A more recent article on diagnosis and management of rheumatoid arthritis is available.
Am Fam Physician. 2011;84(11):1245-1252
Author disclosure: No relevant financial affiliations to disclose.
Rheumatoid arthritis is the most commonly diagnosed systemic inflammatory arthritis. Women, smokers, and those with a family history of the disease are most often affected. Criteria for diagnosis include having at least one joint with definite swelling that is not explained by another disease. The likelihood of a rheumatoid arthritis diagnosis increases with the number of small joints involved. In a patient with inflammatory arthritis, the presence of a rheumatoid factor or anti-citrullinated protein antibody, or elevated C-reactive protein level or erythrocyte sedimentation rate suggests a diagnosis of rheumatoid arthritis. Initial laboratory evaluation should also include complete blood count with differential and assessment of renal and hepatic function. Patients taking biologic agents should be tested for hepatitis B, hepatitis C, and tuberculosis. Earlier diagnosis of rheumatoid arthritis allows for earlier treatment with disease-modifying antirheumatic agents. Combinations of medications are often used to control the disease. Methotrexate is typically the first-line drug for rheumatoid arthritis. Biologic agents, such as tumor necrosis factor inhibitors, are generally considered second-line agents or can be added for dual therapy. The goals of treatment include minimization of joint pain and swelling, prevention of radiographic damage and visible deformity, and continuation of work and personal activities. Joint replacement is indicated for patients with severe joint damage whose symptoms are poorly controlled by medical management.
Rheumatoid arthritis (RA) is the most common inflammatory arthritis, with a lifetime prevalence of up to 1 percent worldwide.1 Onset can occur at any age, but peaks between 30 and 50 years.2 Disability is common and significant. In a large U.S. cohort, 35 percent of patients with RA had work disability after 10 years.3
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Patients with inflammatory joint disease should be referred to a rheumatology subspecialist, especially if symptoms last more than six weeks. | C | 16, 17 |
| In persons with RA, combination therapy with two or more disease-modifying antirheumatic drugs is more effective than monotherapy. However, more than one biologic agent should not be used at one time (e.g., adalimumab [Humira] with abatacept [Orencia]) because of the high risk of adverse effects. | A | 21, 22, 24 |
| A guided exercise program can improve quality of life and muscle strength in patients with RA. | B | 32, 34, 35 |
| Cardiovascular disease is the main cause of mortality in persons with RA; therefore, risk factors for coronary artery disease should be addressed in these patients. | C | 40, 41 |
Etiology and Pathophysiology
