Diabetic ketoacidosis is characterized by a serum glucose level greater than 250 mg per dL, a pH less than 7.3, a serum bicarbonate level less than 18 mEq per L, an elevated serum ketone level, and dehydration. Insulin deficiency is the main precipitating factor. Diabetic ketoacidosis can occur in persons of all ages, with 14 percent of cases occurring in persons older than 70 years, 23 percent in persons 51 to 70 years of age, 27 percent in persons 30 to 50 years of age, and 36 percent in persons younger than 30 years. The case fatality rate is 1 to 5 percent. About one-third of all cases are in persons without a history of diabetes mellitus. Common symptoms include polyuria with polydipsia (98 percent), weight loss (81 percent), fatigue (62 percent), dyspnea (57 percent), vomiting (46 percent), preceding febrile illness (40 percent), abdominal pain (32 percent), and polyphagia (23 percent). Measurement of A1C, blood urea nitrogen, creatinine, serum glucose, electrolytes, pH, and serum ketones; complete blood count; urinalysis; electrocardiography; and calculation of anion gap and osmolar gap can differentiate diabetic ketoacidosis from hyperosmolar hyperglycemic state, gastroenteritis, starvation ketosis, and other metabolic syndromes, and can assist in diagnosing comorbid conditions. Appropriate treatment includes administering intravenous fluids and insulin, and monitoring glucose and electrolyte levels. Cerebral edema is a rare but severe complication that occurs predominantly in children. Physicians should recognize the signs of diabetic ketoacidosis for prompt diagnosis, and identify early symptoms to prevent it. Patient education should include information on how to adjust insulin during times of illness and how to monitor glucose and ketone levels, as well as information on the importance of medication compliance.
Diabetic ketoacidosis (DKA) continues to have high rates of morbidity and mortality despite advances in the treatment of diabetes mellitus. In a study of 4,807 episodes of DKA, 14 percent occurred in persons older than 70 years, 23 percent in persons 51 to 70 years of age, 27 percent in persons 30 to 50 years of age, and 36 percent in persons younger than 30 years.1 In a second study of 28,770 persons younger than 20 years (mean age of 14 years) with diabetes, 94 percent had no episodes of DKA, 5 percent had one episode, and 1 percent had at least two episodes.2 Additionally, DKA occurred more often in females, in persons with a migration background, and in persons 11 to 15 years of age.2 DKA has a case fatality rate of 1 to 5 percent.3,4 Although the highest rate of mortality is in older adults and persons with comorbid conditions, DKA is the leading cause of death in persons younger than 24 years with diabetes, most often because of cerebral edema.1,4
