Premenstrual syndrome is defined as recurrent moderate psychological and physical symptoms that occur during the luteal phase of menses and resolve with menstruation. It affects 20 to 32 percent of premenopausal women. Women with premenstrual dysphoric disorder experience affective or somatic symptoms that cause severe dysfunction in social or occupational realms. The disorder affects 3 to 8 percent of premenopausal women. Proposed etiologies include increased sensitivity to normal cycling levels of estrogen and progesterone, increased aldosterone and plasma renin activity, and neurotransmitter abnormalities, particularly serotonin. The Daily Record of Severity of Problems is one tool with which women may self-report the presence and severity of premenstrual symptoms that correlate with the criteria for premenstrual dysphoric disorder in the Diagnostic and Statistical Manual of Mental Disorders, 4th ed., text revision. Symptom relief is the goal for treatment of premenstrual syndrome and premenstrual dysphoric disorder. There is limited evidence to support the use of calcium, vitamin D, and vitamin B6 supplementation, and insufficient evidence to support cognitive behavior therapy. Serotonergic antidepressants (citalopram, escitalopram, fluoxetine, sertraline, venlafaxine) are first-line pharmacologic therapy.
Premenstrual syndrome (PMS) is clinically diagnosed if certain symptoms that impair some facet of a woman's life occur only during the luteal phase of the menstrual cycle (one week before menstruation), and if other diagnoses that may better explain the symptoms are excluded.1 Table 1 lists the physical, psychological, and behavioral symptoms associated with PMS and premenstrual dysphoric disorder (PMDD).2,3 With PMDD, more severe and debilitating affective symptoms impact the patient's relationships and social and occupational realms (Table 2).2 PMS and PMDD symptoms may recur with each ovulatory cycle until menopause, although the severity and frequency of different symptoms may vary over time.3,4 Duration of symptoms each month averages six days, with severity usually peaking anywhere from two days before to the first day of the menstrual flow.5 PMS and PMDD have been shown to negatively affect relationships, work attendance, productivity, and health care costs and utilization.6
