The incidence of nephrolithiasis (kidney stones) is rising worldwide, especially in women and with increasing age. Kidney stones are associated with chronic kidney disease. Preventing recurrence is largely specific to the type of stone (e.g., calcium oxalate, calcium phosphate, cystine, struvite [magnesium ammonium phosphate]), and uric acid stones); however, even when the stone cannot be retrieved, urine pH and 24-hour urine assessment provide information about stone-forming factors that can guide prevention. Medications, such as protease inhibitors, antibiotics, and some diuretics, increase the risk of some types of kidney stones, and patients should be counseled about the risks of using these medications. Managing diet, medication use, and nutrient intake can help prevent the formation of kidney stones. Obesity increases the risk of kidney stones. However, weight loss could undermine prevention of kidney stones if associated with a high animal protein intake, laxative abuse, rapid loss of lean tissue, or poor hydration. For prevention of calcium oxalate, cystine, and uric acid stones, urine should be alkalinized by eating a diet high in fruits and vegetables, taking supplemental or prescription citrate, or drinking alkaline mineral waters. For prevention of calcium phosphate and struvite stones, urine should be acidified; cranberry juice or betaine can lower urine pH. Antispasmodic medications, ureteroscopy, and metabolic testing are increasingly being used to augment fluid and pain medications in the acute management of kidney stones.
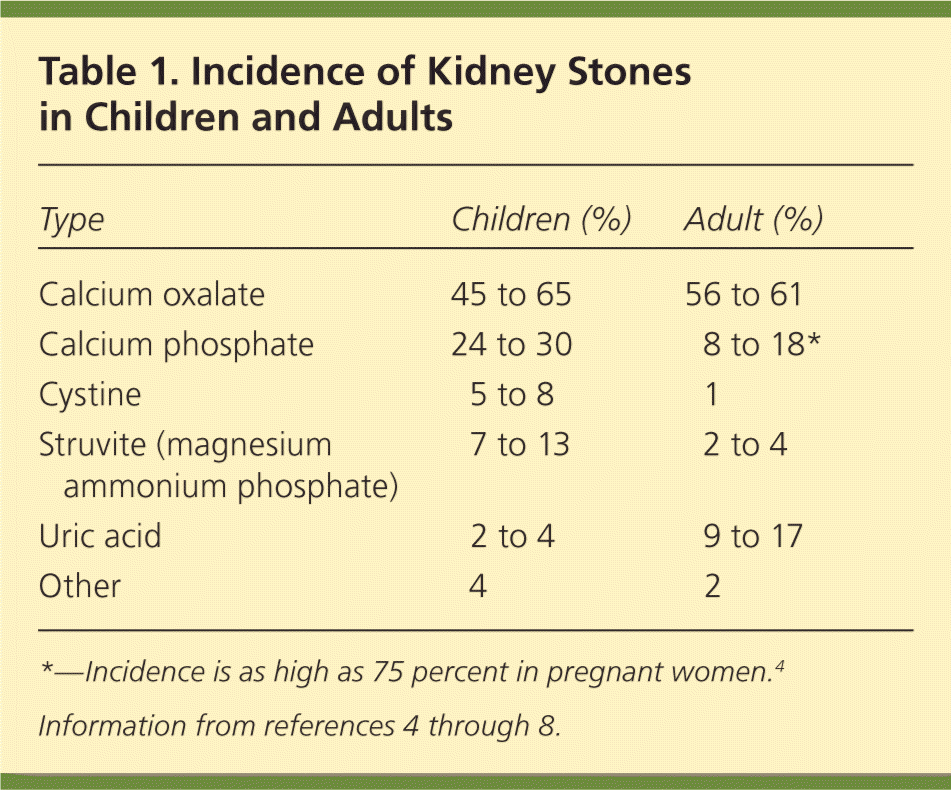
The prevalence of nephrolithiasis (kidney stones) is increasing in women and with increasing age. The risk of developing kidney stones is 10 to 15 percent in the United States, although this number is trending higher.1–3 Table 1 includes rates of different types of kidney stones in children and adults.4–8 Contributing risk factors for kidney stones are obesity, insulin resistance, gastrointestinal pathology, living in warmer climates, and certain dietary patterns and medications.2,9
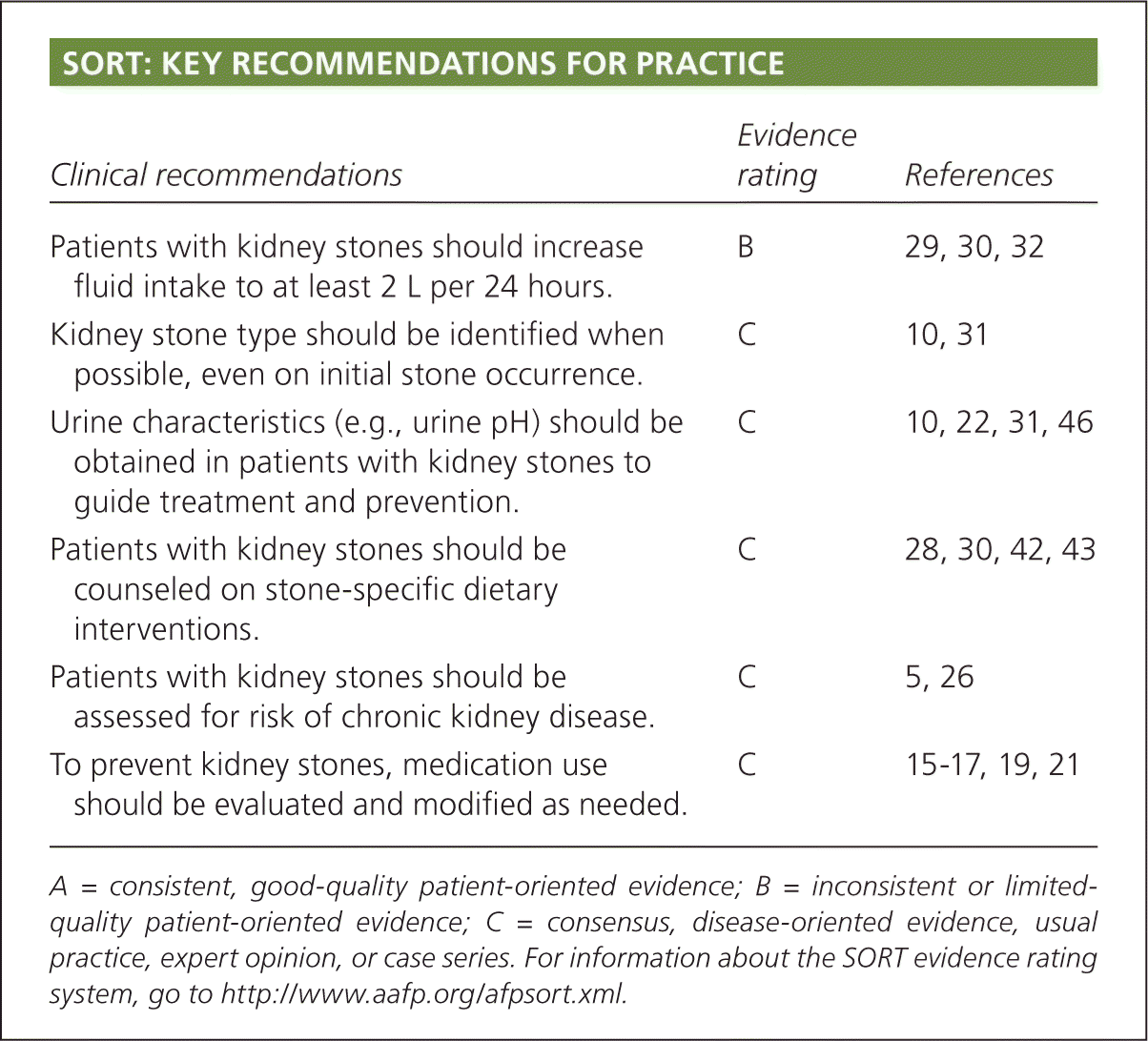
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendations | Evidence rating | References |
|---|---|---|
| Patients with kidney stones should increase fluid intake to at least 2 L per 24 hours. | B | 29, 30, 32 |
| Kidney stone type should be identified when possible, even on initial stone occurrence. | C | 10, 31 |
| Urine characteristics (e.g., urine pH) should be obtained in patients with kidney stones to guide treatment and prevention. | C | 10, 22, 31, 46 |