Although persons with DKA typically have a history of diabetes, 27 to 37 percent have newly diagnosed diabetes.5,6 This is especially true in young children. Most persons with DKA have type 1 diabetes. There is also a subgroup of persons with type 2 diabetes who have ketosis-prone diabetes; this subgroup represents 20 to 50 percent of persons with DKA.7 Persons with ketosis-prone diabetes have impaired insulin secretion; however, with proper glucose management, beta cell function improves and the clinical course resembles that of type 2 diabetes.8 These persons are often black or Latino, male, middle-aged, overweight or obese, have a family history of diabetes, and have newly diagnosed diabetes.9
SORT: KEY RECOMMENDATIONS FOR PRACTICE
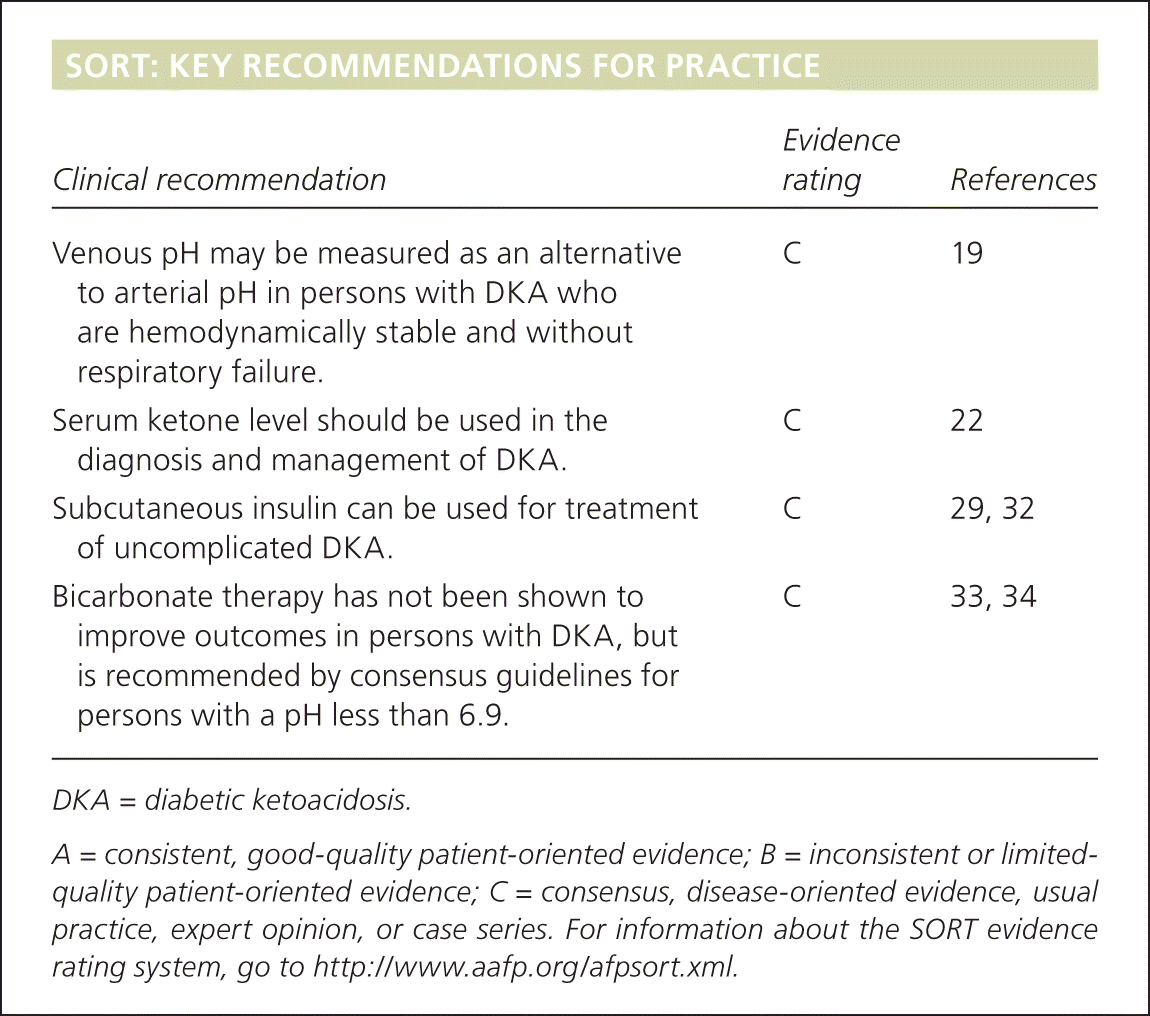
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Venous pH may be measured as an alternative to arterial pH in persons with DKA who are hemodynamically stable and withoutrespiratory failure. | C | 19 |
| Serum ketone level should be used in the diagnosis and management of DKA. | C | 22 |
| Subcutaneous insulin can be used for treatment of uncomplicated DKA. | C | 29, 32 |
| Bicarbonate therapy has not been shown to improve outcomes in persons with DKA, but is recommended by consensus guidelines for persons with a pH less than 6.9. | C | 33, 34 |
DKA = diabetic ketoacidosis.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Pathophysiology
DKA results from insulin deficiency from new-onset diabetes, insulin noncompliance, prescription or illicit drug use, and increased insulin need because of infection (Table 1).4,10–16 This insulin deficiency stimulates the elevation of the counterregulatory hormones (glucagon, catecholamines, cortisol, and growth hormone). Without the ability to use glucose, the body needs alternative energy sources. Lipase activity increases, causing a breakdown of adipose tissue that yields free fatty acids. These components are converted to acetyl coenzyme A, some of which enter the Krebs cycle for energy production; the remainder are broken down into ketones (acetone, acetoacetate, and β-hydroxybutyrate). Ketones can be used for energy, but accumulate rapidly. Glycogen and proteins are catabolized to form glucose. Together, these factors promote hyperglycemia, which leads to an osmotic diuresis resulting in dehydration, metabolic acidosis, and a hyperosmolar state (eFigure A).
