
This is a corrected version of the article that appeared in print.
Am Fam Physician. 2012;85(9):910-911
Author disclosure: No relevant financial affiliations to disclose.
Tapentadol (Nucynta) is a centrally-acting opioid analgesic approved for the relief of moderate to severe pain in adults. It has two mechanisms of action: in addition to acting at the mu opioid receptor, tapentadol blocks norepinephrine uptake, although the clinical relevance of this additional action is not known. [ corrected] It is currently available in an immediate-release form, which is approved for adults with severe acute pain, and an extended-release form, which is used for moderate to severe chronic pain. Extended-release tapentadol is not intended for the management of acute or postoperative pain. Tapentadol is classified as a U.S. Drug Enforcement Administration (DEA) schedule II controlled substance.
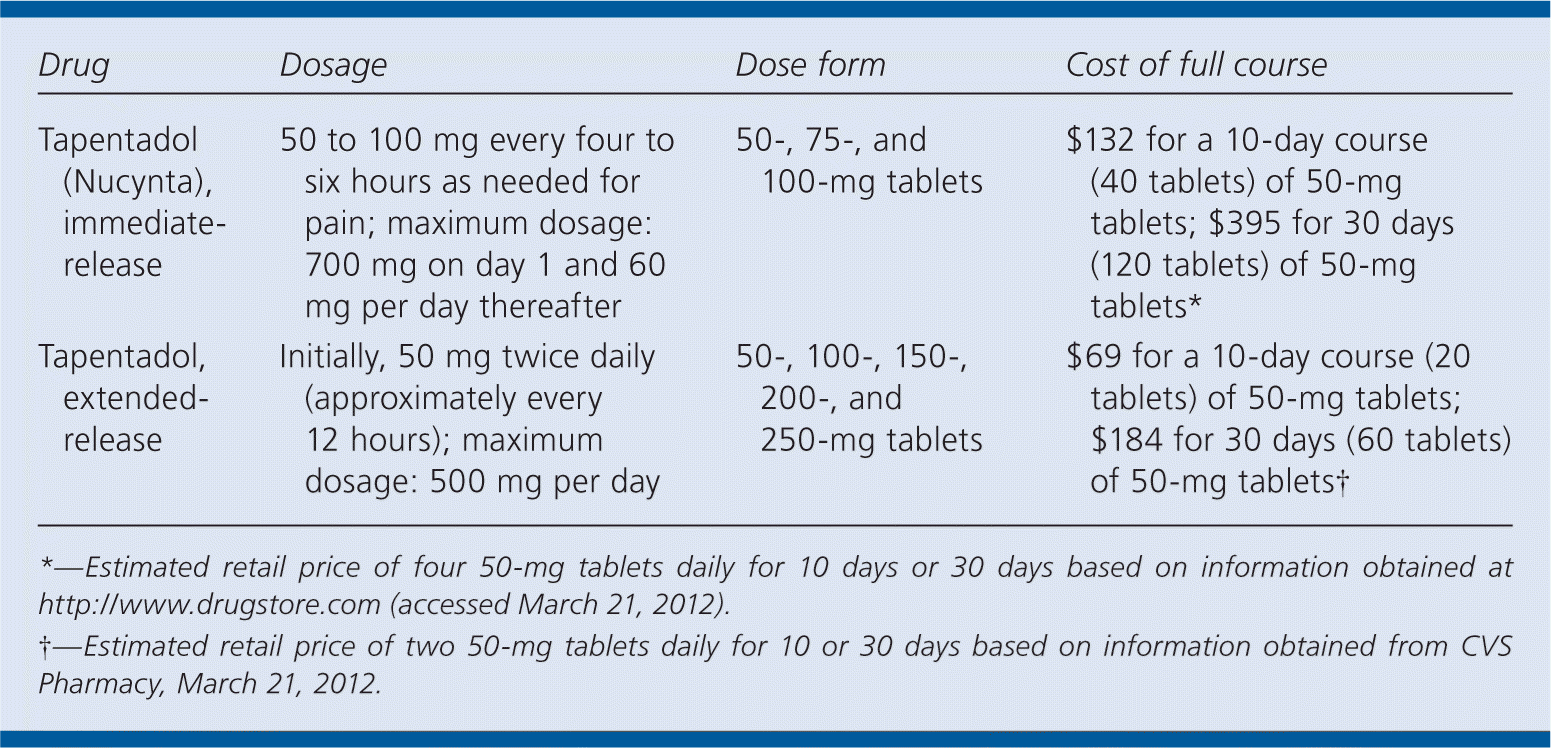
| Drug | Dosage | Dose form | Cost of full course |
|---|---|---|---|
| Tapentadol (Nucynta), immediate-release | 50 to 100 mg every four to six hours as needed for pain; maximum dosage: 700 mg on day 1 and 60 mg per day thereafter | 50-, 75-, and 100-mg tablets | $132 for a 10-day course tablets; $395 for 30 days (120 tablets) of 50-mg tablets* |
| Tapentadol, extended-release | Initially, 50 mg twice daily (approximately every 12 hours); maximum dosage: 500 mg per day | 50-, 100-, 150-, 200-, and 250-mg tablets | $69 for a 10-day course (20 tablets) of 50-mg tablets; $184 for 30 days (60 tablets) of 50-mg tablets† |
SAFETY
The primary safety concern with tapentadol is respiratory depression. It should not be used in patients with severe respiratory illness. Older or debilitated patients and those with an underlying respiratory illness should take the lowest effective dose. Tapentadol should not be used concurrently with monoamine oxidase inhibitors because of the risk of hemodynamic lability; there is also a theoretical risk of serotonin syndrome in patients taking serotonin reuptake inhibitors. Concurrent use of extended-release tapentadol and alcohol can be fatal. The extended-release tablets must not be split, crushed, or broken because rapid absorption may be fatal. Tapentadol is a U.S. Food and Drug Administration pregnancy category C drug and should not be used during breastfeeding. It has not been studied in children. As with all DEA schedule II narcotics, the usual risks of opiate addiction, diversion, and misuse apply.1,2
TOLERABILITY
