This is a corrected version of the article that appeared in print.
Am Fam Physician. 2012;86(10):931-938
A more recent article on chlamydial and gonococcal infections is available.
Related letter: Testing Options for the Detection of Gonorrhea and Chlamydia
Patient information: See related handout on gonorrhea, written by the authors of this article.
Author disclosure: No relevant financial affiliations to disclose.
Neisseria gonorrhoeae causes urogenital, anorectal, conjunctival, and pharyngeal infections. Urogenital tract infections are most common. Men with gonorrhea may present with penile discharge and dysuria, whereas women may present with mucopurulent discharge or pelvic pain; however, women often are asymptomatic. Neonatal infections include conjunctivitis and scalp abscesses. If left untreated, gonorrhea may cause pelvic inflammatory disease in women, or it may disseminate, causing synovial and skin manifestations. Urogenital N. gonorrhoeae infection can be diagnosed using culture or nucleic acid amplification testing. Urine nucleic acid amplification tests have a sensitivity and specificity comparable to those of cervical and urethral samples. Fluoroquinolones are no longer recommended for the treatment of gonorrhea because of antimicrobial resistance. [corrected] First-line treatment for uncomplicated urogenital, anorectal, or pharyngeal gonococcal infections is a single intramuscular injection of ceftriaxone, 250 mg, accompanied by either azithromycin, 1 g orally, or doxycycline, 100 mg orally twice daily for seven days, to prevent bacterial resistance and address the likelihood of coinfection with Chlamydia trachomatis. This dosage is more effective for common pharyngeal infections than the previously recommended dose of 125 mg. Azithromycin may be used as an alternative treatment option for patients with previous allergic reactions to penicillin, but because of the likelihood of antimicrobial resistance, its use should be limited. Gonococcal infection should prompt physicians to test for other sexually transmitted infections, including human immunodeficiency virus. Because of high reinfection rates, patients should be retested in three to six months. The U.S. Preventive Services Task Force recommends screening for gonorrhea in all sexually active women at increased risk of infection. It also recommends intensive behavioral counseling for persons with or at increased risk of contracting sexually transmitted infections. Condom use is an effective strategy to reduce the risk of infection.
According to the Centers for Disease Control and Prevention (CDC), approximately 700,000 new cases of Neisseria gonorrhoeae infection occur annually in the United States, making it the second most frequently reported bacterial sexually transmitted infection (STI) after Chlamydia trachomatis.1 Reported rates of chlamydia and gonorrhea are highest among females 15 to 19 years of age.1 Incarcerated women younger than 35 years also exhibit high rates of infection.2
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| The USPSTF recommends that all newborns receive ocular topical medication for prophylaxis against gonococcal ophthalmia neonatorum. | A | 15 |
| Fluoroquinolones should not be used to treat gonorrhea in the United States because of the emergence of quinolone-resistant Neisseria gonorrhoeae. | C | 18 |