Table 1. Causes of Diabetic Ketoacidosis

| Drugs |
| Antipsychotic agents: clozapine (Clozaril),10 olanzapine (Zyprexa),11 risperidone (Risperdal)12 |
| Illicit drugs (cocaine13) and alcohol4 |
| Others: corticosteroids, glucagon, interferon,14 pentamidine,14 sympathomimetic agents,14 thiazide diuretics4 |
| Infection |
| Pneumonia, sepsis, urinary tract infection |
| Lack of insulin |
| Insulin pump failure |
| Nonadherence to insulin treatment plans: body image issues,15 financial problems, psychological factors |
| Unrecognized symptoms of new-onset diabetes mellitus |
| Other physiologic stressors |
| Acromegaly,14 arterial thrombosis,14 cerebrovascular accident, Cushing disease,16 hemochromatosis,14 myocardial infarction, pancreatitis,14 pregnancy,14 psychological stress,16 shock/hypovolemia, trauma16 |
eFigure A. Pathophysiology of Diabetic Ketoacidosis
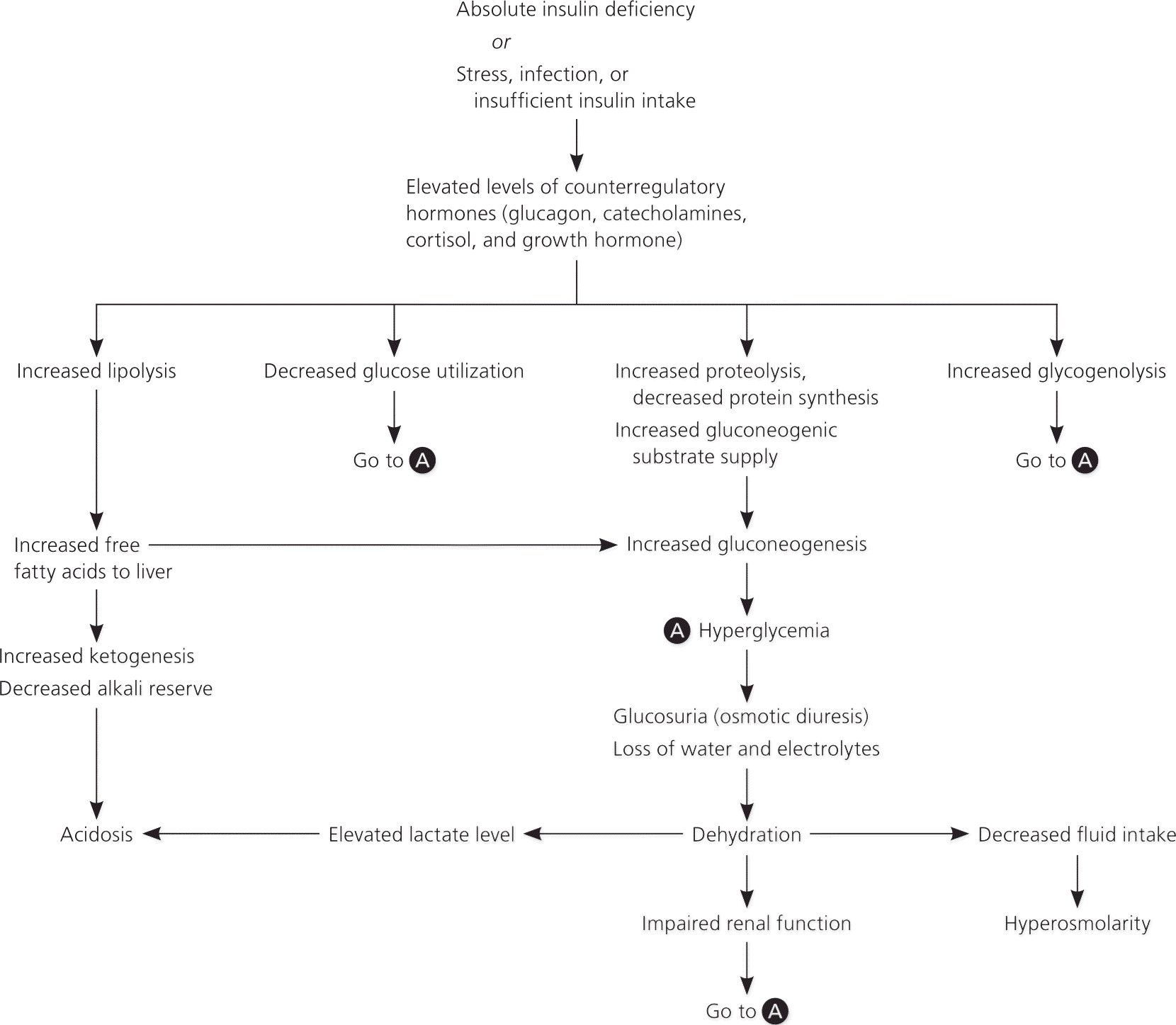
Adapted with permission from Wolfsdorf J, Glaser N, Spearing MA. Diabetic ketoacidosis in infants, children, and adolescents: A consensus statement from the American Diabetes Association. Diabetes Care. 2006;29(5):1151. Copyright 2006 American Diabetes Association.
Diagnosis
TYPICAL CLINICAL PRESENTATION
The presentation of DKA varies with severity and comorbid conditions. Polyuria with polydipsia is the most common presenting symptom and was found in 98 percent of persons in one study of childhood type 1 diabetes. Other common symptoms included weight loss (81 percent), fatigue (62 percent), dyspnea (57 percent), vomiting (46 percent), preceding febrile illness (40 percent), abdominal pain (32 percent), and polyphagia (23 percent).17 Dehydration causes tachycardia, poor skin turgor, dry mucous membranes, and orthostatic hypotension. The metabolic acidosis may lead to compensatory deep (Kussmaul) respirations, whereas increased acetone can be sensed as a fruity smell on the patient's breath. Mental status can vary from somnolence to lethargy and coma. A detailed evaluation may reveal precipitating factors, especially nonadherence to medical regimens and infection, which are common causes of DKA.