SORT: KEY RECOMMENDATIONS FOR PRACTICE
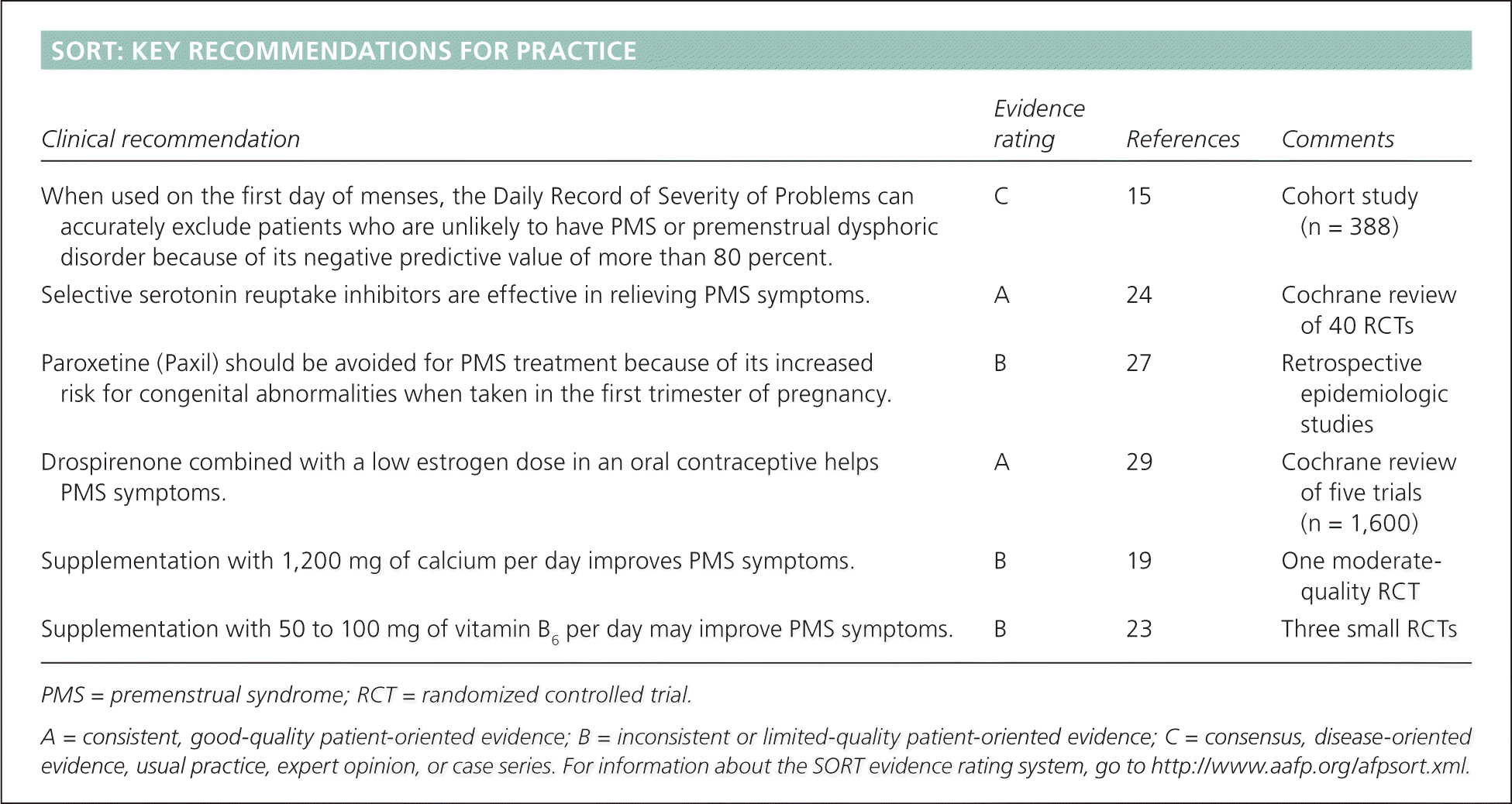
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| When used on the first day of menses, the Daily Record of Severity of Problems can accurately exclude patients who are unlikely to have PMS or premenstrual dysphoric disorder because of its negative predictive value of more than 80 percent. | C | 15 | Cohort study (n = 388) |
| Selective serotonin reuptake inhibitors are effective in relieving PMS symptoms. | A | 24 | Cochrane review of 40 RCTs |
| Paroxetine (Paxil) should be avoided for PMS treatment because of its increased risk for congenital abnormalities when taken in the first trimester of pregnancy. | B | 27 | Retrospective epidemiologic studies |
| Drospirenone combined with a low estrogen dose in an oral contraceptive helps PMS symptoms. | A | 29 | Cochrane review of five trials (n = 1,600) |
| Supplementation with 1,200 mg of calcium per day improves PMS symptoms. | B | 19 | One moderate-quality RCT |
| Supplementation with 50 to 100 mg of vitamin B6 per day may improve PMS symptoms. | B | 23 | Three small RCTs |
PMS = premenstrual syndrome; RCT = randomized controlled trial.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Table 1. Symptoms Associated with Premenstrual Syndrome and Premenstrual Dysphoric Disorder
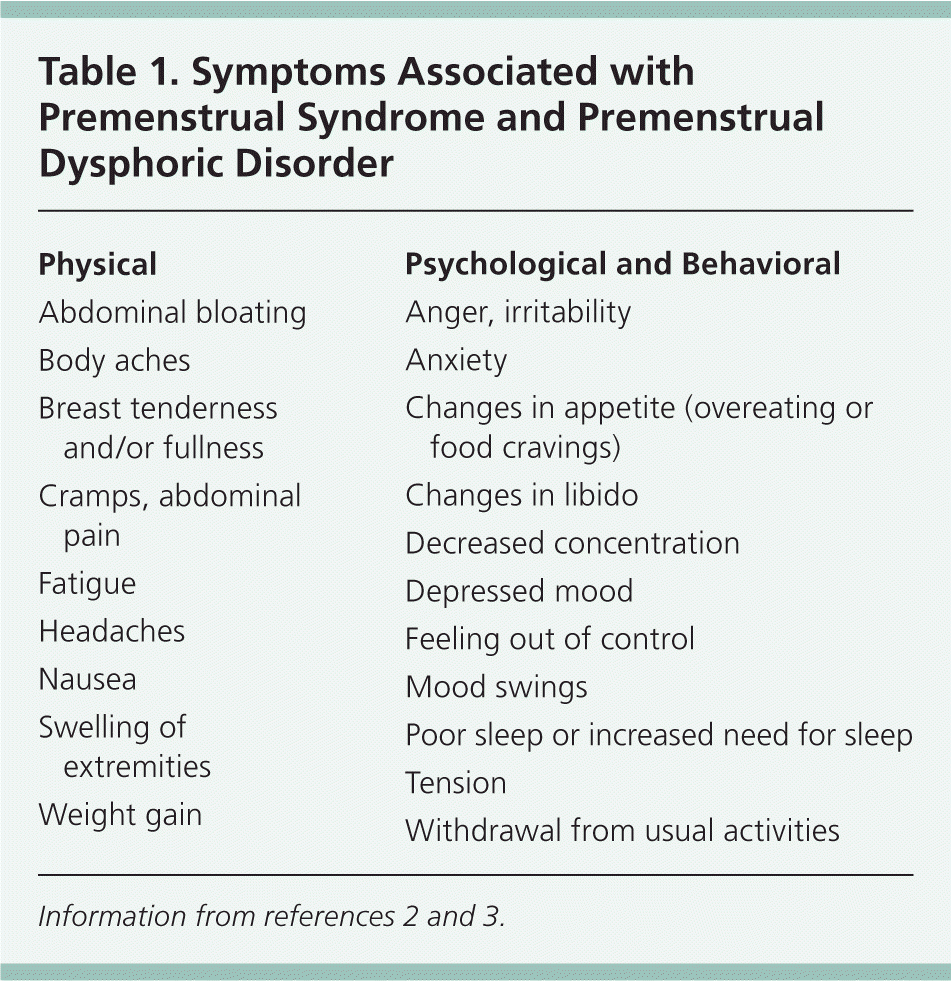
| Physical |
| Abdominal bloating |
| Body aches |
| Breast tenderness and/or fullness |
| Cramps, abdominal pain |
| Fatigue |
| Headaches |
| Nausea |
| Swelling of extremities |
| Weight gain |
| Psychological and Behavioral |
| Anger, irritability |
| Anxiety |
| Changes in appetite (overeating or food cravings) |
| Changes in libido |
| Decreased concentration |
| Depressed mood |
| Feeling out of control |
| Mood swings |
| Poor sleep or increased need for sleep |
| Tension |
| Withdrawal from usual activities |
Epidemiology