| [corrected] Uncomplicated gonococcal infections of the cervix, urethra, rectum, or pharynx should be treated with a single dose of ceftriaxone (Rocephin), 250 mg intramuscularly, and either azithromycin (Zithromax), 1 g orally, or doxycycline, 100 mg orally twice daily for seven days. | C | 1, 14 |
| Retesting for gonococcal infection is recommended three to six months after treatment in men and women to detect reinfection, regardless of partner treatment. | C | 23, 24 |
| The USPSTF recommends routine screening for gonorrhea in all sexually active women if they are at increased risk of infection, including those who are pregnant. | B | 3 |
| The USPSTF recommends against routine screening for gonorrhea in men and women who are at low risk of infection. | A | 3 |
| The CDC recommends that all men who have sex with men and practice insertive anal intercourse be screened at least annually for urethral gonococcal infection with a urine nucleic acid amplification test. | C | 1, 2 |
| The CDC recommends that all men who have sex with men and practice receptive anal or oral intercourse be screened at least annually for rectal or pharyngeal gonococcal infection with a rectal or pharyngeal nucleic acid amplification swab test. | C | 1, 2 |
| The USPSTF recommends high-intensity behavioral counseling to prevent sexually transmitted infections in all sexually active adolescents and in adults at increased risk. | B | 31 |
Gonococcal infections include urogenital, anorectal, pharyngeal, and conjunctival infections. Risk factors include high-risk sexual behavior and age younger than 25 years (Table 1).1–4 The differential diagnosis depends on the site of mucocutaneous infection (Table 2).1,2 Untreated infections may lead to pelvic inflammatory disease (PID), disseminated gonococcal infections, and, although rare, endocarditis or meningitis.5
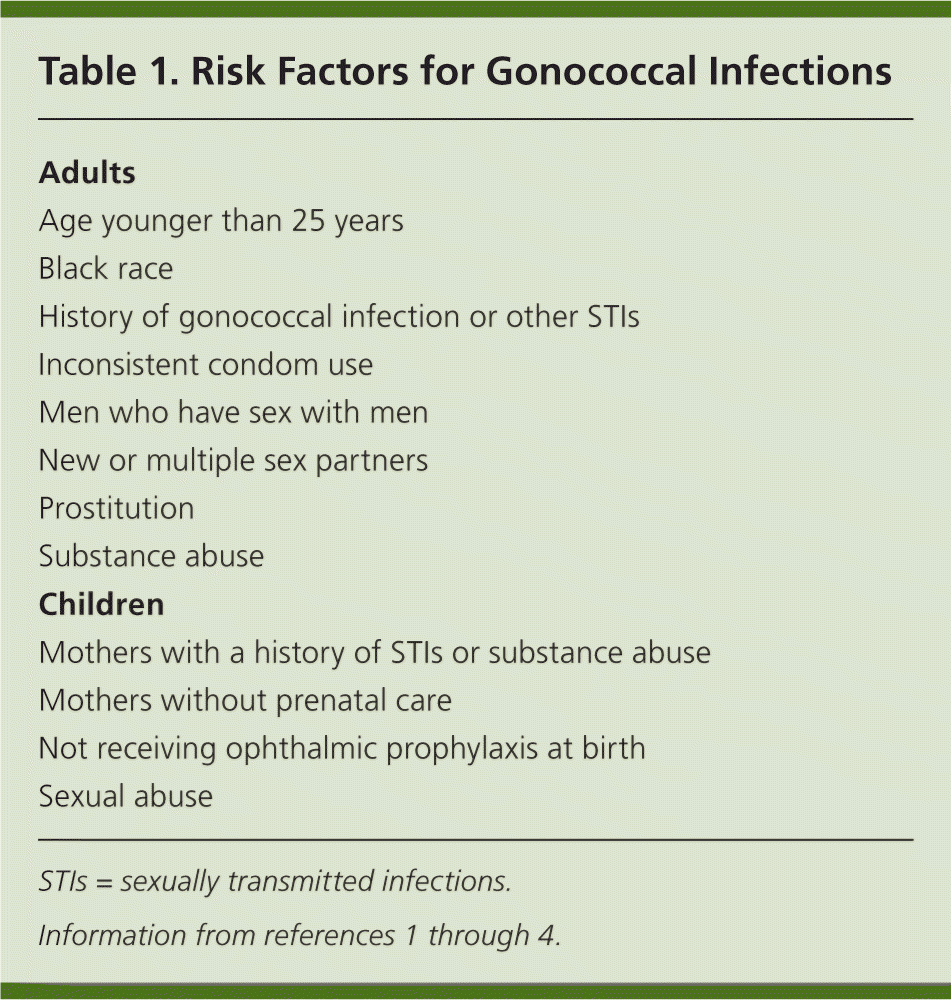
| Adults |
| Age younger than 25 years |
| Black race |
| History of gonococcal infection or other STIs |
| Inconsistent condom use |
| Men who have sex with men |
| New or multiple sex partners |
| Prostitution |
| Substance abuse |
| Children |
| Mothers with a history of STIs or substance abuse |
| Mothers without prenatal care |
| Not receiving ophthalmic prophylaxis at birth |
| Sexual abuse |

| Diagnosis | Causes | |
|---|---|---|
| Cervicitis | Bacterial vaginosis | |
| Chlamydia trachomatis infection | ||
| Frequent douching or exposure to another irritant | ||
| Genital herpes simplex virus infection | ||
| Mycoplasma genitalium infection | ||
| Trichomonas vaginalis infection | ||
| Neonatal conjunctivitis | C. trachomatis infection | |
| Moraxella catarrhalis infection | ||
| Other Neisseria species infection | ||
| Urethritis (nongonococcal) | Adenovirus | |
| C. trachomatis infection (15 to 40 percent) | ||
| Enteric bacteria (associated with insertive anal intercourse) | ||
| Genital herpes simplex virus | ||
| M. genitalium infection (15 to 25 percent) | ||
| Nonchlamydial nongonococcal urethritis | ||
| T. vaginalis infection | ||
| If urethritis persists after treatment, consider doxycycline-resistant Ureaplasma urealyticum or M. genitalium infection | ||
| If urethritis still persists, consider chronic nonbacterial prostatitis or chronic pelvic pain syndrome | ||
| Vaginal discharge | Common infections | |
| Candidiasis | ||
| Cervicitis | ||
| Gardnerella vaginalis | ||
| T. vaginalis | ||
| Less common infections | ||
