on lumbar puncture in nine patients. The bottom line is that in this clinical setting, CT is only 93 percent sensitive for detecting SAH.6
What should the family physician do?
Bob: I do not believe you can abandon the “head CT followed by lumbar puncture” dogma in cases of suspected SAH. In my career, I have identified two cases of SAH in patients with negative head CT. I hate to think of the consequences if I had not done the lumbar puncture.
Andrea: I agree. I sense that one of the reasons everyone is trying to find a way around the lumbar puncture is perhaps the lack of experience with it. I see many young physicians who have done very few, if any, lumbar punctures before leaving residency. If you are uncomfortable doing lumbar punctures, consider doing it under ultrasound guidance. It is a technique that is easy and can be quickly learned.
Mark: And please avoid getting overly enamored with technology. Too many physicians have been sued because they based management decisions solely on the results of advanced imaging studies.
Main points
• In cases of suspected SAH, normal head CT should be followed by a lumbar puncture.
EBM Points
• Retrospective studies are generally suspect because they must rely on complete and accurate medical record documentation.
• Retrospective studies need to follow common, agreed-upon methods of data abstraction.
• A diagnostic test can perform differently in dissimilar patient populations. This is known as spectrum bias.