| Patients with kidney stones should be counseled on stone-specific dietary interventions. | C | 28, 30, 42, 43 |
| Patients with kidney stones should be assessed for risk of chronic kidney disease. | C | 5, 26 |
| To prevent kidney stones, medication use should be evaluated and modified as needed. | C | 15–17, 19, 21 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Table 1. Incidence of Kidney Stones in Children and Adults
Acute Diagnosis and Management
CLINICAL PRESENTATION
The characteristic cramping and intermittent abdominal and flank pain occur as kidney stones travel within the urinary tract. The pain is often accompanied by hematuria, nausea or vomiting, and malaise; fever and chills may also be present. However, stones in the renal pelvis may be asymptomatic. The differential diagnosis includes infections in the urinary tract or abdomen, malignancies, and musculoskeletal inflammation or spasm (Table 2).9
Table 2. Differential Diagnosis of Urinary Calculi

| Clinical clues | Suggested diagnoses | |
|---|---|---|
| Dysuria | Urinary tract infection, interstitial cystitis (pelvic pain syndrome), vaginitis, prostatitis | |
| Fevers, chills | Nonspecific response to infection or inflammation | |
| Frequency | High fluid intake, urinary tract infection, bladder spasms, benign prostatic hyperplasia, hyperglycemia | |
| Hematuria | Kidney stones, uroepithelial or prostatic tumors, urinary tract infection, renal glomerular disease | |
| Nausea, vomiting | Nonspecific response to pain, intestinal or urinary obstruction, gastrointestinal disease | |
| Pain and tenderness | ||
| Abdominal | Kidney stones, gastrointestinal disease | |
| Flank | Kidney stones, musculoskeletal inflammation or spasm, referred pain from gallbladder (on right side), pyelonephritis | |
| Groin or pelvic | Kidney or bladder stones, urethritis, prostatitis, vaginitis, pelvic inflammatory disease, pelvic pain syndrome | |
Adapted from Pietrow PK, Karellas ME. Medical management of common urinary calculi. Am Fam Physician. 2006;74(1):88.
DIAGNOSTIC WORKUP
The initial workup of a patient with suspected kidney stones (Figure 1) should include urinalysis for blood. Urine cultures are crucial if the patient is febrile or leukocytes are detected in the urine. Diagnosis is sometimes made by visualizing a stone on a plain radiograph. Uric acid stones and stones associated with protease inhibitor use may not be visible on a radiograph. Ultrasonography or spiral computed tomography can detect all types of kidney stones and may be necessary if the diagnosis is in question.