| M. genitalium | ||
| Mobiluncus species | ||
| Prevotella species | ||
| U. urealyticum | ||
Urogenital Infections
INFECTIONS IN WOMEN
More than 95 percent of women with gonorrhea have no symptoms.4 Gonorrhea may infect the endocervix, Bartholin glands, Skene ducts, vagina, rectum, or pharynx.4 The most common manifestation is cervicitis, which usually occurs five to 10 days after exposure.6 Ten to 20 percent of women with cervical gonorrhea also have a pharyngeal infection.4 If symptoms occur, they are usually mild, and may mimic acute cystitis or vaginitis. Metrorrhagia also may be a sign of an acute gonococcal infection.4
Untreated gonorrhea causes 10 to 20 percent of PID cases, and 15 percent of women with PID develop infertility from tubal scarring.7 One-half of patients who have had three or more episodes of PID develop infertility.7 The CDC recommends that symptomatic women be examined for PID by palpating for cervical or adnexal tenderness, and tested for gonorrhea, chlamydia, bacterial vaginosis, and trichomoniasis with endocervical swabs for light microscopy and nucleic acid amplification testing. Because light microscopy is only 50 percent sensitive for Trichomonas vaginalis, the CDC recommends that culture also be obtained when a wet-mount preparation is undertaken.1
INFECTIONS IN MEN
Men with gonorrhea usually are symptomatic, but asymptomatic urethral infections may occur in at least 10 percent of cases.1,6 When symptoms occur, they typically appear two to five days after infection, but may take as long as 30 days to appear. Common signs and symptoms include dysuria and purulent penile discharge. Unilateral epididymitis without discharge also may be present.1,5
Chlamydia causes 15 to 40 percent of nongonococcal urethritis cases in men.8 One to 2 percent of men who have nongonococcal urethritis develop sexually acquired reactive arthritis (formerly known as Reiter syndrome), which also includes uveitis, and often skin and mucosal lesions.8,9 Sexually acquired reactive arthritis is associated more commonly with chlamydia and less commonly with gonococcal urethritis.8,9 The development of reactive arthritis correlates with the presence of human leukocyte antigen-B27, which is found in 90 to 96 percent of patients with reactive arthritis.8,9
Anorectal Infections
More than 50 percent of rectal N. gonorrhoeae infections in men and women may be asymptomatic.6 The highest prevalence of anorectal gonorrhea is in men who have sex with men.1 Symptoms include anal pruritus, rectal pain, mucopurulent discharge, and tenesmus.4 Untreated rectal infections cause symptomatic proctitis.4 The CDC recommends nucleic acid amplification testing as the preferred approach to evaluating pharyngeal and rectal specimens.1
Pharyngeal Infections
Gonorrhea can be transmitted through unprotected sexual contact with the oropharynx.1,10 Physicians should ask patients about oral sexual exposure if gonorrhea is suspected. Up to 90 percent of pharyngeal gonorrhea in men and women may be asymptomatic.6 Up to 20 percent of women with cervical gonorrhea are coinfected with pharyngeal gonorrhea.10 Approximately 11 to 26 percent of adolescent women with infection could remain undiagnosed without pharyngeal testing.4 Pharyngeal infections also are common in men who have sex with men, patients with human immunodeficiency virus (HIV) infection, and those presenting to STI clinics.