Like many autoimmune diseases, the etiology of RA is multifactorial. Genetic susceptibility is evident in familial clustering and monozygotic twin studies, with 50 percent of RA risk attributable to genetic factors.4 Genetic associations for RA include human leukocyte antigen-DR45 and -DRB1, and a variety of alleles called the shared epitope.6,7 Genome-wide association studies have identified additional genetic signatures that increase the risk of RA and other autoimmune diseases, including STAT4 gene and CD40 locus.5 Smoking is the major environmental trigger for RA, especially in those with a genetic predisposition.8 Although infections may unmask an autoimmune response, no particular pathogen has been proven to cause RA.9
RA is characterized by inflammatory pathways that lead to proliferation of synovial cells in joints. Subsequent pannus formation may lead to underlying cartilage destruction and bony erosions. Overproduction of proinflammatory cytokines, including tumor necrosis factor (TNF) and interleukin-6, drives the destructive process.10
RISK FACTORS
Older age, a family history of the disease, and female sex are associated with increased risk of RA, although the sex differential is less prominent in older patients.1 Both current and prior cigarette smoking increases the risk of RA (relative risk [RR] = 1.4, up to 2.2 for more than 40-pack-year smokers).11
Pregnancy often causes RA remission, likely because of immunologic tolerance.12 Parity may have long-lasting impact; RA is less likely to be diagnosed in parous women than in nulliparous women (RR = 0.61).13,14 Breastfeeding decreases the risk of RA (RR = 0.5 in women who breastfeed for at least 24 months), whereas early menarche (RR = 1.3 for those with menarche at 10 years of age or younger) and very irregular menstrual periods (RR = 1.5) increase risk.14 Use of oral contraceptive pills or vitamin E does not affect RA risk.15
Diagnosis
TYPICAL PRESENTATION
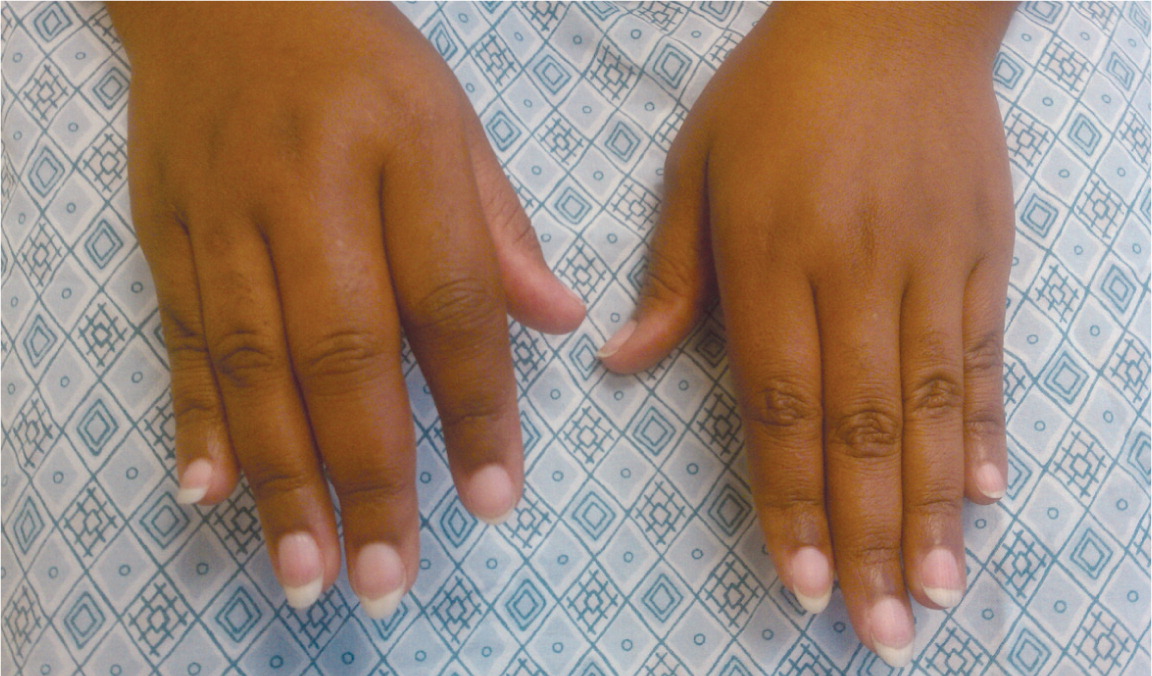
Patients with RA typically present with pain and stiffness in multiple joints. The wrists, proximal interphalangeal joints, and metacarpophalangeal joints are most commonly involved. Morning stiffness lasting more than one hour suggests an inflammatory etiology. Boggy swelling due to synovitis may be visible (Figure 1), or subtle synovial thickening may be palpable on joint examination. Patients may also present with more indolent arthralgias before the onset of clinically apparent joint swelling. Systemic symptoms of fatigue, weight loss, and low-grade fever may occur with active disease.
DIAGNOSTIC CRITERIA
In 2010, the American College of Rheumatology and European League Against Rheumatism collaborated to create new classification criteria for RA (Table 1).16 The new criteria are an effort to diagnose RA earlier in patients who may not meet the 1987 American College of Rheumatology classification criteria. The 2010 criteria do not include presence of rheumatoid nodules or radiographic erosive changes, both of which are less likely in early RA. Symmetric arthritis is also not required in the 2010 criteria, allowing for early asymmetric presentation.