Figure 1. Diagnosis and Management of Acute Kidney Stones
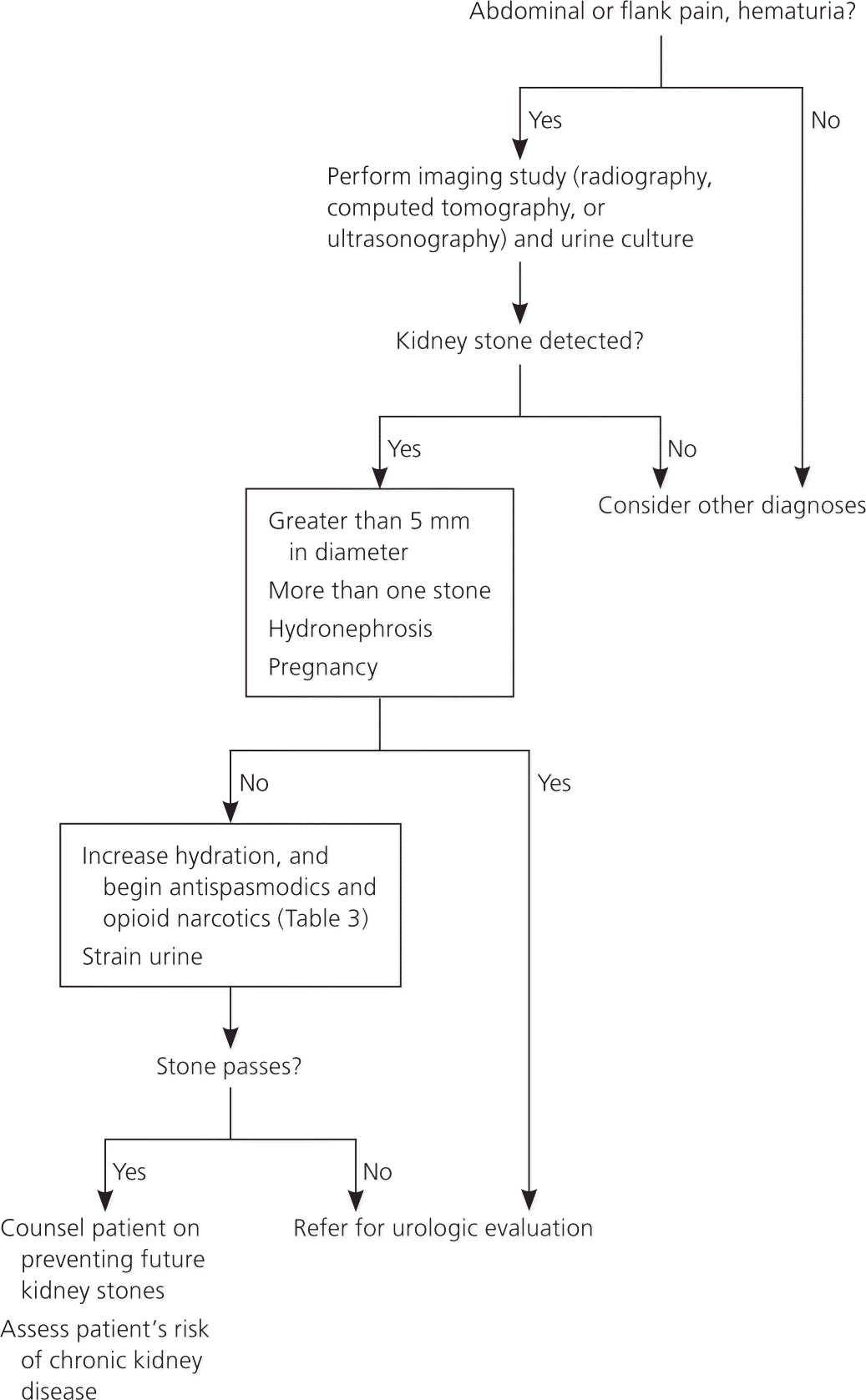
Algorithm for the diagnosis and management of acute kidney stones.