Physical examination findings may include oropharyngeal erythema or exudates, and cervical lymphadenopathy. Pharyngeal gonorrhea is more challenging to treat than urogenital and endocervical infections.4 Oral cephalosporins are up to 90 percent effective, compared with 99 percent for intramuscular ceftriaxone (Rocephin).11 Patients are rarely coinfected with pharyngeal chlamydia, but because concurrent genital coinfection is possible, the CDC recommends treatment for both.1
Disseminated Infection
Disseminated gonococcal infection is rare, affecting 0.4 to 3 percent of patients with gonorrhea,4 but it is the most common cause of infectious arthritis in sexually active, previously healthy patients. Because about 15 percent of patients presenting in a primary care setting have joint pain as a primary symptom, disseminated gonococcal infection should be considered.12 Antibioticresponsive, culture-negative acute arthritis in a sexually active young person is most often caused by disseminated gonococcal infection.1 Skin lesions are present in 75 percent of patients with bacteremia, including painless or painful petechiae, macules, papules, pustules, vesicles, and bullae.12,13 At presentation, some patients have asymmetric arthropathies or polyarthralgia. Joints commonly affected with tenosynovitis or septic arthritis include wrists, ankles, hands, and feet. Rare disease progression may result in perihepatitis, meningitis, or endocarditis.12,13
Infections in Infants
Gonococcal infections can occur in neonates from exposure to infected cervical secretions during delivery.5 Sepsis, neonatal conjunctivitis (ophthalmia neonatorum), meningitis, and arthritis are the most severe complications.1 Other manifestations include pharyngitis, rhinitis, vaginitis, urethritis, and, rarely, pneumonia.1,5 Infants may develop localized scalp infections or abscesses from open wounds, such as those caused by fetal scalp electrodes. Up to 30 percent of cases of ophthalmia neonatorum are caused by C. trachomatis, although gonorrhea still accounts for two or three cases in every 10,000 live births. Other bacteria (e.g., Haemophilus influenzae, Escherichia coli, Staphylococcus aureus) also may cause ophthalmia neonatorum.