DIFFERENTIAL DIAGNOSIS
Although hyperosmolar hyperglycemic state can be confused with DKA, ketone levels are low or absent in persons with hyperosmolar hyperglycemic state. Other causes of high anion gap metabolic acidosis, such as alcoholic ketoacidosis and lactic acidosis, must be ruled out. Table 2 provides the differential diagnosis of DKA.14,18
Table 2. Differential Diagnosis of Diabetic Ketoacidosis
DIAGNOSTIC TESTING
The diagnosis of DKA (Table 3) is based on an elevated serum glucose level (greater than 250 mg per dL [13.88 mmol per L]), an elevated serum ketone level, a pH less than 7.3, and a serum bicarbonate level less than 18 mEq per L (18 mmol per L).4 Although arterial blood gas measurement remains the most widely recommended test for determining pH, measurement of venous blood gas has gained acceptance. One review indicated that venous and arterial pH are clinically interchangeable in persons who are hemodynamically stable and without respiratory failure.19 Traditionally, the severity of DKA is determined by the arterial pH, bicarbonate level, anion gap, and mental status of the patient (Table 3).4 An anion gap greater than 16 mEq per L (16 mmol per L) confirms metabolic acidosis. Although persons with DKA usually have a glucose level greater than 250 mg per dL, a few case reports document DKA in pregnant women who were euglycemic.20,21 Persons with hyperglycemia have pseudohyponatremia, and serum sodium concentration should be corrected. Table 4 provides formulas to calculate the anion gap, serum osmolality, osmolar gap, and serum sodium correction.16 [corrected]
Table 3. Diagnostic Criteria for Diabetic Ketoacidosis and Hyperosmolar Hyperglycemic State

| Criterion | Diabetic ketoacidosis | Hyperosmolar hyperglycemic state | ||
|---|---|---|---|---|
| Mild (serum glucose > 250 mg per dL [13.88 mmol per L]) | Moderate (serum glucose > 250 mg per dL) | Severe (serum glucose > 250 mg per dL) | Serum glucose > 600 mg per dL (33.30 mmol per L) | |
| Anion gap* | > 10 mEq per L (10 mmol per L) | > 12 mEq per L (12 mmol per L) | > 12 mEq per L (12 mmol per L) | Variable |
| Arterial pH | 7.24 to 7.30 | 7.00 to < 7.24 | < 7.00 | > 7.30 |
| Effective serum osmolality* | Variable | Variable | Variable | > 320 mOsm per kg (320 mmol per kg) |
| Mental status | Alert | Alert/drowsy | Stupor/coma | Stupor/coma |
| Serum bicarbonate | 15 to 18 mEq per L (15 to 18 mmol per L) | 10 to < 15 mEq per L (10 to < 15 mmol per L) | < 10 mEq per L (10 mmol per L) | > 18 mEq per L (18 mmol per L) |
| Serum ketone† | Positive | Positive | Positive | Small |
| Urine ketone† | Positive | Positive | Positive | Small |
*—Calculated osmolality and anion gap.
†—Nitroprusside (Nitropress) reaction method.
Adapted with permission from Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN. Hyperglycemic crisis in adult patients with diabetes. Diabetes Care. 2009;32(7):1336. Copyright 2009 American Diabetes Association.
Table 4. Calculations for the Evaluation of Diabetic Ketoacidosis

| Value | Purpose | Formula | Normal value |
|---|---|---|---|
| Anion gap | Essential for evaluation of acid base disorders | Na– (Cl + HCO3) | 7 to 13 mEq per L (7 to 13 mmol per L) |
| Osmolar gap | Difference between measured osmolality and calculated osmolality | Osmolality (measured) – osmolality (calculated) | < 10 mmol per L* |
| Serum osmolality | Measure of particles in a fluid compartment | 2(Na + K) + (glucose/18) + (blood urea nitrogen/2.8) | 285 to 295 mOsm per kg (285 to 295 mmol per kg) of water |
| Serum sodium correction | Hyperglycemia causes pseudohyponatremia | Na + 0.016(glucose – 100) | 135 to 140 mEq per L (135 to 140 mmol per L) |
Cl = chloride; HCO3= bicarbonate; K = potassium; Na = sodium.
*—Varies by institution.
Adapted with permission from Wilson JF. In clinic. Diabetic ketoacidosis. Ann Intern Med. 2010;152(1):ITC1-1–ITC1-15.
Urinalysis measures only acetone and acetoacetate, not β-hydroxybutyrate, which is the primary ketone in DKA. In one study, the urine dipstick test was negative for ketones in six of 18 persons. Ketonemia was defined as a ketone level greater than 0.42 mmol per L.22 In a second study of point-of-care testing in the emergency department, urine dipstick testing for ketones had a sensitivity of 98 percent, specificity of 35 percent, and a positive predictive value of 15 percent. Serum testing for β-hydroxybutyrate had a sensitivity of 98 percent, a specificity of 79 percent, and a positive predictive value of 34 percent (using a cutoff of greater than 1.5 mmol per L), allowing for more accurate diagnosis of DKA.23 The American Diabetes Association has revised its position on ketone analysis in favor of serum testing, and has concluded that capillary measurement is equivalent to venous measurement.4,22,24
Further initial laboratory studies should include measurement of electrolytes, phosphate, blood urea nitrogen, and creatinine; urinalysis; complete blood count with differential; and electrocardiography (Table 5).16 Potassium level is normal or low in persons with DKA, despite renal losses caused by the acidic environment. An initial potassium level less than 3.3 mEq per L (3.3 mmol per L) indicates profound hypokalemia. Amylase and lipase levels may be increased in persons with DKA, even in those without associated pancreatitis; however, 10 to 15 percent of persons with DKA do have concomitant pancreatitis.18,25
Table 5. Suggested Laboratory Evaluation for Persons with Diabetic Ketoacidosis

| Test | Comments |
|---|---|
| For all patients | |
| A1C | To determine level of glycemic control in persons with diabetes mellitus |
| Anion gap (electrolytes) | Usually greater than 15 mEq per L (15 mmol per L) |
| Arterial blood gas measurement | Below 7.3 |
| Arterial blood gas measurement is the most widely recommended test for determining pH, but measurement of venous blood gas has gained acceptance | |
| Blood urea nitrogen, creatinine levels | Usually elevated because of dehydration and decreased renal perfusion |
| Complete blood count (with differential) | May be elevated in persons with DKA, but without pancreatitis |
| Diagnosis of pancreatitis should be based on clinical judgment and imaging | |
| Electrocardiography | Assesses effect of potassium status; rules out ischemia or myocardial infarction |
| Serum bicarbonate level | Less than 18 mEq per L (18 mmol per L) |
| Serum glucose level | Point-of-care testing at presentation |
| Usually greater than 250 mg per dL (13.88 mmol per L) | |
| Pregnant women may have low to normal levels | |
| Serum ketone level | Point-of-care testing at presentation |
| Usually 7 to 10 mmol per L, or greater than 1:2 dilution | |
| Serum magnesium level | Can be low or normal because of osmotic diuresis |
| Serum osmolality | Greater than 320 mOsm per kg (320 mmol per kg) |
| Serum phosphate level | May be normal or elevated initially, but usually decreases with treatment |
| Serum potassium level | May be low, normal, or elevated |
| Serum sodium level | Usually low |
| Patient may have pseudohyponatremia that should be corrected | |
| Urinalysis | Confirms the presence of glucose and ketones, and will help assess for presence of a urinary tract infection |
| If clinically indicated | |
| Chest radiography | Perform if pneumonia or pulmonary disorder is suspected |
| Serum amylase/lipase level | May be elevated in persons with DKA, even in those without associated pancreatitis |
| Diagnosis of pancreatitis should be based on clinical judgment and imaging | |
| Serum creatine kinase and troponin levels | May be elevated in persons with DKA in the absence of myocardial infarction |
| Diagnosis of myocardial infarction should be based on clinical judgment and imaging | |
| Serum hepatic transaminase levels | Mild increases can occur, especially in persons with fatty liver disease |
| Urine and blood cultures | Perform if infection is suspected |
DKA = diabetic ketoacidosis.
