Summary of Recommendations and Evidence
This recommendation statement applies to women who have a cervix, regardless of sexual history (Table 1). It does not apply to women who have received a diagnosis of a high-grade precancerous cervical lesion or cervical cancer, women with in utero exposure to diethylstilbestrol, or women who are immunocompromised (such as those who are positive for human immunodeficiency virus [HIV]).
Table 1. Screening for Cervical Cancer: Clinical Summary of the USPSTF Recommendation
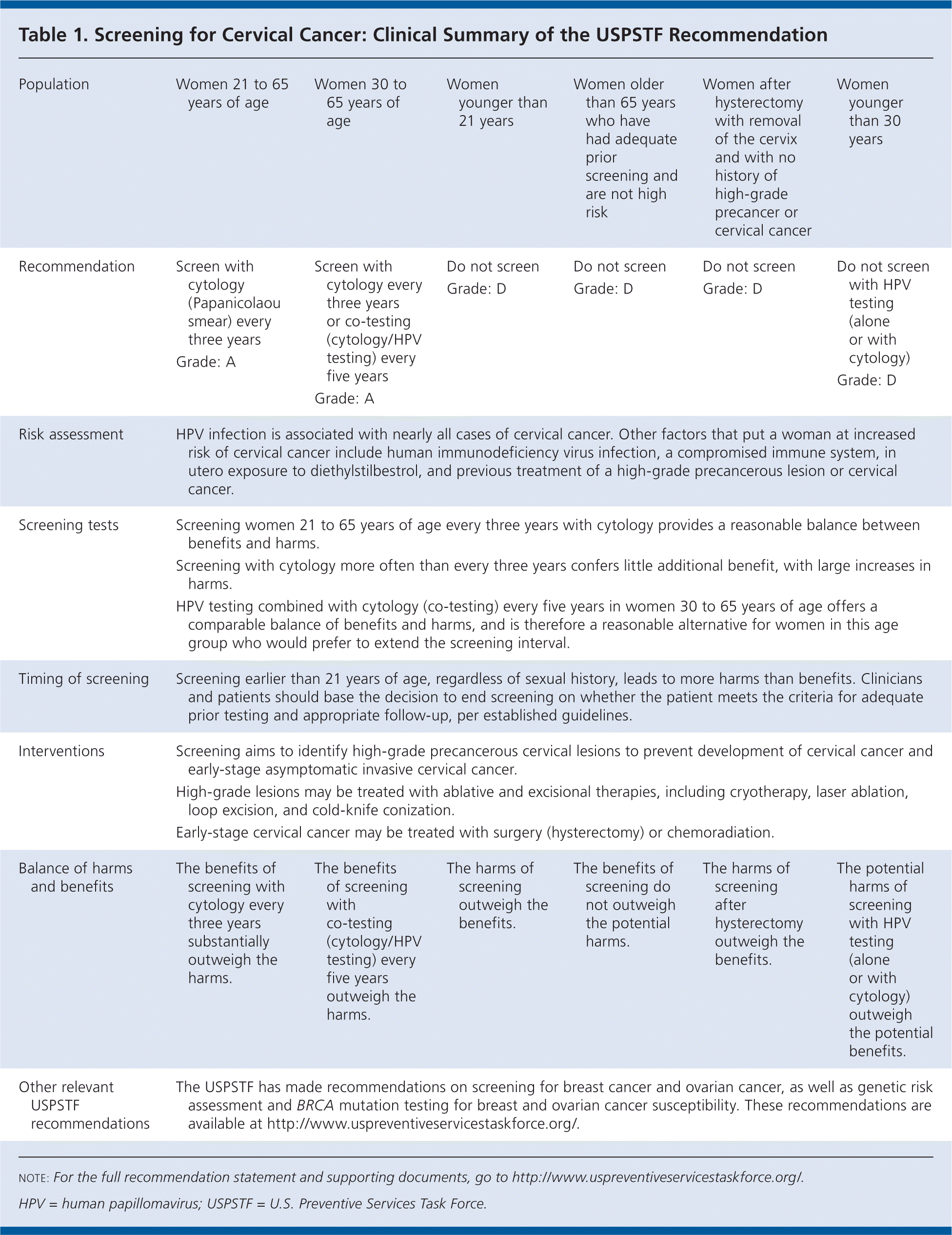
| Population | Women 21 to 65 years of age | Women 30 to 65 years of age | Women younger than 21 years | Women older than 65 years who have had adequate prior screening and are not high risk | Women after hysterectomy with removal of the cervix and with no history of high-grade precancer or cervical cancer | Women younger than 30 years |
| Recommendation | Screen with cytology (Papanicolaou smear) every three years Grade: A | Screen with cytology every three years or co-testing (cytology/HPV testing) every five years Grade: A | Do not screen Grade: D | Do not screen Grade: D | Do not screen Grade: D | Do not screen with HPV testing (alone or with cytology) Grade: D |
| Risk assessment | HPV infection is associated with nearly all cases of cervical cancer. Other factors that put a woman at increased risk of cervical cancer include human immunodeficiency virus infection, a compromised immune system, in utero exposure to diethylstilbestrol, and previous treatment of a high-grade precancerous lesion or cervical cancer. | |||||
| Screening tests | Screening women 21 to 65 years of age every three years with cytology provides a reasonable balance between benefits and harms. | |||||
| Screening with cytology more often than every three years confers little additional benefit, with large increases in harms. | ||||||
| HPV testing combined with cytology (co-testing) every five years in women 30 to 65 years of age offers a comparable balance of benefits and harms, and is therefore a reasonable alternative for women in this age group who would prefer to extend the screening interval. | ||||||
| Timing of screening | Screening earlier than 21 years of age, regardless of sexual history, leads to more harms than benefits. Clinicians and patients should base the decision to end screening on whether the patient meets the criteria for adequate prior testing and appropriate follow-up, per established guidelines. | |||||