Gonococcal conjunctivitis has an average incubation period of six days. Gram stain smears of conjunctival exudates showing elevated white blood cell counts or gramnegative intracellular diplococci are sufficient for presumptive treatment, but only gonococcal cultures provide a definitive diagnosis. Culture should be performed in the infant and mother, including testing for chlamydia. Blood culture or Gram stain smears of cerebrospinal fluid or joint aspirate may be necessary if sepsis is suspected. Treatment of ophthalmia neonatorum is important to prevent globe perforation and blindness1,2 (Table 34,14 ). The U.S. Preventive Services Task Force (USPSTF) recommends that all newborns receive ocular topical medication for prophylaxis against gonococcal ophthalmia neonatorum.15
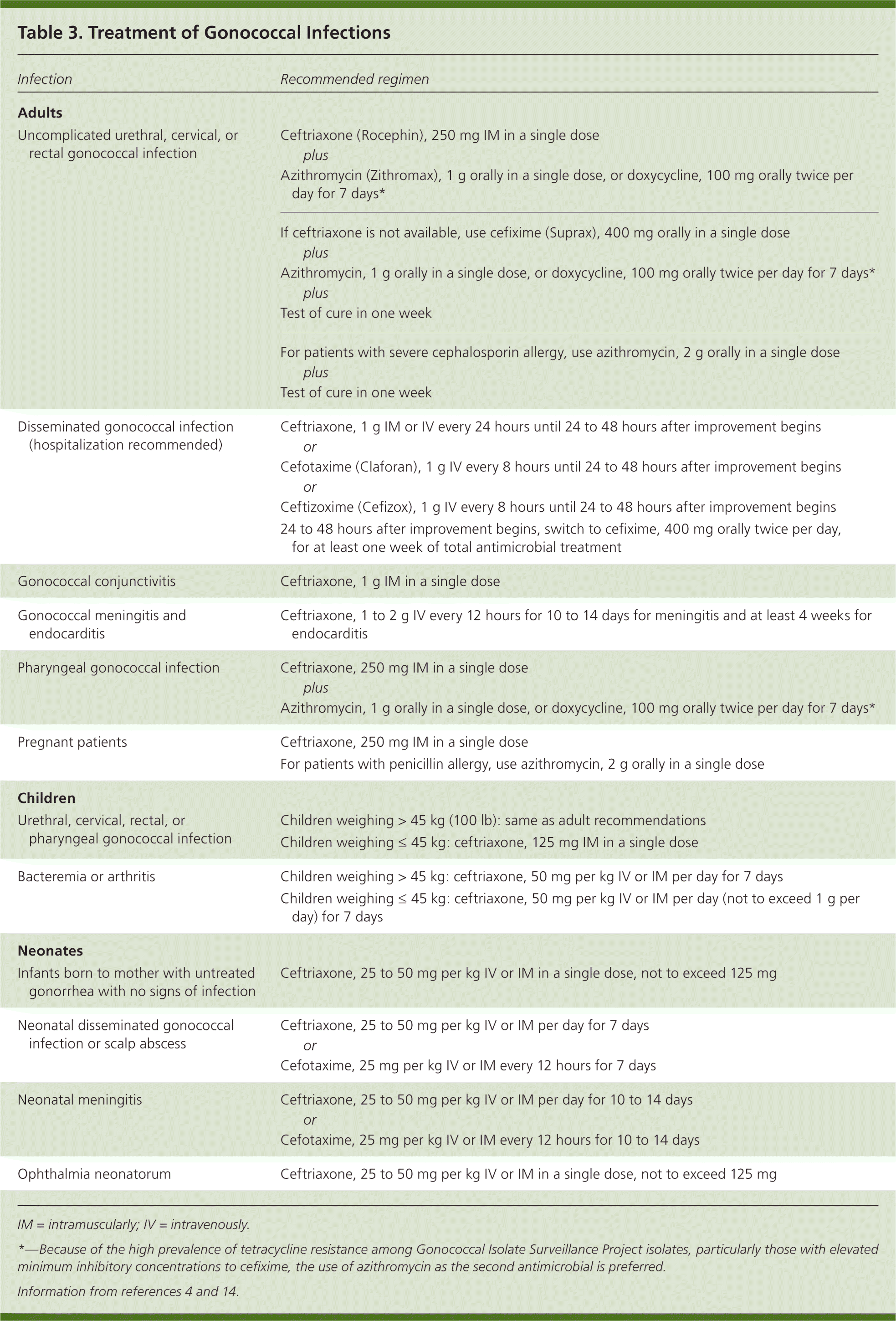
| Infection | Recommended regimen | |
|---|---|---|
| Adults | ||
| Uncomplicated urethral, cervical, or rectal gonococcal infection | Ceftriaxone (Rocephin), 250 mg IM in a single dose | |
| plus | ||
| Azithromycin (Zithromax), 1 g orally in a single dose, or doxycycline, 100 mg orally twice per day for 7 days* | ||
| If ceftriaxone is not available, use cefixime (Suprax), 400 mg orally in a single dose | ||
| plus | ||
| Azithromycin, 1 g orally in a single dose, or doxycycline, 100 mg orally twice per day for 7 days* | ||
| plus | ||
| Test of cure in one week | ||
| For patients with severe cephalosporin allergy, use azithromycin, 2 g orally in a single dose | ||
| plus | ||
| Test of cure in one week | ||
| Disseminated gonococcal infection (hospitalization recommended) | Ceftriaxone, 1 g IM or IV every 24 hours until 24 to 48 hours after improvement begins | |
| or | ||
| Cefotaxime (Claforan), 1 g IV every 8 hours until 24 to 48 hours after improvement begins | ||
| or | ||
| Ceftizoxime (Cefizox), 1 g IV every 8 hours until 24 to 48 hours after improvement begins 24 to 48 hours after improvement begins, switch to cefixime, 400 mg orally twice per day, for at least one week of total antimicrobial treatment | ||
| Gonococcal conjunctivitis | Ceftriaxone, 1 g IM in a single dose | |