| Criteria | Score | |
|---|---|---|
| Target population (who should be tested?): patients who | ||
| 1) have at least one joint with definite clinical synovitis (swelling)* | ||
| 2) with the synovitis not better explained by another disease† | ||
| Classification criteria for RA (score-based algorithm: add score of categories A through D; a score of ≥ 6 out of 10 is needed for classification of a patient as having definite RA)‡ | ||
| A. Joint involvement§ | ||
| One large joint|| | 0 | |
| Two to 10 large joints | 1 | |
| One to three small joints (with or without involvement of large joints)¶ | 2 | |
| Four to 10 small joints (with or without involvement of large joints) | 3 | |
| > 10 joints (at least one small joint)** | 5 | |
| B. Serology (at least one test result is needed for classification)†† | ||
| Negative RF and negative ACPA | 0 | |
| Low positive RF or low positive ACPA | 2 | |
| High positive RF or high positive ACPA | 3 | |
| C. Acute phase reactants (at least one test result is needed for classification)‡‡ | ||
| Normal CRP and normal ESR | 0 | |
| Abnormal CRP or normal ESR | 1 | |
| D. Duration of symptoms§§ | ||
| < six weeks | 0 | |
| ≥ six weeks | 1 | |

| Patient characteristics | Points | |
|---|---|---|
| Age | Years × 0.02 | |
| Female sex | 1.0 | |
| Distribution of affected joints (patients may receive points for more than one item) | ||
| Small joints of hands or feet | 0.5 | |
| Symmetric | 0.5 | |
| Upper extremities | 1.0 | |
| Upper and lower extremities | 1.5 | |
| Score for morning stiffness on a 100-mm visual analog scale | ||
| 26 to 90 mm | 1.0 | |
| > 90 mm | 2.0 | |
| Number of tender joints | ||
| Four to 10 | 0.5 | |
| ≥ 11 | 1.0 | |
| Number of swollen joints | ||
| Four to 10 | 0.5 | |
| ≥ 11 | 1.0 | |
| C-reactive protein level | ||
| 5 to 50 mg per L (47.62 to 476.20 nmol per L) | 0.5 | |
| ≥ 51 mg per L (485.72 nmol per L) | 1.5 | |
| Positive for rheumatoid factor | 1.0 | |
| Positive for anti-citrullinated protein antibody | 2.0 | |
| Total: | ______ | |
DIAGNOSTIC TESTS
Autoimmune diseases such as RA are often characterized by the presence of autoantibodies. Rheumatoid factor is not specific for RA and may be present in patients with other diseases, such as hepatitis C, and in healthy older persons. Anti-citrullinated protein antibody is more specific for RA and may play a role in disease pathogenesis.6 Approximately 50 to 80 percent of persons with RA have rheumatoid factor, anti-citrullinated protein antibody, or both.10 Patients with RA may have a positive antinuclear antibody test result, and the test is of prognostic importance in juvenile forms of this disease.19 C-reactive protein levels and erythrocyte sedimentation rate are often increased with active RA, and these acute phase reactants are part of the new RA classification criteria.16 C-reactive protein levels and erythrocyte sedimentation rate may also be used to follow disease activity and response to medication.
Baseline complete blood count with differential and assessment of renal and hepatic function are helpful because the results may influence treatment options (e.g., a patient with renal insufficiency or significant thrombocytopenia likely would not be prescribed a nonsteroidal anti-inflammatory drug [NSAID]). Mild anemia of chronic disease occurs in 33 to 60 percent of all patients with RA,20 although gastrointestinal blood loss should also be considered in patients taking corticosteroids or NSAIDs. Methotrexate is contraindicated in patients with hepatic disease, such as hepatitis C, and in patients with significant renal impairment.21 Biologic therapy, such as a TNF inhibitor, requires a negative tuberculin test or treatment for latent tuberculosis. Hepatitis B reactivation can also occur with TNF inhibitor use.22 Radiography of hands and feet should be performed to evaluate for characteristic periarticular erosive changes, which may be indicative of a more aggressive RA subtype.10
DIFFERENTIAL DIAGNOSIS
Skin findings suggest systemic lupus erythematosus, systemic sclerosis, or psoriatic arthritis. Polymyalgia rheumatica should be considered in an older patient with symptoms primarily in the shoulder and hip, and the patient should be asked questions related to associated temporal arteritis. Chest radiography is helpful to evaluate for sarcoidosis as an etiology of arthritis.
Patients with inflammatory back symptoms, a history of inflammatory bowel disease, or inflammatory eye disease may have spondyloarthropathy. Persons with less than six weeks of symptoms may have a viral process, such as parvovirus. Recurrent self-limited episodes of acute joint swelling suggest crystal arthropathy, and arthrocentesis should be performed to evaluate for monosodium urate monohydrate or calcium pyrophosphate dihydrate crystals. The presence of numerous myofascial trigger points and somatic symptoms may suggest fibromyalgia, which can coexist with RA. To help guide diagnosis and determine treatment strategy, patients with inflammatory arthritis should be promptly referred to a rheumatology subspecialist.16,17
Treatment
After RA has been diagnosed and an initial evaluation performed, treatment should begin. Recent guidelines have addressed the management of RA,21,22 but patient preference also plays an important role. There are special considerations for women of childbearing age because many medications have deleterious effects on pregnancy. Goals of therapy include minimizing joint pain and swelling, preventing deformity (such as ulnar deviation) and radiographic damage (such as erosions), maintaining quality of life (personal and work), and controlling extra-articular manifestations. Disease-modifying antirheumatic drugs (DMARDs) are the mainstay of RA therapy.