| Interventions | Screening aims to identify high-grade precancerous cervical lesions to prevent development of cervical cancer and early-stage asymptomatic invasive cervical cancer. | |||||
| High-grade lesions may be treated with ablative and excisional therapies, including cryotherapy, laser ablation, loop excision, and cold-knife conization. | ||||||
| Early-stage cervical cancer may be treated with surgery (hysterectomy) or chemoradiation. | ||||||
| Balance of harms and benefits | The benefits of screening with cytology every three years substantially outweigh the harms. | The benefits of screening with co-testing (cytology/HPV testing) every five years outweigh the harms. | The harms of screening outweigh the benefits. | The benefits of screening do not outweigh the potential harms. | The harms of screening after hysterectomy outweigh the benefits. | The potential harms of screening with HPV testing (alone or with cytology) outweigh the potential benefits. |
| Other relevant USPSTF recommendations | The USPSTF has made recommendations on screening for breast cancer and ovarian cancer, as well as genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility. These recommendations are available at http://www.uspreventiveservicestaskforce.org/. | |||||
note: For the full recommendation statement and supporting documents, go to http://www.uspreventiveservicestaskforce.org/.
HPV = human papillomavirus; USPSTF = U.S. Preventive Services Task Force.
The U.S. Preventive Services Task Force (USPSTF) recommends screening for cervical cancer in women 21 to 65 years of age with cytology (Papanicolaou [Pap] smear) every three years or, for women 30 to 65 years of age who want to lengthen the screening interval, screening with a combination of cytology and human papillomavirus (HPV) testing every five years. See the Clinical Considerations section for discussion of cytology method, HPV testing, and screening interval. A recommendation.
The USPSTF recommends against screening for cervical cancer in women younger than 21 years. D recommendation
The USPSTF recommends against screening for cervical cancer in women older than 65 years who have had adequate prior screening and are not otherwise at high risk of cervical cancer. See the Clinical Considerations section for discussion of adequacy of prior screening and risk factors. D recommendation.
The USPSTF recommends against screening for cervical cancer in women who have had a hysterectomy with removal of the cervix and who do not have a history of a high-grade precancerous lesion (cervical intraepithelial neoplasia [CIN] grade 2 or 3) or cervical cancer. D recommendation.