| Gonococcal meningitis and endocarditis | Ceftriaxone, 1 to 2 g IV every 12 hours for 10 to 14 days for meningitis and at least 4 weeks for endocarditis | |
| Pharyngeal gonococcal infection | Ceftriaxone, 250 mg IM in a single dose | |
| plus | ||
| Azithromycin, 1 g orally in a single dose, or doxycycline, 100 mg orally twice per day for 7 days* | ||
| Pregnant patients | Ceftriaxone, 250 mg IM in a single dose | |
| For patients with penicillin allergy, use azithromycin, 2 g orally in a single dose | ||
| Children | ||
| Urethral, cervical, rectal, or pharyngeal gonococcal infection | Children weighing > 45 kg (100 lb): same as adult recommendations | |
| Children weighing ≤ 45 kg: ceftriaxone, 125 mg IM in a single dose | ||
| Bacteremia or arthritis | Children weighing > 45 kg: ceftriaxone, 50 mg per kg IV or IM per day for 7 days | |
| Children weighing ≤ 45 kg: ceftriaxone, 50 mg per kg IV or IM per day (not to exceed 1 g per day) for 7 days | ||
| Neonates | ||
| Infants born to mother with untreated gonorrhea with no signs of infection | Ceftriaxone, 25 to 50 mg per kg IV or IM in a single dose, not to exceed 125 mg | |
| Neonatal disseminated gonococcal infection or scalp abscess | Ceftriaxone, 25 to 50 mg per kg IV or IM per day for 7 days | |
| or | ||
| Cefotaxime, 25 mg per kg IV or IM every 12 hours for 7 days | ||
| Neonatal meningitis | Ceftriaxone, 25 to 50 mg per kg IV or IM per day for 10 to 14 days | |
| or | ||
| Cefotaxime, 25 mg per kg IV or IM every 12 hours for 10 to 14 days | ||
| Ophthalmia neonatorum | Ceftriaxone, 25 to 50 mg per kg IV or IM in a single dose, not to exceed 125 mg | |
Infections in Children
In preadolescent children, gonococcal infection is likely indicative of sexual abuse. Vaginitis is the presenting symptom in preadolescent girls.1 Pharyngeal and rectal infections may coexist with vaginitis but are often asymptomatic. Specimens from all sites should be collected. Gram stains are inadequate for diagnosis in children. Culture techniques remain the preferred testing modality, and nucleic acid amplification testing may be appropriate if collecting culture swabs is not feasible.1,2 If gonorrhea is suspected, children also should be evaluated for coinfection with HIV, chlamydia, and syphilis.