DMARDS
DMARDs can be biologic or nonbiologic (Table 3).23 Biologic agents include monoclonal antibodies and recombinant receptors to block cytokines that promote the inflammatory cascade responsible for RA symptoms. Methotrexate is recommended as the first-line treatment in patients with active RA, unless contraindicated or not tolerated.21 Leflunomide (Arava) may be used as an alternative to methotrexate, although gastrointestinal adverse effects are more common. Sulfasalazine (Azulfidine) or hydroxychloroquine (Plaquenil) is recommended as monotherapy in patients with low disease activity or without poor prognostic features (e.g., seronegative, nonerosive RA).21,22
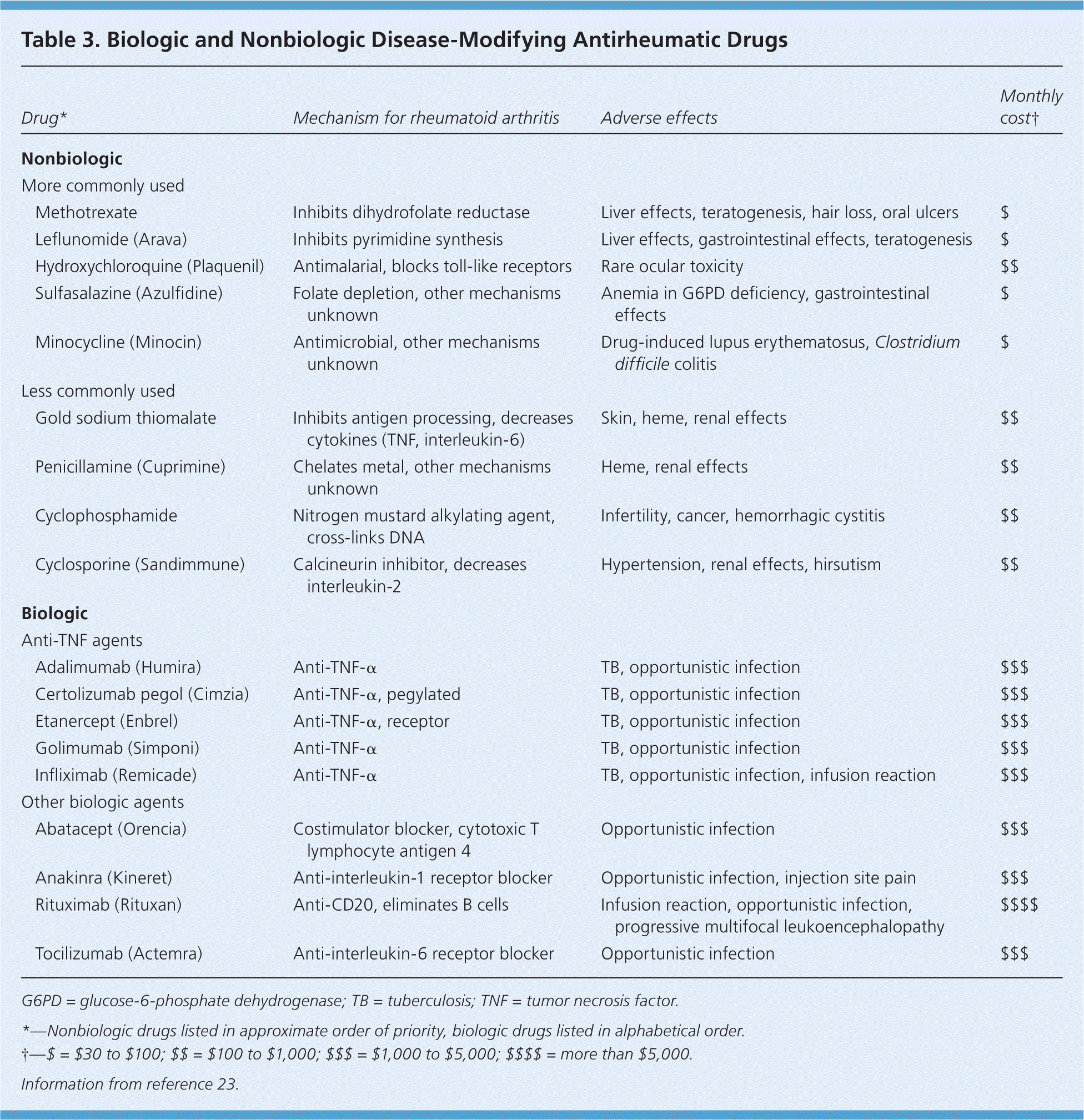
| Drug* | Mechanism for rheumatoid arthritis | Adverse effects | Monthly cost † | |
|---|---|---|---|---|
| Nonbiologic | ||||
| More commonly used | ||||
| Methotrexate | Inhibits dihydrofolate reductase | Liver effects, teratogenesis, hair loss, oral ulcers | $ | |
| Leflunomide (Arava) | Inhibits pyrimidine synthesis | Liver effects, gastrointestinal effects, teratogenesis | $ | |
| Hydroxychloroquine (Plaquenil) | Antimalarial, blocks toll-like receptors | Rare ocular toxicity | $$ | |
| Sulfasalazine (Azulfidine) | Folate depletion, other mechanisms unknown | Anemia in G6PD deficiency, gastrointestinal effects | $ | |
| Minocycline (Minocin) | Antimicrobial, other mechanisms unknown | Drug-induced lupus erythematosus, Clostridium difficile colitis | $ | |
| Less commonly used | ||||
| Gold sodium thiomalate | Inhibits antigen processing, decreases cytokines (TNF, interleukin-6) | Skin, heme, renal effects | $$ | |
| Penicillamine (Cuprimine) | Chelates metal, other mechanisms unknown | Heme, renal effects | $$ | |
| Cyclophosphamide | Nitrogen mustard alkylating agent, cross-links DNA | Infertility, cancer, hemorrhagic cystitis | $$ | |
| Cyclosporine (Sandimmune) | Calcineurin inhibitor, decreases interleukin-2 | Hypertension, renal effects, hirsutism | $$ | |
| Biologic | ||||
| Anti-TNF agents | ||||
| Adalimumab (Humira) | Anti-TNF-α | TB, opportunistic infection | $$$ | |
| Certolizumab pegol (Cimzia) | Anti-TNF-α, pegylated | TB, opportunistic infection | $$$ | |
| Etanercept (Enbrel) | Anti-TNF-α, receptor | TB, opportunistic infection | $$$ | |
| Golimumab (Simponi) | Anti-TNF-α | TB, opportunistic infection | $$$ | |
| Infliximab (Remicade) | Anti-TNF-α | TB, opportunistic infection, infusion reaction | $$$ | |
| Other biologic agents | ||||
| Abatacept (Orencia) | Costimulator blocker, cytotoxic T lymphocyte antigen 4 | Opportunistic infection | $$$ | |
| Anakinra (Kineret) | Anti-interleukin-1 receptor blocker | Opportunistic infection, injection site pain | $$$ | |
| Rituximab (Rituxan) | Anti-CD20, eliminates B cells | Infusion reaction, opportunistic infection, progressive multifocal leukoencephalopathy | $$$$ | |
| Tocilizumab (Actemra) | Anti-interleukin-6 receptor blocker | Opportunistic infection | $$$ | |