The USPSTF recommends against screening for cervical cancer with HPV testing, alone or in combination with cytology, in women younger than 30 years. D recommendation.
Rationale
IMPORTANCE
The age-adjusted annual incidence rate of cervical cancer is 6.6 cases per 100,000 women, according to data from 2008.1–3 An estimated 12,200 new cases of cervical cancer and 4,210 deaths occurred in the United States in 2010.1 Cervical cancer deaths in the United States have decreased dramatically since the implementation of widespread cervical cancer screening. Most cases of cervical cancer occur in women who have not been appropriately screened.2,3 Strategies that aim to ensure that all women are screened at the appropriate interval and receive adequate follow-up are most likely to be successful in further reducing cervical cancer incidence and mortality in the United States.
DETECTION
Screening with cervical cytology or testing for multiple oncogenic HPV types (a test for the presence of more than two high-risk or carcinogenic HPV types [i.e., HPV testing]) can lead to detection of high-grade precancerous cervical lesions and cervical cancer.
BENEFITS OF DETECTION AND EARLY INTERVENTION/TREATMENT
Women 21 to 65 years of age. There is convincing evidence that screening women 21 to 65 years of age with cytology every three years substantially reduces cervical cancer incidence and mortality. Among women 30 to 65 years of age, there is adequate evidence that screening with a combination of cytology and HPV testing (co-testing) every five years provides benefits similar to those seen with cytology screening alone every three years.
Among women younger than 30 years, there is adequate evidence that screening with HPV testing (alone or in combination with cytology) confers little to no benefit.
Women younger than 21 years. There is adequate evidence that screening women younger than 21 years (regardless of sexual history) does not reduce cervical cancer incidence and mortality compared with beginning screening at 21 years of age.4
Women older than 65 years. There is adequate evidence that screening women older than 65 years who have had adequate prior screening and are not otherwise at high risk provides little to no benefit.
Women after hysterectomy. There is convincing evidence that continued screening after hysterectomy with removal of the cervix for indications other than a high-grade precancerous lesion or cervical cancer provides no benefit.
HARMS OF DETECTION AND EARLY INTERVENTION/TREATMENT
Screening with cervical cytology or HPV testing can lead to harms, and the harms of screening can take many forms. Abnormal test results can lead to more frequent testing and invasive diagnostic procedures, such as colposcopy and cervical biopsy. Evidence from randomized controlled trials and observational studies indicates that harms from these diagnostic procedures include vaginal bleeding, pain, infection, and failure to diagnose (due to inadequate sampling). Abnormal screening test results are also associated with mild psychological harms; short-term increases in anxiety, distress, and concern about health have been reported with cytology and HPV testing.
Harms of treatment of screening-detected disease. The harms of treatment include risks from the treatment procedure itself and the potential downstream consequences of treatment. Summary evidence from observational studies indicates that some treatments for precancerous lesions (such as cold-knife conization and loop excision) are associated with adverse pregnancy outcomes, such as preterm delivery, that can lead to low birth weight in infants and perinatal death.2 Evidence is convincing that many precancerous cervical lesions will regress and that other lesions are so indolent and slow-growing that they will not become clinically important over a woman's lifetime; identification and treatment of these lesions constitute overdiagnosis. It is difficult to estimate the precise magnitude of overdiagnosis associated with any screening or treatment strategy, but it is of concern because it confers no benefit and leads to unnecessary surveillance, diagnostic tests, and treatments with the associated harms.
Women 21 to 65 years of age. There is adequate evidence that the harms of screening for cervical cancer with cytology alone or in combination with HPV testing in women 30 to 65 years of age are moderate. Positive screening results are more common with strategies that include HPV testing than with strategies that use cytology alone. Therefore, the likelihood of prolonged surveillance and overtreatment may increase with strategies that incorporate HPV testing. Cervical treatments may increase the risk of adverse pregnancy outcomes (e.g., cervical insufficiency, preterm delivery) in women who have not yet completed childbearing.