Up to 80 percent of women report one or more physical, psychological, or behavioral symptoms during the luteal phase of their menstrual cycle without experiencing substantial disruption to their daily functioning.5 PMS, in which mild to moderate symptoms affect some facet of the woman's life, occurs in 20 to 32 percent of premenopausal women; the more severe symptoms of PMDD affect 3 to 8 percent of premenopausal women.5 However, the prevalence of PMDD varies substantially among studies, likely because of different study definitions for PMDD. Initially, PMS and PMDD appeared to be limited to women in Western cultures, but more recent studies have demonstrated symptoms suggesting that PMS and PMDD occur at similar rates internationally.7–9
Etiology
The etiologies of PMS and PMDD are not definitive, but several theories are postulated. Women with PMS or PMDD appear to be more physiologically sensitive to and express more symptoms with normal cycling levels of estrogen and progesterone.10 In women whose normal cycles were blocked with administration of a gonadotropin-releasing hormone (GnRH) agonist and who were then given exogenous hormones, those with PMS experienced more symptoms of sadness, anxiety, irritability, bloating, and impaired function than those without PMS.10 Premenstrual symptoms may alter as women transition toward menopause; however, women who experienced PMS appear to have greater risk of menopausal symptoms associated with hormonal fluctuations (study participants were most affected by hot flashes, depressed mood, poor sleep, and decreased libido).11 Increases in aldosterone and plasma renin activity are the hypothetical mechanisms for PMS- and PMDD-associated fluid retention and bloating symptoms.12 Neurotransmitters, particularly serotonin and γ-amino-butyric acid, appear to be involved.6 Twin concordance studies suggest a genetic predisposition.13
Diagnosis
Clinical history is key to the diagnosis of PMS or PMDD. Other affective disorders such as depression and anxiety may have premenstrual cyclic worsening, but do not have the symptom-free interval during the mid-follicular phase (days 6 through 10 of the menstrual cycle) needed for the clinical diagnosis of PMS or PMDD. Some medical or gynecologic conditions such as hypothyroidism, anemia, endometriosis, or physiologic ovarian cysts may replicate physical symptoms of PMS. Physicians could check thyroid function or hemoglobin levels to rule out hypothyroidism and anemia, respectively.