Combination therapy with two or more DMARDs is more effective than monotherapy; however, adverse effects may also be greater.24 If RA is not well controlled with a nonbiologic DMARD, a biologic DMARD should be initiated.21,22 TNF inhibitors are the first-line biologic therapy and are the most studied of these agents. If TNF inhibitors are ineffective, additional biologic therapies can be considered. Simultaneous use of more than one biologic therapy (e.g., adalimumab [Humira] with abatacept [Orencia]) is not recommended because of an unacceptable rate of adverse effects.21
NSAIDS AND CORTICOSTEROIDS
COMPLEMENTARY THERAPIES
Dietary interventions, including vegetarian and Mediterranean diets, have been studied in the treatment of RA without convincing evidence of benefit.25,26 Despite some favorable outcomes, there is a lack of evidence for the effectiveness of acupuncture in placebo-controlled trials of patients with RA.27,28 In addition, thermotherapy and therapeutic ultrasound for RA have not been studied adequately.29,30 A Cochrane review of herbal treatments for RA concluded that gamma-linolenic acid (from evening primrose or black currant seed oil) and Tripterygium wilfordii (thunder god vine) have potential benefits.31 It is important to inform patients that serious adverse effects have been reported with use of herbal therapy.31
EXERCISE AND PHYSICAL THERAPY
Results of randomized controlled trials support physical exercise to improve quality of life and muscle strength in patients with RA.32,33 Exercise training programs have not been shown to have deleterious effects on RA disease activity, pain scores, or radiographic joint damage.34 Tai chi has been shown to improve ankle range of motion in persons with RA, although randomized trials are limited.35 Randomized controlled trials of Iyengar yoga in young adults with RA are underway.36
DURATION OF TREATMENT
Remission is obtainable in 10 to 50 percent of patients with RA, depending on how remission is defined and the intensity of therapy.10 Remission is more likely in males, nonsmokers, persons younger than 40 years, and in those with late-onset disease (patients older than 65 years), with shorter duration of disease, with milder disease activity, without elevated acute phase reactants, and without positive rheumatoid factor or anti- citrullinated protein antibody findings.37
After the disease is controlled, medication dosages may be cautiously decreased to the minimum amount necessary. Patients will require frequent monitoring to ensure stable symptoms, and prompt increase in medication is recommended with disease flare-ups.22
JOINT REPLACEMENT
Joint replacement is indicated when there is severe joint damage and unsatisfactory control of symptoms with medical management. Long-term outcomes are good, with only 4 to 13 percent of large joint replacements requiring revision within 10 years.38 The hip and knee are the most commonly replaced joints.