Referral to a urologist is warranted when more than one stone is present, symptoms worsen with fever, renal function is impaired, stone passage is prolonged, hydronephrosis is diagnosed based on imaging findings, the patient is pregnant, or the stone is larger than 5 mm in diameter as measured using computed tomography or ultrasonography. Smaller stones pass spontaneously in 90 percent of patients.10 Urologists are increasingly using ureteroscopy to remove stones and evaluate for changes in the uroepithelial lining.11 Interstitial deposits containing calcium oxalate (Randall plaques) are visible on ureteroscopy as whitish deposits.12
MANAGEMENT
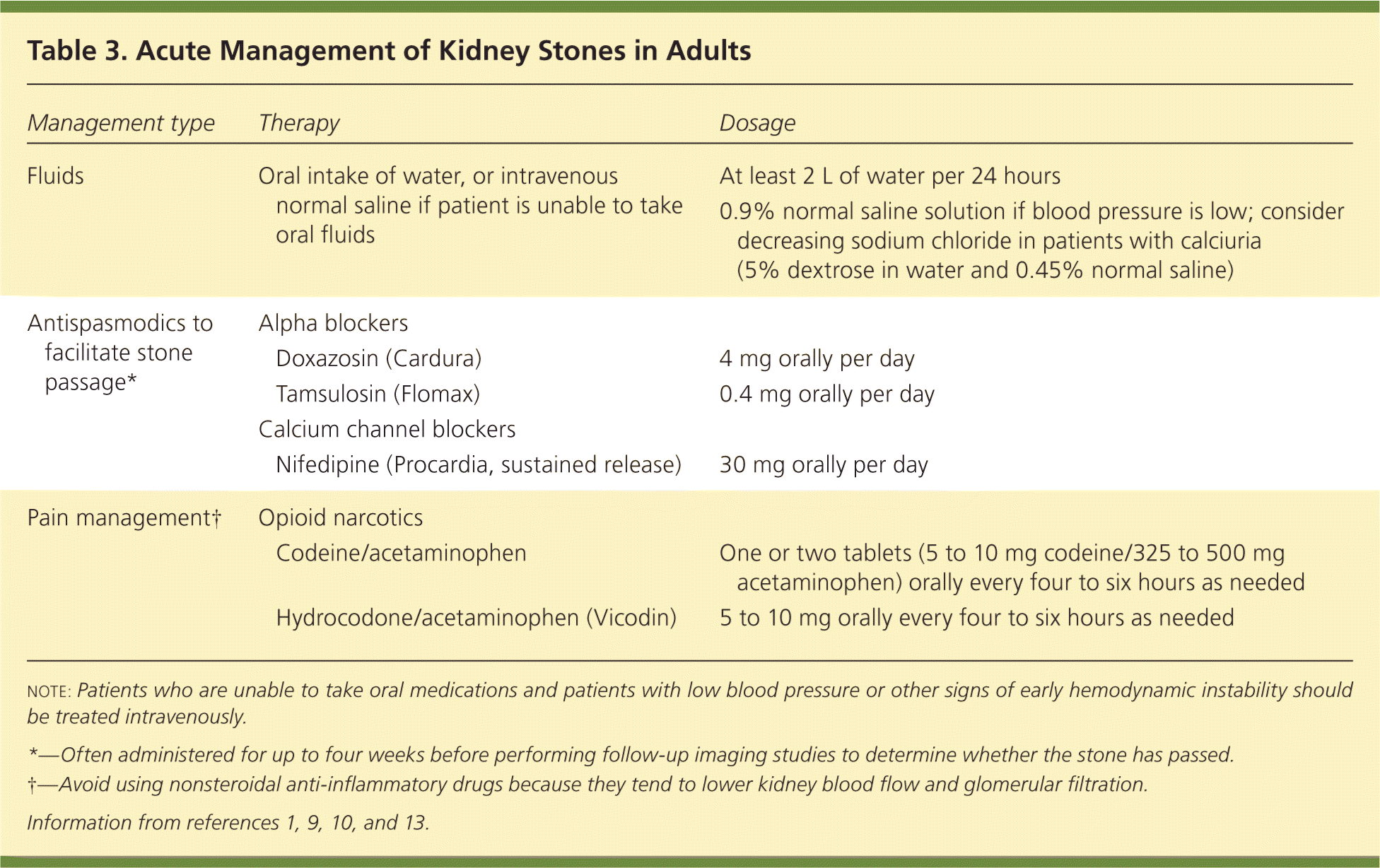
Oral hydration and pain management are part of the acute treatment of all stone types (Table 31,9,10,13 and Figure 1). For stones measuring 10 mm or less, antispasmodics such as calcium channel blockers and alpha blockers relax the smooth muscle of the ureters and have been shown to hasten stone passage by five to seven days.10 Coadministration of oral corticosteroids leads to little or no improvement in outcomes.14 Patients who are unable to take oral fluids or medications and patients with low blood pressure or other signs of early hemodynamic instability should be treated intravenously. If signs of possible infection are present (e.g., fever, pyuria), initial management should include empiric antibiotics that cover gram-negative bacilli (e.g., Enterobacteriaceae species) and gram-positive cocci (e.g., staphylococci, enterococci) according to local susceptibility patterns. Referral to a urologist should be expedited if the patient also shows radiologic evidence of obstruction (hydronephrosis).
Table 3. Acute Management of Kidney Stones in Adults
| Management type | Therapy | Dosage | |
|---|---|---|---|
| Fluids | Oral intake of water, or intravenous normal saline if patient is unable to take oral fluids | At least 2 L of water per 24 hours | |
| 0.9% normal saline solution if blood pressure is low; consider decreasing sodium chloride in patients with calciuria (5% dextrose in water and 0.45% normal saline) | |||
| Antispasmodics to facilitate stone passage* | Alpha blockers | ||
| Doxazosin (Cardura) | 4 mg orally per day | ||
| Tamsulosin (Flomax) | 0.4 mg orally per day | ||
| Calcium channel blockers | |||
| Nifedipine (Procardia, sustained release) | 30 mg orally per day | ||
| Pain management† | Opioid narcotics | ||
| Codeine/acetaminophen | One or two tablets (5 to 10 mg codeine/325 to 500 mg acetaminophen) orally every four to six hours as needed | ||
| Hydrocodone/acetaminophen (Vicodin) | 5 to 10 mg orally every four to six hours as needed | ||
note: Patients who are unable to take oral medications and patients with low blood pressure or other signs of early hemodynamic instability should be treated intravenously.
*—Often administered for up to four weeks before performing follow-up imaging studies to determine whether the stone has passed.
†—Avoid using nonsteroidal anti-inflammatory drugs because they tend to lower kidney blood flow and glomerular filtration.