The American College of Obstetricians and Gynecologists suggests diagnosing PMS based on prospective symptom diaries; many women will have non–luteal-phase symptoms consistent with other medical or psychological disorders.1 One tool to chart symptoms associated with PMS and PMDD is the Daily Record of Severity of Problems (Figure 1).14 Symptoms listed correspond to commonly used diagnostic criteria, such as the Diagnostic and Statistical Manual of Mental Disorders, 4th ed., text revision, for PMDD (Table 2).2 Because symptom charting for two cycles entails a significant time commitment for patients and a potential delay in diagnosis, one-time or retrospective assessments of symptoms may be helpful. Administering the Daily Record of Severity of Problems on the first day of menses is not sufficient for making a diagnosis (positive predictive value = 53.8 percent), but the tool can accurately exclude patients who are unlikely to have PMS or PMDD because of its negative predictive value (83.4 percent).15
Table 2. Research Criteria for Premenstrual Dysphoric Disorder

A. In most menstrual cycles during the past year, five (or more) of the following symptoms were present for most of the time during the last week of the luteal phase, began to remit within a few days after the onset of the follicular phase, and were absent in the week postmenses, with at least one of the symptoms being either 1, 2, 3, or 4:
|
| B. The disturbance markedly interferes with work or school or with usual social activities and relationships with others (e.g., avoidance of social activities, decreased productivity and efficiency at work or school). |
| C. The disturbance is not merely an exacerbation of the symptoms of another disorder, such as Major Depressive Disorder, Panic Disorder, Dysthymic Disorder, or a Personality Disorder (although it may be superimposed on any of these disorders). |
| D. Criteria A, B, and C must be confirmed by prospective daily ratings during at least two consecutive symptomatic cycles. (The diagnosis may be made provisionally prior to this confirmation.) |
Reprinted with permission from American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed., text revision. Washington, DC: American Psychiatric Association; 2000:774.
Treatment
Because the etiology of PMS and PMDD is not clear, symptom relief is the goal of treatment. General strategies address the proposed physiologic causes of symptoms, such as the ovulatory hormonal cyclicity of menstruation or the central nervous system neurotransmitters affecting mood (e.g., serotonin).
NONPHARMACOLOGIC
Lifestyle Modifications. Although some physicians recommend increasing exercise or decreasing intake of caffeine, salt, and refined sugar for PMS symptom relief, no current evidence substantiates those recommendations. A literature review found only one small randomized trial examining the effect of exercise on PMS symptoms that had been published in the past 25 years.16 Improved diet and exercise should be recommended for good health, but not as evidence-based treatment for PMS or PMDD.
However, dietary calcium and vitamin D intake may play a role in PMS symptoms. Women who had a high dietary calcium intake (mean: 1,283 mg per day) were 30 percent less likely to have PMS symptoms compared with women who had a low intake (mean: 529 mg per day; relative risk = 0.70; 95% confidence interval, 0.50 to 0.97).17 Women with high daily vitamin D intake were also less likely to have PMS symptoms,17,18 but whether vitamin D supplementation decreases these symptoms is unknown. Women (n = 466) whose calcium intake was supplemented with 1,200 mg of calcium carbonate per day for the duration of three menstrual cycles had significantly lower symptom scores for the second and third cycles of treatment compared with placebo (48 versus 30 percent reduction, respectively).19 Supplementation or high intake of dietary calcium and vitamin D may be considered for symptom relief in women with PMS or PMDD.
Cognitive Behavior Therapy. Cognitive behavior therapy uses psychotherapy techniques that focus on modifying problematic thoughts, emotions, and behaviors. It appears to be effective for other affective and somatic disorders such as anxiety and pain; thus, it theoretically could be useful for PMS treatment. However, a 2008 systematic review found a dearth of evidence for its use in treating PMS and PMDD; the review included only seven small randomized controlled trials (RCTs), all with large attrition rates.20
Complementary and Alternative Therapies. A 2009 systematic review found 62 studies of herbal supplements, vitamins, and minerals with published claims for PMS symptom relief, only 10 of which were RCTs.21 The review found that data are insufficient to evaluate the effectiveness of ginkgo, saffron, St. John's wort, soy, or vitamin E, because only one study existed for each. However, results suggested that chasteberry and vitamin B6 may reduce PMS symptoms.21
Chasteberry (Vitex agnus-castus, also known as chaste tree) is a small tree or shrub grown in southern Europe. Women in one RCT (n = 170) treated with 20 mg of chasteberry per day were more likely to have a decrease in PMS symptoms (irritability, mood swings, anger, headache, breast fullness) of more than 50 percent compared with placebo (52 versus 24 percent; P = .001).22 No patients withdrew from the study because of adverse effects.22
One systematic review of nine RCTs suggests pyridoxine (vitamin B6) supplementation relieved PMS symptoms by more than twofold compared with placebo (odds ratio = 2.32; 95% confidence interval, 1.95 to 2.54).23 Because doses higher than 100 mg showed no greater response than doses below 100 mg,23 and high doses of pyridoxine (greater than 300 mg) may be associated with peripheral neuropathy, moderate-dosage (i.e., 50 to 100 mg per day) vitamin B6 supplementation could be considered for relieving PMS symptoms and premenstrual depression with minimal risks of adverse effects.