Long-term Monitoring

| Manifestation | Characteristics |
|---|---|
| Cardiac | |
| Accelerated atherosclerosis | Leading cause of death in patients with RA |
| Pericarditis | Present in 30 to 50 percent of persons with RA on autopsy, rarely leads to tamponade |
| Eye | |
| Episcleritis/scleritis | Acute, red, painful eye; occurs in less than 1 percent of patients with RA |
| Keratoconjunctivitis sicca | Secondary Sjögren syndrome, dry mouth may also occur |
| Peripheral ulcerative keratitis | More severe scleritis, if untreated can perforate anterior chamber |
| Hematologic | |
| Amyloidosis | Caused by chronic inflammation |
| Felty syndrome | Splenomegaly, neutropenia, and thrombocytopenia |
| Nervous system | |
| Cervical myelopathy | Caused by C1–C2 subluxation, seen on flexion-extension radiography |
| Neuropathy | Carpal tunnel, mononeuritis multiplex (foot drop) |
| Pulmonary | |
| Caplan syndrome | Nodules and pneumoconiosis (e.g., in coal miners) |
| Interstitial lung disease | May resemble bronchiolitis obliterans with organizing pneumonia, idiopathic pulmonary fibrosis, patient may also have pulmonary arterial hypertension |
| Pleural effusion | Exudative effusion with markedly low glucose level |
| Pulmonary nodules | Can be asymptomatic |
| Skin | |
| Rheumatoid nodules | Firm or rubbery, located on pressure areas (e.g., olecranon) |
| Vasculitis | Poor prognosis, increased mortality, rare but occurs with severe RA (i.e., erosive, deforming, and seropositive disease) |
Patients with RA have a twofold increased risk of lymphoma, which is thought to be caused by the underlying inflammatory process, and not a consequence of medical treatment.39 Patients with RA are also at an increased risk of coronary artery disease, and physicians should work with patients to modify risk factors, such as smoking, high blood pressure, and high cholesterol.40,41 Class III or IV congestive heart failure (CHF) is a contraindication for using TNF inhibitors, which can worsen CHF outcomes.21 In patients with RA and malignancy, caution is needed with continued use of DMARDs, especially TNF inhibitors. Biologic DMARDs, methotrexate, and leflunomide should not be initiated in patients with active herpes zoster, significant fungal infection, or bacterial infection requiring antibiotics.21 Complications of RA and its treatments are listed in Table 5.1,2,10

| Complication | Comments | |
|---|---|---|
| Depression | Affects up to 40 percent of patients with RA, can also be caused by corticosteroid use | |
| Infection | May be caused by RA itself or use of immunosuppressants | |
| Malignancy | ||
| Lymphoma | Risk is double in persons with RA, independent of immunosuppressant use | |
| Lung cancer | Caused by smoking, underlying interstitial lung disease | |
| Skin cancer | Risk may be increased with immunosuppressant use | |
Prognosis
Patients with RA live three to 12 years less than the general population.40 Increased mortality in these patients is mainly due to accelerated cardiovascular disease, especially in those with high disease activity and chronic inflammation. The relatively new biologic therapies may reverse progression of atherosclerosis and extend life in those with RA.41
Data Sources: A PubMed search was completed in Clinical Queries using the key terms rheumatoid arthritis, extra-articular manifestations, and disease-modifying antirheumatic agents. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Also searched were the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, Essential Evidence, and UpToDate. Search date: September 20, 2010.