Further Evaluation
Further evaluation identifies modifiable risk factors and guides individualized treatment and prevention. The medical history should identify conditions associated with increased risk of kidney stones (e.g., inflammatory bowel disease, bowel surgery, gout, diabetes mellitus, obesity or recent changes in weight, metabolic syndromes, hyperparathyroidism-associated conditions, frequent urinary tract infections, chronic kidney disease).15 A family medical history should also be obtained.
A medication history establishes temporal associations; identifies medications recently discontinued, off-label use of medications, and use of herbal preparations and supplements; and screens for illicit drug use. Medications contribute to kidney stones (Table 4) through various mechanisms by forming urine crystals and altering urine characteristics (e.g., changing urine pH, reducing urine volume).16–23 For example, carbonic anhydrase inhibitors contribute to calcium phosphate stone formation by causing a mild systemic acidosis and paradoxically high urine pH, hypercalciuria, and low urine citrate.22,23 Some antibiotics may increase urine oxalate by reducing the intestinal bacteria that break down oxalate.
Table 4. Medications Associated with Kidney Stone Formation
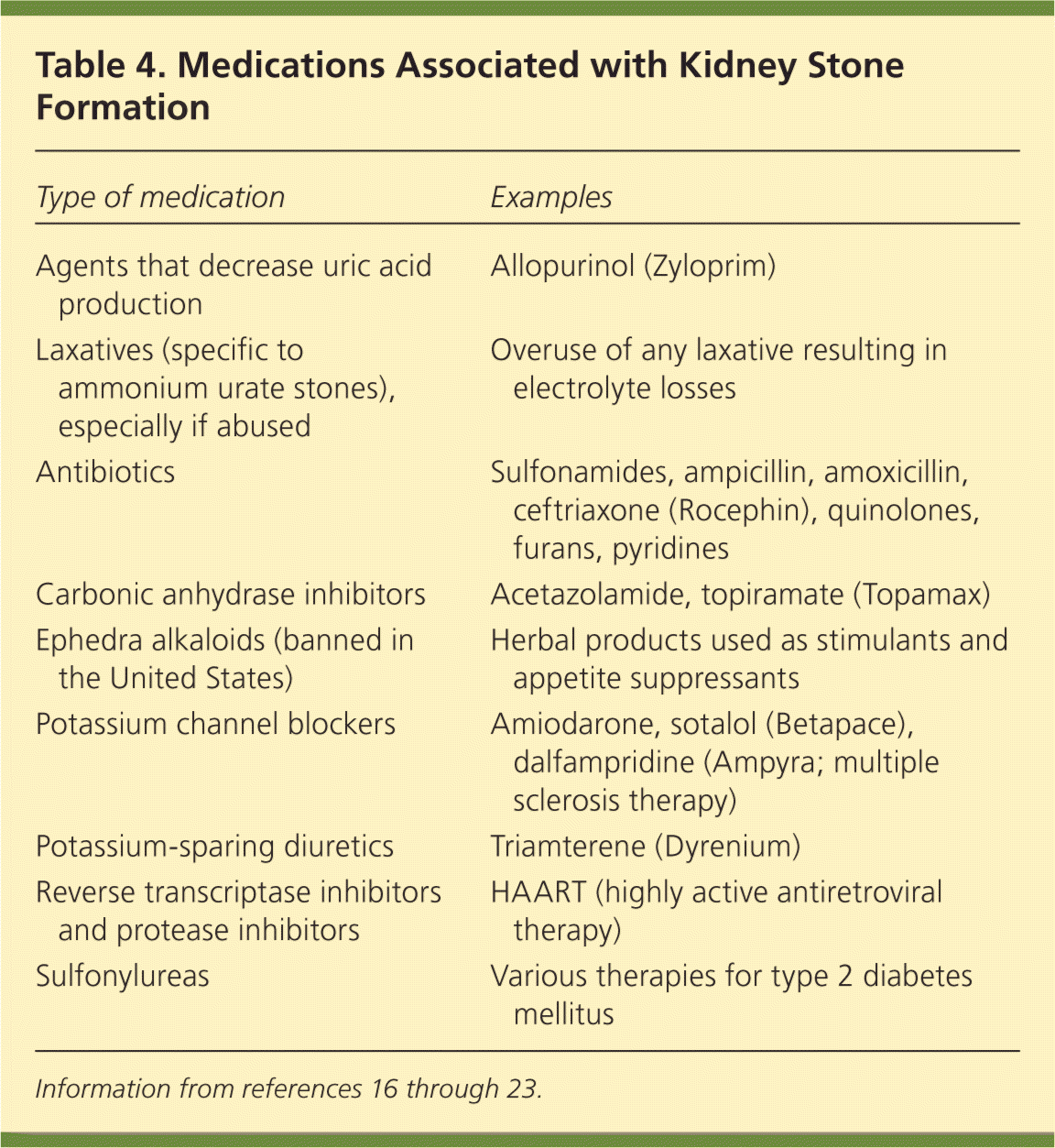
| Type of medication | Examples |
|---|---|
| Agents that decrease uric acid production [ | Allopurinol (Zyloprim) |
| Laxatives (specific to ammonium urate stones), especially if abused | Overuse of any laxative resulting in electrolyte losses |
| Antibiotics | Sulfonamides, ampicillin, amoxicillin, ceftriaxone (Rocephin), quinolones, furans, pyridines |
| Carbonic anhydrase inhibitors | Acetazolamide, topiramate (Topamax) |
| Ephedra alkaloids (banned in the United States) | Herbal products used as stimulants and appetite suppressants |
| Potassium channel blockers | Amiodarone, sotalol (Betapace), dalfampridine (Ampyra; multiple sclerosis therapy) |
| Potassium-sparing diuretics | Triamterene (Dyrenium) |
| Reverse transcriptase inhibitors and protease inhibitors | HAART (highly active antiretroviral therapy) |
| Sulfonylureas | Various therapies for type 2 diabetes mellitus |
Management and prevention are largely specific to the type of kidney stone. However, stones often pass without being retrieved, even when urine is strained. With the increasing incidence and prevalence of kidney stones and chronic kidney disease, further evaluation after a first kidney stone may be beneficial in all patients.24 Risk factors for kidney stone formation can often be assessed even after the stone has passed, through urine and blood chemistry.