Diagnostic Tests
Gonorrhea is characterized by Gram stain of a urethral smear or cervical swab showing gram-negative intracellular diplococci. Speculum examination may not be necessary to diagnose gonorrhea in women because of the comparable effectiveness of blind vaginal swabs.16 Urine nucleic acid amplification testing in women and men (and urine polymerase chain reaction testing in men) has comparable sensitivity and specificity to cervical and urethral samples. Urine polymerase chain reaction testing in women has a 55.6 percent sensitivity,17 which is too low to recommend routine use. Cultures from skin lesions in disseminated gonococcal infections are usually negative, but joint aspirate may have greater yield. The workup should include evaluation for other causes of arthritis, and oral, genital, blood, and fluid aspirate cultures.12,13 Table 4 lists the diagnostic features of gonococcal infections.1,2,4

| Infection | Diagnostic features | |
|---|---|---|
| Cervicitis | History | |
| Dyspareunia | ||
| Vaginal bleeding after sex | ||
| Physical examination | ||
| Inguinal lymphadenopathy | ||
| Mucopurulent or purulent endocervical exudate | ||
| Sustained endocervical bleeding easily induced by swab | ||
| Endocervical Gram stain showing increased PMNL count or leukorrhea with more than 10 WBCs per high-power field (low positive predictive value) and gram-negative intracellular diplococci (50 percent of women) | ||
| Positive culture or nucleic acid amplification test | ||
| Gonococcal conjunctivitis (ophthalmia neonatorum) | Conjunctival erythema with mucopurulent eye discharge | |
| Gram stain smear of conjunctival exudates showing elevated PMNL count or gram-negative intracellular diplococci | ||
| Gonococcal culture | ||
| Pelvic inflammatory disease | Minimal criteria | |
| Cervical motion, uterine, or adnexal tenderness | ||
| Additional criteria | ||
| Abnormal cervical or vaginal mucopurulent discharge | ||
| Temperature > 101oF (38.3oC) | ||
| Laboratory confirmation of cervical infection with Neisseria gonorrhoeae or Chlamydia trachomatis | ||
| Elevated erythrocyte sedimentation rate | ||
| Elevated C-reactive protein level | ||
| WBCs present on saline microscopy (wet-mount preparation) of vaginal secretions | ||
| Specific criteria | ||
| Endometrial biopsy with histopathologic evidence of endometritis | ||
| Transvaginal sonography or magnetic resonance imaging showing thickened, fluid-filled tubes with or without free pelvic fluid or tubo-ovarian complex, or Doppler studies suggesting pelvic infection | ||
| Laparoscopic abnormalities consistent with pelvic inflammatory disease | ||
| Urethritis | Mucopurulent or purulent discharge on examination | |
| Urethral smear with Gram stain showing more than five PMNLs per oil immersion field and WBCs containing gram-negative intracellular diplococci | ||
| First void urine with positive leukocyte esterase on urine dip and microscopic examination with more than 10 WBCs per high-power field | ||
| Positive culture or nucleic acid amplification test | ||
Treatment
Treatment of gonococcal infections is outlined in Table 3.4,14 Patients' sex partners within 60 days before symptom onset should also be treated. Fluoroquinolones are not recommended in the United States for treatment of gonorrhea or associated conditions because of the emergence of quinolone-resistant N. gonorrhoeae.1,18 [corrected] Ceftriaxone, 250 mg in a single intramuscular injection, plus either azithromycin (Zithromax), 1 g orally, or doxycycline, 100 mg orally twice daily for seven days, is effective for treating uncomplicated cervical, urethral, anorectal, and pharyngeal infections.11 This dosage is recommended over the 125-mg dose because of reported treatment failures and limited effectiveness for treating pharyngeal infections at the lower dose. Although doxycycline is an alternative regimen in combination with a cephalosporin, azithromycin is preferred because of the high prevalence of tetracycline resistance.14 Up to 10 percent of persons with penicillin allergies have cross-reactivity to first-generation cephalosporins; however, cross-reactivity to third-generation cephalosporins is less common.19 A history of severe reaction to penicillin, such as anaphylaxis or toxic epidermal necrolysis, should be the only contraindication. The CDC recommends consultation with an infectious disease specialist for patients with a history of severe allergic reactions to cephalosporins. Desensitization is not recommended.1
Patients with persistent symptoms after treatment should be evaluated by culture and tested for antimicrobial susceptibility.1 The CDC recommends empiric, broad-spectrum coverage for treating suspected PID to avoid sequelae.1 Hospitalization is recommended for initiation of therapy for disseminated gonococcal infection or for patients who might not be compliant.1 Treatment of sexually acquired reactive arthritis includes rest with restriction of physical activity, physical therapy to prevent muscle wasting, cold compresses and nonsteroidal anti-inflammatory drugs, and antimicrobial therapy for three months or longer.8,9