Adapted with permission from Wilson JF. In clinic. Diabetic ketoacidosis. Ann Intern Med. 2010;152(1):ITC1-1–ITC1-15.
Leukocytosis can occur even in the absence of infection; bandemia more accurately predicts infection. One study showed that an elevated band count in persons with DKA had a sensitivity for predicting infection of 100 percent (19 out of 19 cases) and a specificity of 80 percent.26 Chest radiography and urine and blood cultures should be added for further evaluation of infection. An elevated hemoglobin level caused by dehydration may also exist. Elevated hepatic transaminase levels may occur, especially in persons with fatty liver disease.27 Mild increases in creatine kinase and troponin levels may occur in the absence of myocardial damage; one study demonstrated that increased troponin levels occurred in 26 out of 96 persons with DKA without a coronary event.28 Finally, the A1C level indicates the degree of glycemic control in persons known to have diabetes.
Treatment
Figure 14,29 provides the treatment approach for DKA in adults, and Figure 224,30 provides the treatment approach for DKA in persons younger than 20 years. Both approaches are recommended by the American Diabetes Association. Specific issues for the adult patient are discussed in more detail below. For persons younger than 20 years, insulin should be administered gradually, and fluid and electrolyte replacement should be done cautiously because of limited data and concern for precipitating cerebral edema.
Figure 1. Management of Diabetic Ketoacidosis in Adults
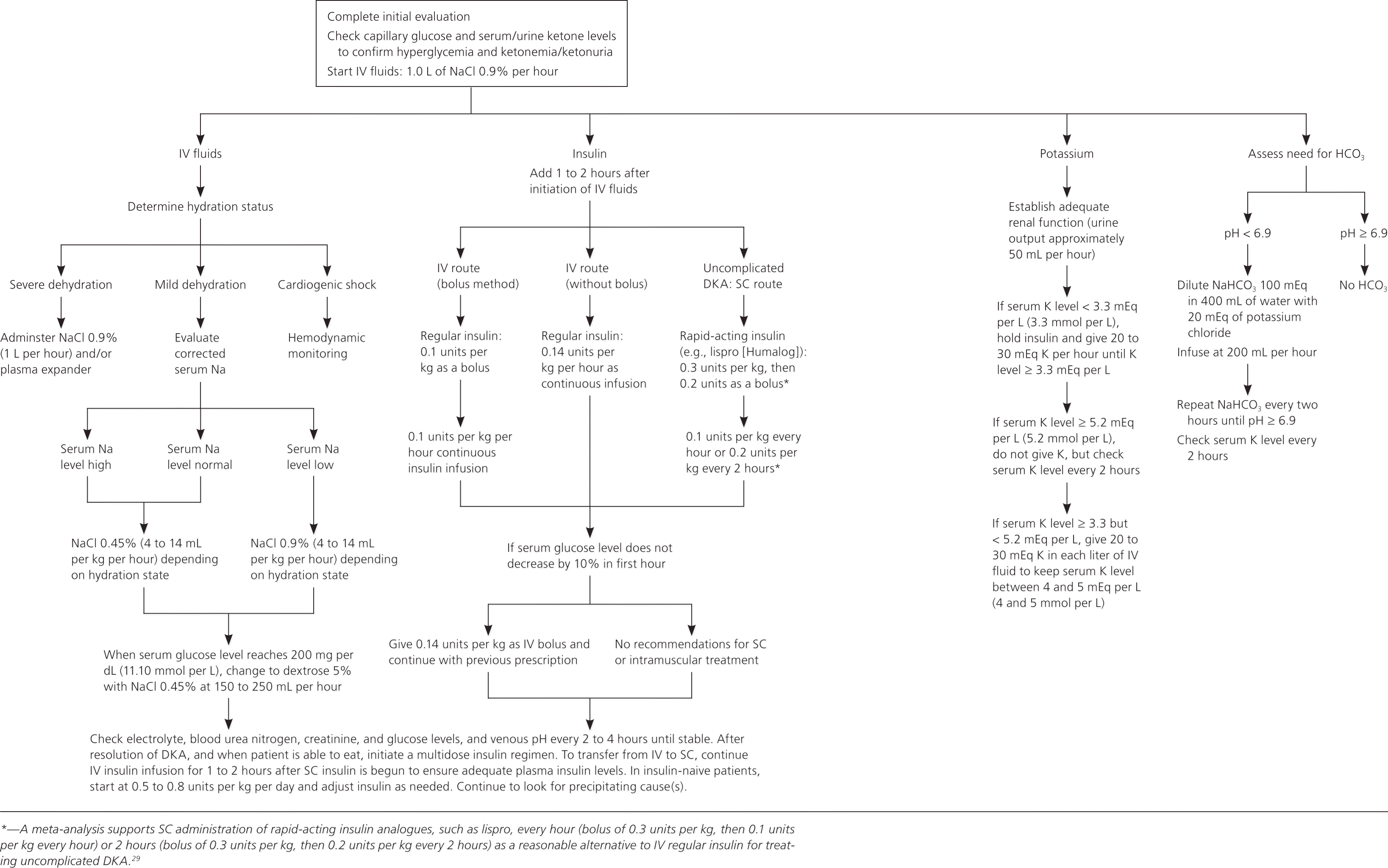
Adapted with permission from Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN. Hyperglycemic crisis in adult patients with diabetes. Diabetes Care. 2009;32(7):1339. Copyright 2009 American Diabetes Association. Additional information from reference 29.