Urine pH is an important factor in the production of kidney stones. Uric acid, cystine, and calcium oxalate stones tend to form in acidic urine, whereas struvite (magnesium ammonium phosphate) and calcium phosphate stones form in alkaline urine. A 24-hour urine collection can be analyzed for calcium, phosphorus, magnesium, uric acid, and oxalate to determine stone composition, and for the stone-inhibiting factors citrate and phytate. Urine calcium excretion is another important risk factor for stone formation and is elevated with increased diet acid loads, increased salt intake, and both inadequate and excessive vitamin D levels. Elevated serum calcium levels may suggest primary hyperparathyroidism as the cause of calcium stone formation.
Kidney stones are a risk factor for chronic kidney disease and progression to end-stage renal disease. 5 Persons with kidney stones are more likely to have traditional risk factors for chronic kidney disease (e.g., hypertension, preexisting kidney disease, diabetes, proteinuria, albuminuria), as well as non-traditional factors (e.g., interstitial nephritis, chronic pyelonephritis, female sex). 25 [ corrected] The American Society of Nephrology suggests referral to a nephrologist if the estimated glomerular filtration rate is 60 mL per minute per 1.73 m2 or less (stage 3 chronic kidney disease) or if macroalbuminuria is present.26
Special Considerations
CHILDREN
More children are developing kidney stones, which is attributed to the corresponding rise in diabetes, obesity, and hypertension in this population.2,3 Because increasing age is a risk factor for kidney stones, adolescents are more likely to form stones than younger children. The underlying causes and resulting treatments differ in children and adults. Children with kidney stones are more likely to have anatomic and metabolic abnormalities,3 increased urinary calcium excretion, decreased urinary oxalate and citrate excretion, and much higher urinary calcium oxalate saturations than children with no history of kidney stones.2 Children with cystinuria and other hereditary forms of kidney stones are at increased risk of decline in renal function compared with age-matched controls, although progression to end-stage renal disease is uncommon.2
PREGNANT WOMEN
Pregnant women are twice as likely to have calcium phosphate stones compared with age-matched nonpregnant women, and are two to three times more likely to have calcium phosphate stones than oxalate stones.4 The incidence of kidney stones during pregnancy increases in the second and third trimesters. Women have an increased glomerular filtration rate and higher urinary calcium excretion throughout pregnancy, with higher urine pH in the second and third trimesters, which may predispose them to calcium phosphate stones. Ultrasonography is considered the imaging modality of choice in pregnant women. Kidney stones during pregnancy increase the risk of urinary tract infections, and pregnant women with renal colic have nearly double the risk of pre-term delivery compared with women who do not have kidney stones.27
Prevention
Measures to prevent kidney stones include dietary modifications, nutritional supplements, and medications, depending on the specific type of kidney stone and urine characteristics (Table 5).13,28–33 General concepts on prevention are presented below.
Table 5. Recommendations for the Management of Kidney Stones Based on Stone Type and Urine Properties
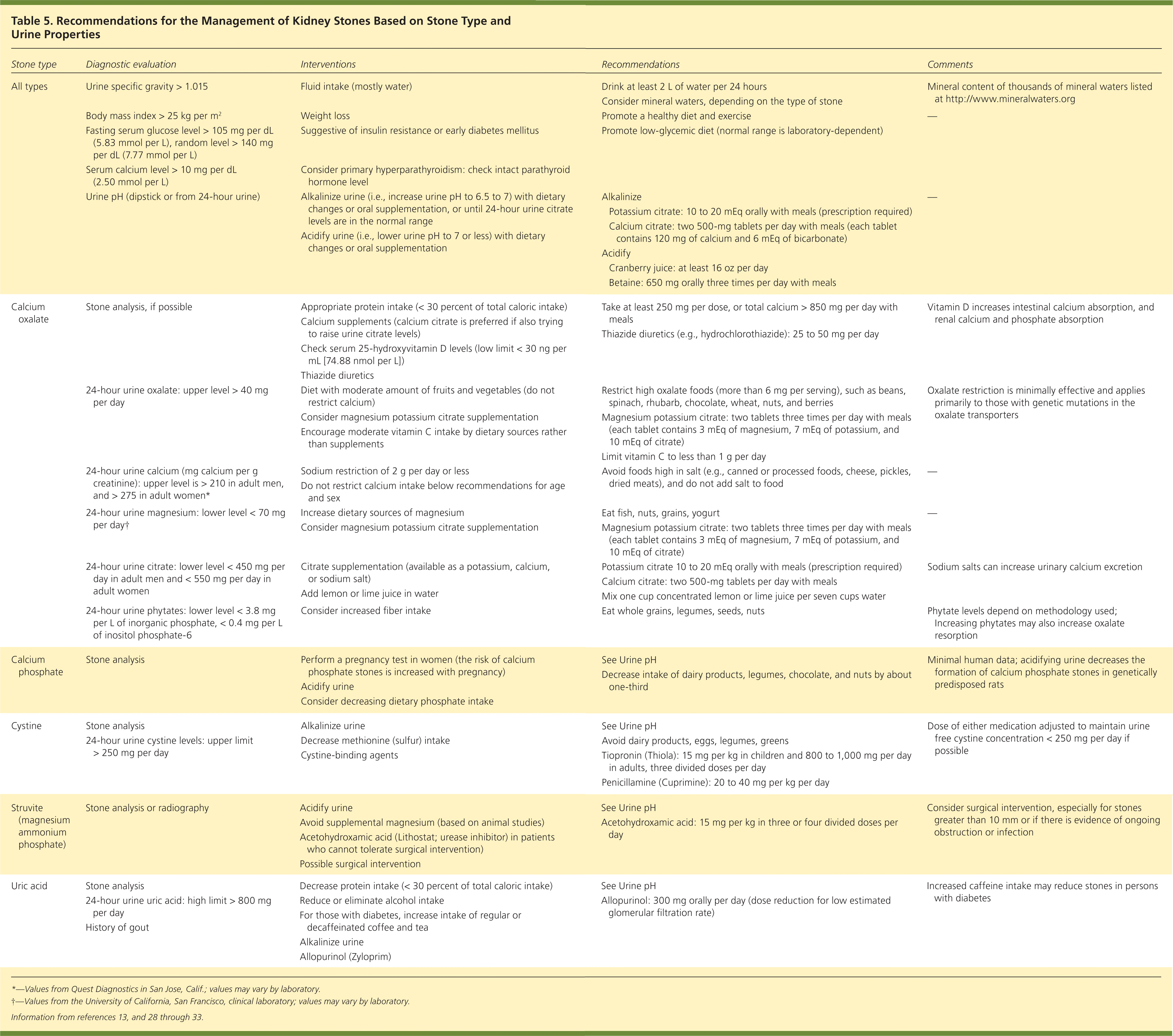
| Stone type | Diagnostic evaluation | Interventions | Recommendations | Comments |
|---|---|---|---|---|
| All types | Urine specific gravity > 1.015 | Fluid intake (mostly water) |
| Mineral content of thousands of mineral waters listed at http://www.mineralwaters.org |
| Body mass index > 25 kg per m2 | Weight loss |
| — | |
| Fasting serum glucose level > 105 mg per dL (5.83 mmol per L), random level > 140 mg per dL (7.77 mmol per L) | Suggestive of insulin resistance or early diabetes mellitus |