PHARMACOLOGIC
Serotonergic Antidepressants. Medications affecting serotonin are first-line pharmacologic treatments for severe PMS or PMDD.1,24 The selective serotonin reuptake inhibitors (SSRIs) citalopram (Celexa), escitalopram (Lexapro), fluoxetine (Prozac), and sertraline (Zoloft)—taken daily or only during the luteal phase of menstruation—significantly decrease physical and psychological symptoms of PMS compared with placebo.24 In a study of PMDD treatment with the serotonin-norepinephrine reuptake inhibitor (SNRI) venlafaxine (Effexor), symptom scores were reduced by at least one-half in 60 percent of participants treated with venlafaxine (50 to 200 mg per day) compared with 35 percent of participants in the placebo group; 80 percent of the PMS symptom reduction with venlafaxine occurred within the first month of treatment.25 SSRIs and SNRIs may need to be administered for three to four weeks to affect symptoms of depression; PMS symptoms, however, appear to improve more rapidly.26 Daily use of an SSRI or SNRI with an increased dose during the luteal phase, especially if PMS symptoms are comorbid with major depression or generalized anxiety, is a reasonable alternative.26 Several SSRIs and SNRIs are available in generic forms that are relatively inexpensive. Nausea, insomnia, headache, and decreased libido are common adverse effects of SSRIs and SNRIs, and the benefit of symptom relief must be balanced against these potential effects.
In 2005, the U.S. Food and Drug Administration listed paroxetine (Paxil) as pregnancy category D based on studies demonstrating increased risk of major congenital heart defects from first-trimester exposure.27 Therefore, physicians should avoid prescribing paroxetine to women of childbearing age without reliable contraception or sterilization.
Oral Contraceptives. Few data support the effectiveness of oral contraceptives in treating PMS.1 A small RCT of a triphasic combined oral contraceptive (35 mcg of ethinyl estradiol and 0.5 mg, 1.0 mg, 0.5 mg of norethindrone weekly) versus placebo showed improvement in premenstrual breast tenderness and bloating, but not in mood symptoms.28 Combined oral contraceptives that include drospirenone, a spironolactone (Aldactone) derivative, appear to improve PMS and PMDD symptoms compared with placebo.29 Twenty-four days of drospirenone, 3 mg/ethinyl estradiol, 20 mcg (Yasmin) followed by four days of placebo improved mood and physical symptoms in women with PMDD,30 whereas the same doses of drospirenone/ethinyl estradiol for 21 days followed by seven days of placebo over three cycles showed no significant improvement in mood.31 Theoretically, the placebo week of oral contraceptives may allow ovarian hormonal function to initiate, causing PMS and PMDD symptoms to occur. Therefore, suppressing ovarian function with continuous oral contraceptive use may ameliorate symptoms. Use of drospirenone, 3 mg/ethinyl estradiol, 30 mcg for 42 to 168 days decreased physical symptoms of PMS such as edema, breast tenderness, and bloating compared with a standard 28-day cycle.32 Other oral contraceptive formulations do not have recently published studies that specifically address PMS symptoms.
GnRH Agonists. Because they suppress ovarian function, the GnRH agonists goserelin (Zoladex), histrelin (Vantas), leuprolide (Lupron), and nafarelin (Synarel) have been tried off-label to reduce severe physical symptoms of PMS and PMDD.33 However, adverse effects, especially hot flashes and decreased bone density, limit their use to only a few months.1 Estrogen can be added back, but this may cause PMS and PMDD symptoms to recur.10 Because of these limitations and their substantial cost, GnRH agonists do not appear to be appropriate agents for the conventional treatment of PMS and PMDD.
Other Medications. Irritability and anxiety are common symptoms in women with PMS or PMDD. For patients with anxiety symptoms, physicians may consider treatment with 0.25 mg of alprazolam (Xanax) intermittently up to three or four times per day during the luteal phase, but its use should be limited to carefully selected patients because of its dependence, tolerance, and abuse potential.34 When used continuously, buspirone (Buspar) decreased only irritability and not physical symptoms of PMS compared with placebo (83 versus 54 percent decrease in visual analog scale from baseline, respectively; P = .03).35 Spironolactone, the potassium-sparing diuretic with antiandrogenic effects, significantly improved symptoms of breast tenderness, bloating, weight gain, and depressed mood compared with placebo, and physicians may consider it a pharmacologic option for treating PMS symptoms.36
Data Sources: A PubMed search was done using the key terms premenstrual syndrome and premenstrual dysphoric disorder. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Cochrane Database of Systematic Reviews, Essential Evidence Plus, and Clinical Evidence 2009 Search date: April 2, 2010.