Figure 2. Management of Diabetic Ketoacidosis in Persons Younger than 20 Years
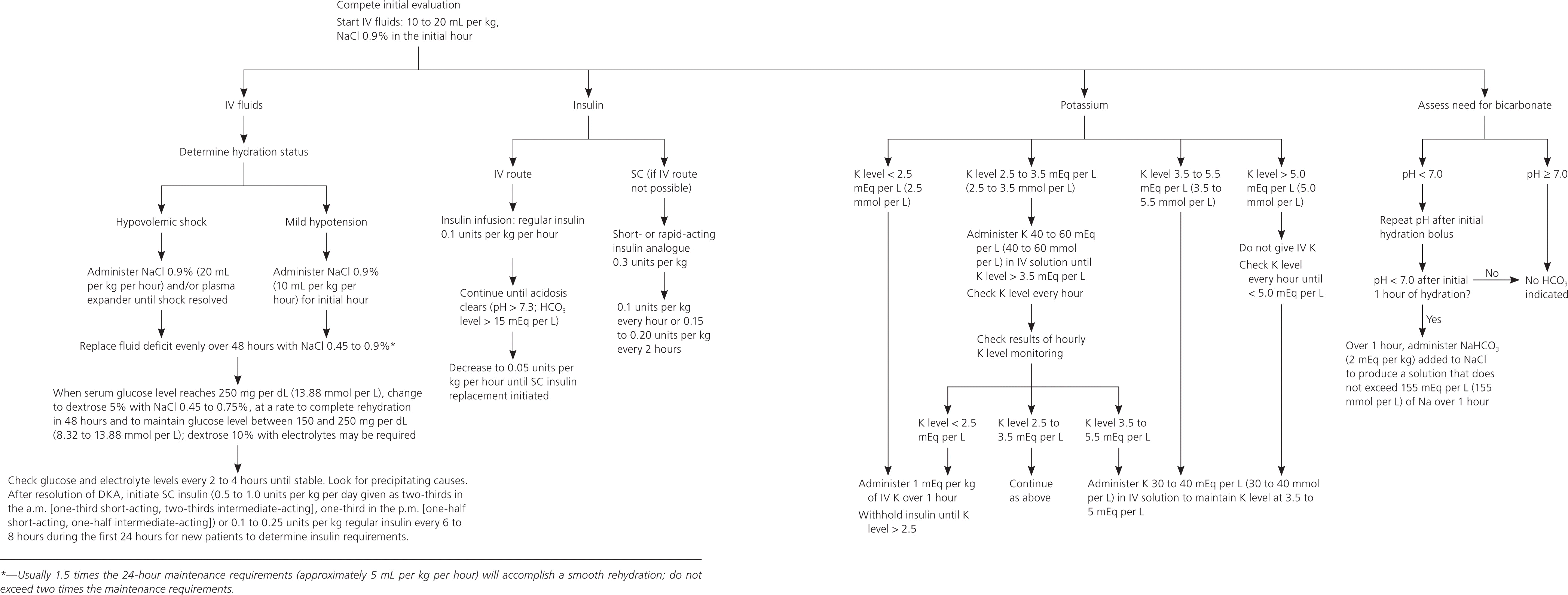
Adapted with permission from Kitabchi AE, Umpierrez GE, Miles JM, et al.; American Diabetes Association. Hyperglycemic crisis in diabetes. Diabetes Care. 2004;27(suppl 1):S98. Copyright 2004 American Diabetes Association. Additional information from reference 30.