| ||
| Serum calcium level > 10 mg per dL (2.50 mmol per L) | Consider primary hyperparathyroidism: check intact parathyroid hormone level | |||
| Urine pH (dipstick or from 24-hour urine) | Alkalinize urine (i.e., increase urine pH to 6.5 to 7) with dietary changes or oral supplementation, or until 24-hour urine citrate levels are in the normal range |
| — | |
| Acidify urine (i.e., lower urine pH to 7 or less) with dietary changes or oral supplementation |
| |||
| Calcium oxalate | Stone analysis, if possible | Appropriate protein intake (< 30 percent of total caloric intake) |
| Vitamin D increases intestinal calcium absorption, and renal calcium and phosphate absorption |
| Calcium supplements (calcium citrate is preferred if also trying to raise urine citrate levels) | ||||
| Check serum 25-hydroxyvitamin D levels (low limit < 30 ng per mL [74.88 nmol per L]) | ||||
| Thiazide diuretics | ||||
| 24-hour urine oxalate: upper level > 40 mg per day | Diet with moderate amount of fruits and vegetables (do not restrict calcium) |
| Oxalate restriction is minimally effective and applies primarily to those with genetic mutations in the oxalate transporters | |
| Consider magnesium potassium citrate supplementation | ||||
| Encourage moderate vitamin C intake by dietary sources rather than supplements | ||||
| 24-hour urine calcium (mg calcium per g creatinine): upper level is > 210 in adult men, and > 275 in adult women* | Sodium restriction of 2 g per day or less |
| — | |
| Do not restrict calcium intake below recommendations for age and sex | ||||
| 24-hour urine magnesium: lower level < 70 mg per day † | Increase dietary sources of magnesium |
| — | |
| Consider magnesium potassium citrate supplementation | ||||
| 24-hour urine citrate: lower level < 450 mg per day in adult men and < 550 mg per day in adult women | Citrate supplementation (available as a potassium, calcium, or sodium salt) |
| Sodium salts can increase urinary calcium excretion | |
| Add lemon or lime juice in water | ||||
| 24-hour urine phytates: lower level < 3.8 mg per L of inorganic phosphate, < 0.4 mg per L of inositol phosphate-6 | Consider increased fiber intake |
| Phytate levels depend on methodology used; increasing phytates may also increase oxalate resorption | |
| Calcium phosphate | Stone analysis | Perform a pregnancy test in women (the risk of calcium phosphate stones is increased with pregnancy) |
| Minimal human data; acidifying urine decreases the formation of calcium phosphate stones in genetically predisposed rats |
| Acidify urine | ||||
| Consider decreasing dietary phosphate intake | ||||
| Cystine | Stone analysis 24-hour urine cystine levels: upper limit > 250 mg per day | Alkalinize urine |
| Dose of either medication adjusted to maintain urine free cystine concentration < 250 mg per day if possible |
| Decrease methionine (sulfur) intake | ||||
| Cystine-binding agents | ||||
| Struvite (magnesium ammonium phosphate) | Stone analysis or radiography | Acidify urine |
| Consider surgical intervention, especially for stones greater than 10 mm or if there is evidence of ongoing obstruction or infection |
| Avoid supplemental magnesium (based on animal studies) | ||||
| Acetohydroxamic acid (Lithostat; urease inhibitor) in patients who cannot tolerate surgical intervention) | ||||
| Possible surgical intervention | ||||
| Uric acid | Stone analysis 24-hour urine uric acid: high limit > 800 mg per day History of gout | Decrease protein intake (< 30 percent of total caloric intake) |
| Increased caffeine intake may reduce stones in persons with diabetes |
| Reduce or eliminate alcohol intake | ||||
| For those with diabetes, increase intake of regular or decaffeinated coffee and tea | ||||
| Alkalinize urine Allopurinol (Zyloprim) |
*—Values from Quest Diagnostics in San Jose, Calif.; values may vary by laboratory.