Nausea is the most common adverse effect associated with immediate-release tapentadol. The 50-mg dose has a lower rate of nausea and other gastrointestinal adverse effects (18 to 35 percent) than a 10-mg dose of oxycodone (Oxycontin; 35 to 63 percent).3–6 At higher doses of tapentadol (75 to 100 mg), rates of nausea are similar to those of oxycodone.5 Extended-release tapentadol has lower rates of nausea and constipation than the controlled-release form of oxycodone.7 Rates of central nervous system adverse effects, such as dizziness, somnolence, headache, and fatigue, are comparable between tapentadol and oxycodone.4
EFFECTIVENESS
Although equianalgesic dosing has not been formally established between tapentadol and any other opioid, tapentadol is roughly equianalgesic to hydromorphone (Dilaudid) at its usual dosing (starting dose of 2 mg).8 In clinical trials, the 50-mg dose of tapentadol has been established as providing pain relief similar to that of 10-mg oxycodone. There are no published comparisons between tapentadol and nonopioid pain relievers.
Immediate-release tapentadol in dosages of 50 to 100 mg every four to six hours has been studied in the treatment of end-stage osteoarthritis3 and in patients undergoing orthopedic surgery.5 Extended-release tapentadol in dosages of 100 to 250 mg twice daily compares favorably with placebo for patients with painful diabetic peripheral neuropathy,9 and is as effective as controlled-release oxycodone (20 to 50 mg) for the treatment of low back pain.7 Tapentadol has not been studied in other painful disorders, but it is labeled for use for any moderate to severe pain. Its long-term effectiveness has not been evaluated.
PRICE
SIMPLICITY
Immediate-release tapentadol is given in dosages of 50, 75, or 100 mg every four to six hours. Extended-release tapentadol is given in dosages of 50 to 250 mg twice a day. No dosing adjustment is necessary for patients with mild to moderate renal impairment or mild hepatic impairment. Because tapentadol is a DEA schedule II controlled substance, it is governed by laws regarding who can prescribe it and how it may be dispensed.
Bottom Line
Tapentadol is a DEA schedule II opioid medication with a safety profile, duration of action, frequency of dosing, and analgesic effect similar to those of other oral narcotics, although it is more expensive. At low doses, immediate-release tapentadol is better tolerated than oxycodone in terms of gastrointestinal adverse effects, and in the long term, extended-release tapentadol is better tolerated than controlled-release oxycodone. The potential for better gastrointestinal tolerability must be weighed against the higher price.