FLUID REPLACEMENT
After determining the level of dehydration, intravenous fluid replacement should be started. In most persons, saline 0.9% is started at 15 to 20 mL per kg per hour, or 1 L per hour initially. Fluid status, cardiac status, urine output, blood pressure, and electrolyte level should be monitored. As the patient stabilizes, fluids can be lowered to 4 to 14 mL per kg per hour, or 250 to 500 mL per hour. Once the corrected sodium concentration is normal or high (greater than 135 mEq per L [135 mmol per L]), the solution can be changed to saline 0.45%. Dextrose is added when the glucose level decreases to 200 mg per dL (11.10 mmol per L).4
INSULIN
To further correct hyperglycemia, insulin should be added to intravenous fluids one to two hours after fluids are initiated. An initial bolus of 0.1 units per kg should be given with an infusion of 0.1 units per kg per hour.4 Some believe this bolus is unnecessary as long as an adequate infusion of insulin is maintained.31 An infusion of 0.14 units per kg per hour is recommended in the absence of a bolus. Glucose level should decrease by about 50 to 70 mg per dL (2.77 to 3.89 mmol per L) per hour, and the insulin infusion should be adjusted to achieve this goal.4 Once the glucose level decreases to 200 mg per dL, the insulin infusion rate should be decreased to 0.05 to 0.1 units per kg per hour, and dextrose should be added to the intravenous fluids to maintain a glucose level between 150 and 200 mg per dL (8.32 and 11.10 mmol per L).4 Subcutaneous insulin is an effective alternative to intravenous insulin in persons with uncomplicated DKA.29 In one prospective randomized trial of 45 persons, 15 received insulin aspart (Novolog) hourly, 15 received insulin aspart every two hours, and 15 received standard intravenous infusion of regular insulin. Physiologic and clinical outcomes were identical in all three groups.32 A meta-analysis supports subcutaneous administration of rapid-acting insulin analogues, such as lispro (Humalog), every hour (bolus of 0.3 units per kg, then 0.1 units per kg) or two hours (bolus of 0.3 units per kg, then 0.2 units per kg) as a reasonable alternative to intravenous regular insulin for treating uncomplicated DKA.29
DKA is resolved when the glucose level is less than 200 mg per dL, the pH is greater than 7.3, and the bicarbonate level is 18 mEq per L or higher.4
Once these levels are achieved and oral fluids are tolerated, the patient can be started on an insulin regimen that includes an intermediate- or long-acting insulin and a short- or rapid-acting insulin. When intravenous insulin is used, it should remain in place for one to two hours after subcutaneous insulin is initiated. Persons known to have diabetes can be started on their outpatient dose, with adjustments to improve control. Those new to insulin should receive 0.5 to 0.8 mg per kg per day in divided doses.4
POTASSIUM
Although potassium is profoundly depleted in persons with DKA, decreased insulin levels, acidosis, and volume depletion cause elevated extracellular concentrations. Potassium levels should be monitored every two to four hours in the early stages of DKA. Hydration alone will cause potassium to drop because of dilution. Improved renal perfusion will increase excretion. Insulin therapy and correction of acidosis will cause cellular uptake of potassium. If the potassium level is in the normal range, replacement can start at 10 to 15 mEq potassium per hour. During treatment of DKA, the goal is to maintain serum potassium levels between 4 and 5 mEq per L (4 and 5 mmol per L). If the potassium level is between 3.3 and 5.2 mEq per L (3.3 and 5.2 mmol per L) and urine output is normal, replacement can start at 20 to 30 mEq potassium per hour. If the potassium level is lower than 3.3 mEq per L, insulin should be held and replacement should be started at 20 to 30 mEq potassium per hour. If the potassium level is greater than 5.2 mEq per L, insulin therapy without potassium replacement should be initiated, and serum potassium levels should be checked every two hours. When the potassium level is between 3.3 and 5.2 mEq per L, potassium replacement should be initiated.4 Some guidelines recommend potassium replacement with potassium chloride, whereas others recommend combining it with potassium phosphate or potassium acetate. Clinical trials are lacking to determine which is best, although in the face of phosphate depletion, potassium phosphate is used.
BICARBONATE
Bicarbonate therapy in persons with DKA is somewhat controversial. Proponents believe that severe acidosis will cause cardiac and neurologic complications. However, studies have not demonstrated improved clinical outcomes with bicarbonate therapy, and treatment has been associated with hypokalemia. In one retrospective quasi-experimental study of 39 persons with DKA and a pH between 6.9 and 7.1, there was no difference in outcomes between those who received bicarbonate therapy and those who did not.33 A second study of 106 adolescents with DKA showed no difference in outcomes in patients treated with and without sodium bicarbonate, but few had a pH below 7 and only one had a pH below 6.9.34
Current American Diabetes Association guidelines continue to recommend bicarbonate replacement in persons with a pH lower than 6.9 using 100 mEq of sodium bicarbonate in 400 mL of sterile water with 20 mEq of potassium chloride at a rate of 200 mL per hour for two hours. This should be repeated every two hours until the patient's pH is 6.9 or greater.4
PHOSPHATE AND MAGNESIUM
Phosphate levels may be normal to elevated on presentation, but decline with treatment as the phosphate enters the intracellular space. Studies have not shown a benefit from phosphate replacement, and it can be associated with hypocalcemia and hypomagnesemia. However, because phosphate deficiency is linked with muscle fatigue, rhabdomyolysis, hemolysis, respiratory failure, and cardiac arrhythmia, replacement is recommended when the phosphate level falls below 1.0 mg per dL (0.32 mmol per L) or when these complications occur.4 Persons with anemia or respiratory problems and congestive heart failure may benefit from phosphate. This can be achieved by adding 20 to 30 mEq of potassium phosphate to the intravenous fluid.4
DKA can cause a drop in magnesium, which can result in paresthesia, tremor, muscle spasm, seizures, and cardiac arrhythmia. It should be replaced if it falls below 1.2 mg per dL or if symptoms of hypomagnesemia develop.35
Complications
Cerebral edema is the most severe complication of DKA. It occurs in 0.5 to 1 percent of all DKA cases,36,37 and carries a mortality rate of 21 to 24 percent.30 Survivors are at risk of residual neurologic problems.38 Cerebral edema predominantly occurs in children, although it has been reported in adults.39 Risk factors include younger age, new-onset diabetes, longer duration of symptoms, lower partial pressure of carbon dioxide, severe acidosis, low initial bicarbonate level, low sodium level, high glucose level at presentation, rapid hydration, and retained fluid in the stomach.30,40 Signs of cerebral edema that require immediate evaluation include headache, persistent vomiting, hypertension, bradycardia, and lethargy and other neurologic changes.