†—Values from the University of California–San Francisco clinical laboratory; values may vary by laboratory.
CAN BACTERIAL INFECTION TRIGGER RECURRENCE?
Bacteria exert both pathogenic and protective roles. Struvite stones are associated with recurrent infections because of high urinary pH levels from urease splitting bacteria and the body's inability to rid the urinary tract of bacteria that become embedded in the stones.34
Oxalobacter formigenes is an anaerobic bacterium that colonizes the intestinal tract, where it metabolizes oxalate to formate and carbon dioxide. Absence of O. formigenes colonization predisposes persons to oxalate stones.35 Preliminary studies of O. formigenes ingestion in healthy patients35 and in patients with primary hyperoxaluria36 demonstrated up to a 90 percent decrease in urinary oxalate levels.36 Larger studies of this potential therapy are ongoing.
DOES OBESITY INCREASE RISK AND WILL WEIGHT LOSS REDUCE RISK?
Obesity contributes to risk of kidney stones more than dietary factors. The associated changes in body composition pose biophysical challenges associated with disturbed thermogenesis and dehydration. Because body fat is hydrophobic, the proportion of body water decreases with increasing obesity, which can lead to dehydration.37 Additionally, the decrease in surface area to body volume complicates heat exchange and metabolic rate.38 Obesity is a proinflammatory state associated with electrolyte imbalances and altered urine chemistry. Obese persons with kidney stones are predisposed to hyperuricemia, gout, hypocitraturia, hyperuricosuria, and uric acid stones.39 A recent retrospective analysis found that patients with diabetes and kidney stones excrete more oxalate and have lower urine pH, which is partly a result of higher sulfate excretion and less acid excreted as ammonium ions.40,41 Patients with kidney disease who are obese or have diabetes may have a lesser genetic predisposition to kidney stones and greater responsiveness to environmental modification, such as a healthy diet and hydration.
Weight loss may improve or undermine management of kidney stones, depending on how it is achieved. Weight loss could be detrimental to prevention of kidney stones if associated with a high animal protein diet, laxative abuse, rapid loss of lean tissue, or poor hydration. High acid diets, such as the Atkins diet, increase the risk of uric acid stones. 42 Therefore, diet advice should be based on the type of kidney stone.
SHOULD PATIENTS REDUCE FRUCTOSE INTAKE?
Increased dietary fructose has been associated with up to a 38 percent higher risk of kidney stones. 43 Increased fructose intake increases urinary calcium excretion in persons with magnesium deficiency, and fructose is the only dietary carbohydrate known to raise uric acid levels. Additionally, sugar-sweetened beverages and orange juice have been linked to gout. 44
HOW CAN URINE BE ALKALINIZED (HIGHER URINE PH)? [ corrected]
For prevention of calcium oxalate, cystine, and uric acid stones, urine should be alkalinized.45,46 Western diets are characteristically high in acid-producing foods, such as grains, dairy products, legumes, and meat. Alkalinizing urine involves eating a diet high in fruits and vegetables, taking supplemental or prescription citrate, or drinking alkaline mineral waters.1
HOW CAN URINE BE ACIDIFIED (LOWER URINE PH)? [ corrected]
For prevention of calcium phosphate and struvite stones, urine should be acidified.47 Cranberry juice or betaine can lower urine pH without the adverse effects associated with acid-producing foods. Although table salt (sodium chloride) also lowers urine pH, it can increase blood pressure, insulin excretion, and urine calcium excretion.
ARE ALTERNATIVE THERAPIES HELPFUL?
Acupuncture and chiropractic manipulation may ease stone passage in patients with nerve impingement. Herbs have been used in acute kidney stone treatment since antiquity, but many uncertainties surround their contemporary use (e.g., quality and safety, interactions with medications or anesthesia, lack of stone-specific effectiveness). However, phytonutrients in green tea, turmeric, and berries may reduce the risk of infection, parsley may promote diuresis, and the traditional herb Agropyron repens may help achieve flushing of the urinary tract.
Data Sources: PubMed searches were completed in Clinical Queries using the key terms kidney stone, nephrolithiasis, chronic kidney disease, pregnancy, children, cystinuria, calcium oxalate, struvite, calcium phosphate, uric acid, magnesium, potassium, citrate, phytate, alkalinizing, aciduria, and uroscopy. PubMed was also searched for specific authors using the key terms, Bushinsky, Coe, Curhan, Moe, and Evans. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Google, Wikipedia, and UpToDate were also searched. Search date: April 17, 2011.