Women younger than 30 years. There is adequate evidence that the harms of HPV testing (alone or in combination with cytology) in women younger than 30 years are moderate.
Women younger than 21 years. There is adequate evidence that the harms of screening in women younger than 21 years are moderate.
Women older than 65 years. There is adequate evidence that the harms of screening in women older than 65 years who have had adequate prior screening and are not otherwise at high risk are at least small.
Women after hysterectomy. There is adequate evidence that screening after hysterectomy among women who do not have a history of a high-grade precancerous lesion or cervical cancer is associated with harms.
USPSTF ASSESSMENT
The USPSTF concludes that for women 21 to 65 years of age, there is high certainty that the benefits of screening with cytology every three years substantially outweigh the harms.
For women 30 to 65 years of age, there is high certainty that the benefits of screening with a combination of cytology and HPV testing (co-testing) every five years outweigh the harms.
For women younger than 21 years, regardless of sexual history, there is moderate certainty that the harms of screening outweigh the benefits.
For women older than 65 years who have had adequate prior screening and are not otherwise at high risk of cervical cancer, there is moderate certainty that the benefits of screening do not outweigh the potential harms.
For women who have had a hysterectomy with removal of the cervix for indications other than a high-grade pre-cancerous lesion or cancer, there is high certainty that the harms of screening outweigh the benefits.
For women younger than 30 years, there is moderate certainty that the potential harms of screening with HPV testing (alone or in combination with cytology) outweigh the potential benefits.
Clinical Considerations
PATIENT POPULATION
This recommendation statement applies to all women who have a cervix, regardless of sexual history. This recommendation statement does not apply to women who have received a diagnosis of a high-grade precancerous cervical lesion or cervical cancer, women with in utero exposure to diethylstilbestrol, or women who are immunocompromised (such as those who are HIV positive).
SCREENING TESTS
The effectiveness of cervical cancer screening observed in the United States over the past several decades is attributed to the use of conventional cytology. Current evidence indicates that there are no clinically important differences between liquid-based cytology and conventional cytology. The USPSTF realizes that the choice of cytology method may not be under the direct control of the clinician, and considers cytology screening in appropriate age groups at appropriate intervals to be of substantial net benefit, regardless of method. HPV testing with Digene Hybrid Capture 2 (HC2; Qiagen, German-town, Md.) is commonly performed in the United States, and HC2 and polymerase chain reaction–based methods have been evaluated in effectiveness trials. Although alternative HPV detection methods are emerging, the clinical comparability and implications of these methods are not completely understood.
SCREENING INTERVAL
Screening women 21 to 65 years of age every three years with cytology provides a reasonable balance between benefits and harms. Among women 30 to 65 years of age, HPV testing combined with cytology (co-testing) every five years offers a comparable balance of benefits and harms and is therefore a reasonable alternative for women in this age group who would prefer to extend the screening interval. Screening with cytology more often than every three years confers little additional benefit, with large increases in harms, including additional procedures and assessment and treatment of transient lesions. Treatment of lesions that would otherwise resolve on their own is harmful because it can lead to procedures with unwanted adverse effects, including the potential for cervical incompetence and preterm labor. Similarly, the frequency of HPV testing with cytology should not be more often than every five years to maintain a reasonable balance of benefits and harms similar to that seen with cytology alone every three years. Maintaining the comparability of the benefits and harms of co-testing and cytology alone demands that patients, clinicians, and health care organizations adhere to currently recommended screening intervals, protocols for repeat testing, cytologic thresholds for further diagnostic testing (i.e., colposcopy) and treatments, and extended surveillance as recommended by current American Cancer Society/American Society for Colposcopy and Cervical Pathology/American Society for Clinical Pathology (ACS/ASCCP/ASCP) guidelines.