Follow-up
Writing patients a prescription to give to their sex partners (known as expedited partner therapy or patient-delivered partner therapy), or contacting patients' partners directly to offer treatment without an examination, reduces the risk of recurrent infection with gonorrhea.20 Because expedited or patient-delivered partner therapy is prohibited in some states, physicians should consult the CDC to obtain updated information on individual jurisdictions (http://www.cdc.gov/std/ept).21 The CDC recommends that any patient-delivered partner therapy medication or prescription be accompanied by treatment instructions, appropriate warnings, general health counseling, and a statement advising that partners seek personal medical evaluation.1 Patient-delivered partner therapy has been shown to reduce the recurrence of gonorrhea at follow-up by about 50 percent.22
Retesting men and women is recommended three to six months after treatment, regardless of partner treatment, because of high rates of reinfection within six months of therapy.1,23,24 If using alternative regimens, such as cefixime (Suprax) when ceftriaxone is unavailable or azithromycin in the case of severe penicillin or cephalosporin allergy, the CDC now recommends a test of cure within one week.14 Co-treatment for chlamydial infection is recommended, even if the nucleic acid amplification test result is negative for chlamydia.2 Pregnant women with first-trimester gonococcal infection should be retested within three to six months, in addition to routine test of cure, preferably in the third trimester. Uninfected pregnant women who remain at high risk should be retested during the third trimester.1
As with other STIs, gonorrhea increases shedding of HIV. Treatment of gonorrhea reduces HIV shedding and subsequent transmission.25 All patients who test positive for gonorrhea should be tested for other STIs, including chlamydia, syphilis, and HIV. The CDC recommends that patients with gonococcal infections be treated routinely with a regimen effective against chlamydia.26,27
Screening and Prevention
At-risk patients and those in a high-prevalence area should undergo routine screening for STIs.17 The USPSTF recommends routine screening for gonorrhea in all sexually active women at increased risk of infection, including during pregnancy, but recommends against screening low-risk men and women.3 Routine screening for gonorrhea and other curable STIs should be performed at least annually in sexually active patients with HIV infection. The CDC recommends that men who have sex with men be screened at least annually for gonorrhea at urethral, rectal, and pharyngeal sites for a history of insertive anal, receptive anal, and receptive oral intercourse, respectively.1,2 Screening for urethral infections should be performed with urine nucleic acid amplification testing, whereas rectal or pharyngeal screening should be performed with nucleic acid amplification swab. Screening every three to six months also is recommended for men who have sex with men if they have multiple or anonymous partners because they are at highest risk of contracting STIs.28
Condom use can reduce the risk of gonorrhea and other STIs. It also reduces the risk of PID by decreasing lower genital tract infections.29 According to observational studies, using a diaphragm may protect against cervical gonorrhea.30 Individual and group-based prevention counseling effectively reduce the risk of contracting gonorrhea. The USPSTF recommends high-intensity behavioral counseling for all sexually active adolescents and for adults at increased risk of STIs.31 High-intensity behavioral counseling refers to multiple sessions in a primary care or STI clinic setting. Intensive counseling also increases adherence to treatment in women and contraceptive use in adolescent males, and decreases nonsexual risky behavior and pregnancy in sexually active adolescent females.32 Culturally appropriate intensive behavioral interventions may be more effective than standard prevention counseling for reducing the rate of reinfection with gonorrhea (number needed to treat = 12).33
Data Sources: A PubMed search was conducted for gonorrhea-related topics, including clinical reviews, randomized controlled trials, and meta-analyses. Search terms used were Neisseria gonorrhoeae, gonorrhea, and sexually transmitted infections. Also reviewed were relevant publications from the Cochrane database, National Guideline Clearinghouse, U.S. Preventive Services Task Force, Centers for Disease Control and Prevention, and Dynamed. Search date: February 1, 2011.