Other complications of DKA include hypokalemia, hypoglycemia, acute renal failure, and shock. Less common problems can include rhabdomyolysis,41 thrombosis and stroke,42 pneumomediastinum,43 prolonged corrected QT interval,44 pulmonary edema,45 and memory loss with decreased cognitive function in children.46
Prevention
Physicians should recognize signs of diabetes in all age groups, and should educate patients and caregivers on how to recognize them as well (eTable A). In one study, persons with DKA had symptoms of diabetes for 24.5 days before developing DKA.17 Persons with diabetes and their caregivers should be familiar with adjusting insulin during times of illness. This includes more frequent glucose monitoring; continuing insulin, but at lower doses, during times of decreased food intake; and checking urine ketone levels with a dipstick test if the glucose level is greater than 240 mg per dL (13.32 mmol per L).47 More accessible home measurement of serum ketones with a commercial glucometer may allow for earlier detection of DKA and decreased hospital visits.48 Persons with an insulin pump need to know their pump settings, and should maintain a prescription for basal insulin in case of pump failure.
eTable A. Preventive Strategies for Diabetic Ketoacidosis

| Education for physicians on early recognition of diabetes mellitus symptoms for prompt diagnosisA1 | |
| Education for patients and caregivers on diabetes care | |
| 24-hour hotline for urgent questions | |
| Group visitsA2 | |
| Referral for diabetes education with certified educator or pharmacistA3, A4 | |
| TelecommunicationA5 | |
| Web-based educationA6 (http://dtc.ucsf.edu and http://www.learningaboutdiabetes.org) | |
| Sick day managementA7 | |
| Early contact with clinician | |
| Insulin reduction rather than elimination | |
| Measurement of urine or serum ketone level | |
| Backup insulin protocol in case of insulin pump failure | |
| Psychological counseling for those who eliminate insulin for body image concerns, and those who have major depression or other psychological illnesses that interfere with proper management | |
| Disparities in care | |
| Assess reasons for discontinuation of insulin (e.g., access to health care; social, cultural, economic barriers) | |
| Referral to community resources | |
| Copay reduction for medicationA8 | |
Information from:
A1 . Vanelli M, Chiari G, Ghizzoni L, Costi G, Giacalone T, Chiarelli F. Effectiveness of a prevention program for diabetic ketoacidosis in children. An 8-year study in schools and private practices. Diabetes Care. 1999;22(1):7–9.
A2 . Riley SB, Marshall ES. Group visits in diabetes care: a systematic review. Diabetes Educ. 2010;36(6):936–944.
A3. Funnell MM, Brown TL, Childs BP, et al. National standards for diabetes self-management education. Diabetes Care. 2010;33(suppl 1):S89–S96.
A4. Taveira TH, Friedmann PD, Cohen LB, et al. Pharmacist-led group medical appointment model in type 2 diabetes. Diabetes Educ. 2010;36(1):109–117.
A5 . Mayes PA, Silvers A, Prendergast JJ. New direction for enhancing quality in diabetes care: utilizing telecommunications and paraprofessional outreach workers backed by an expert medical team. Telemed J E Health. 2010;16(3):358–363.
A6. Hall DL, Drab SR, Campbell RK, Meyer SM, Smith RB. A Web-based interprofessional diabetes education course. Am J Pharm Educ. 2007;71(5):93.
A7. Brink S, Laffel L, Likitmaskul S, et al. Sick day management in children and adolescents with diabetes. Pediatr Diabetes. 2009;10(suppl 12):146–153.
A8. Nair KV, Miller K, Park J, Allen RR, Saseen JJ, Biddle V. Prescription co-pay reduction program for diabetic employees. Popul Health Manag. 2010;13(5):235–245.
Nonadherence to medical regimens is often the cause of recurrent DKA. Physicians need to recognize patient barriers to getting care, such as financial, social, psychological, and cultural reasons. Diabetes education with certified educators and pharmacists enhances patient care.49,50 Other prevention techniques include group visits, telecommunication, web-based learning, and copay reduction for diabetes medications; however, evidence for their effectiveness is mixed.51–55
Data Sources: In July 2010, an initially broad search of PubMed, Essential Evidence Plus, and sources such as the Cochrane database and Clinical Evidence was conducted using the key term diabetic ketoacidosis. In the fall of 2010, another search was conducted using additional key terms, such as incidence and prevalence. As information was collected, individual questions were then searched to add finer points to the documentation. The searches were repeated with each draft of the manuscript.