Women choosing co-testing to increase their screening interval (and potentially decrease testing) should be aware that positive screening results are more likely with HPV-based strategies than with cytology alone, and that some women may require prolonged surveillance with additional frequent testing if they have persistently positive HPV results. Because HPV test results may be positive among women who would otherwise be advised to end screening at 65 years of age on the basis of previously normal cytology results alone, the likelihood of continued testing may increase with HPV testing. The percentage of U.S. women undergoing co-testing who will have a normal cytology test result and a positive HPV test result (and who will therefore require additional testing) ranges from 11 percent among women 30 to 34 years of age to 2.6 percent among women 60 to 65 years of age.5,6
TIMING OF SCREENING
Women younger than 21 years. Cervical cancer is rare before 21 years of age. The USPSTF found little evidence to determine whether and how sexual history should affect the age at which to begin screening. Although exposure of cervical cells to sexually transmitted HPV during vaginal intercourse may lead to cervical carcinogenesis, the process has multiple steps, involves regression, and is generally not rapid. There is evidence that screening before 21 years of age, regardless of sexual history, would lead to more harm than benefit.4 The harms are greater in this younger age group because abnormal test results are likely to be transient and to resolve on their own; in addition, resulting treatment may have an adverse effect on future childbearing.
Women older than 65 years. Clinicians and patients should base the decision to end screening on whether the patient meets the criteria for adequate prior testing and appropriate follow-up per established guidelines. The ACS/ASCCP/ASCP guidelines define adequate prior screening as three consecutive negative cytology results or two consecutive negative HPV results within 10 years before cessation of screening, with the most recent test occurring within five years. They further state that routine screening should continue for at least 20 years after spontaneous regression or appropriate management of a high-grade precancerous lesion, even if this extends screening past 65 years of age.7 The ACS further states that screening should not resume after cessation in women older than 65 years, even if a woman reports having a new sex partner.
Women older than 65 years who have never been screened. Screening may be clinically indicated in older women for whom the adequacy of prior screening cannot be accurately accessed or documented. Women with limited access to care, minority women, and women from countries where screening is not available may be less likely to meet the criteria for adequate prior screening. The USPSTF realizes that certain considerations may support screening in women older than 65 years who are otherwise considered high risk (such as women with a high-grade precancerous lesion or cervical cancer, women with in utero exposure to diethylstilbestrol, or women who are immunocompromised).
ASSESSMENT OF RISK
It is well established that HPV infection is associated with nearly all cases of cervical cancer. Other factors that put a woman at increased risk of cervical cancer include HIV infection, a compromised immune system, in utero exposure to diethylstilbestrol, and previous treatment of a high-grade precancerous lesion or cervical cancer.
Women who have had a hysterectomy with removal of the cervix and who do not have a history of a high-grade precancerous lesion or cervical cancer are not at risk of cervical cancer and should not be screened. Women who had their cervix removed during surgery for ovarian or endometrial cancer are not at high risk of cervical cancer and would not benefit from screening. Clinicians should confirm through review of surgical records or direct examination that the cervix was removed.
TREATMENT
Screening aims to identify high-grade precancerous cervical lesions to prevent development of cervical cancer and early-stage asymptomatic invasive cervical cancer. High-grade lesions may be treated with ablative and excisional therapies, including cryotherapy, laser ablation, loop excision, and cold-knife conization. Early-stage cervical cancer may be treated with surgery (hysterectomy) or chemoradiation. The treatment of precancerous rather than early-stage cancerous lesions is unique to cervical cancer and is the foundation of the success of cervical cancer screening. Treatment of precancerous lesions is less invasive than treatment of cancer and results in fewer adverse effects.
OTHER APPROACHES TO PREVENTION
Many individuals and clinicians have used the annual Pap smear screening visit as an opportunity to discuss other health problems and preventive measures. Individuals, clinicians, and health systems should seek effective ways to facilitate the receipt of recommended preventive services at intervals that are beneficial to the patient. Efforts should also be made to ensure that individuals are able to seek care for additional health concerns as they present.
The overall effect of HPV vaccination on high-grade precancerous cervical lesions and cervical cancer is not yet known. Current trials do not provide data on long-term efficacy8; therefore, the possibility that vaccination might reduce the need for screening with cytology alone or in combination with HPV testing is not established. Given these uncertainties, women who have been vaccinated should continue to be screened.

