
Am Fam Physician. 2013;88(1):44-53
A more recent article on sepsis is available.
Related AFP Community Blog post: For Sepsis, Protocol-Driven Care Is Not Superior to Clinical Judgment
Author disclosure: No relevant financial affiliations.
Sepsis is a complication of severe infection characterized by a systemic inflammatory response. Mortality rates from sepsis range between 25% to 30% for severe sepsis and 40% to 70% for septic shock. The clinical presentation of sepsis is highly variable depending on the etiology. The most common sites of infection are the respiratory, genitourinary, and gastrointestinal systems, as well as the skin and soft tissue. Fever is often the first manifestation of sepsis, with pneumonia being the most common presentation leading to sepsis. Early goal-directed therapy completed within the first six hours of sepsis recognition significantly decreases in-hospital mortality. Initial management includes respiratory stabilization followed by aggressive fluid resuscitation. Vasopressor therapy is indicated when fluid resuscitation fails to restore adequate mean arterial pressure and organ perfusion. Early antibiotic therapy can improve clinical outcomes, and should be given within one hour of suspected sepsis. Blood product therapy may be required in some cases to correct coagulopathy and anemia, and to improve the central venous oxygen saturation. Insulin therapy may be required to maintain serum glucose levels less than 180 mg per dL. Initiation of low-dose corticosteroids may further improve survival in patients with septic shock that does not respond to vasopressor therapy. Timely initiation of evidence-based protocols should improve sepsis outcomes.
Sepsis is a life-threatening inflammatory disorder and the immune system's response to infection. It affects more than 750,000 persons annually, with a prevalence of three cases per 1,000 persons.1 Advances in pharmacotherapy and supportive care have improved survival rates; however, mortality rates remain between 25% to 30% for severe sepsis and 40% to 70% for septic shock. Sepsis is responsible for 20% of all in-hospital deaths each year (210,000), which equals the number of annual deaths from acute myocardial infarction.2,3
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Fluid resuscitation is the initial treatment for hypotension in patients with septic shock. | A | 9, 16, 17 |
| Vasopressor therapy should be initiated in patients with sepsis when fluid resuscitation fails to restore mean arterial pressure (greater than 65 mm Hg) or continued organ hypoperfusion. | B | 9, 23, 25 |
| Antibiotic therapy should be administered within one hour in patients with suspected sepsis. | B | 11, 23, 29, 31 |
| Sepsis care bundle protocols decrease sepsis mortality and should be implemented in medical centers. | A | 15, 23, 39, 43 |
Defining Sepsis
The American College of Chest Physicians and Society of Critical Care Medicine classify the continuum of an inflammatory response to microorganisms as systemic inflammatory response syndrome, sepsis, severe sepsis, and septic shock4,5 (Table 16 ). Systemic inflammatory response syndrome is identified when two of the four criteria (fever, tachycardia, tachypnea, and leukocytosis or leukopenia) are met. It occasionally occurs in the absence of an infection, such as in autoimmune syndromes, vasculitis, pancreatitis, burns, trauma, or surgery.6 Sepsis is the presence of an infection in association with systemic inflammatory response syndrome, although some patients with sepsis may not meet the criteria for systemic inflammatory response syndrome.

| Systemic inflammatory response syndrome | Two or more of the following: | |
| Body temperature > 38.5°C or < 35.0°C | ||
| Heart rate > 90 beats per minute | ||
| Respiratory rate > 20 breaths per minute or arterial CO2 tension < 32 mm Hg or need for mechanical ventilation | ||
| White blood cell count > 12,000/mm3 or < 4,000/mm3 or immature forms > 10% | ||
| Sepsis | Systemic inflammatory response syndrome and documented infection (culture or gram stain of blood, sputum, urine, or normally sterile body fluid positive for pathogenic microorganism; or focus of infection identified by visual inspection—e.g., ruptured bowel with free air or bowel contents found in abdomen at surgery, wound with purulent discharge) | |
| Severe sepsis | Sepsis and at least one sign of organ hypoperfusion or organ dysfunction: | |
| Areas of mottled skin | ||
| Capillary refilling time ≥ 3 s | ||
| Urinary output < 0.5 mL/kg for at least 1 h or renal replacement therapy | ||
| Lactates > 2 mmol/L | ||
| Abrupt change in mental status or abnormal electroencephalogram | ||
| Platelet counts < 100,000/mL or disseminated intravascular coagulation | ||
| Acute lung injury—acute respiratory distress syndrome | ||
| Cardiac dysfunction (echocardiography) | ||
| Septic shock | Severe sepsis and one of: | |
| Systemic mean blood pressure of < 60 mm Hg (< 80 mm Hg if previous hypertension) after 20–30 mL/kg starch or 40–60 mL/kg serum saline, or pulmonary capillary wedge pressure between 12 and 20 mm Hg | ||
| Need for dopamine > 5 μg/kg per min or norepinephrine or epinephrine < 0.25 μg/kg per min to maintain mean blood pressure above 60 mm Hg (> 80 mm Hg if previous hypertension) | ||
| Refractory septic shock | Need for dopamine > 15 μg/kg per min or norepinephrine or epinephrine > 0.25 μg/kg per min to maintain mean blood pressure above 60 mm Hg (> 80 mm Hg if previous hypertension) | |
Differential Diagnosis
Syndromes that mimic sepsis include hypovolemia, acute blood loss, acute pulmonary embolus, acute myocardial infarction, acute pancreatitis, transfusion reaction, diabetic ketoacidosis, and adrenal insufficiency.7
Etiology
The pathophysiology of sepsis involves a complex interaction of proinflammatory and anti-inflammatory mediators in response to pathogen invasion. These mechanisms lead to endothelium damage, vascular permeability, microvascular dysfunction, coagulation pathway activation, and impaired tissue oxygenation resulting in the cascade of sepsis.8
The systems most commonly infected are the respiratory, genitourinary, and gastrointestinal systems, as well as the skin and soft tissue. These sites comprise more than 80% of all cases of sepsis.6 Pneumonia is the most common presentation leading to sepsis.1,9–11 Bacterial microbes are the most common causative organisms (gram-positive bacteria account for 30% to 50% of total cases)6; however, a small number of patients may manifest with fungal, viral, or parasitic infections.6
Risk Factors
Persons at the extremes of age are at higher risk of developing sepsis. Patients older than 65 years are 13 times more likely to develop sepsis and have a twofold higher risk of death from sepsis independent of race, sex, comorbid conditions, and severity of illness.2 Additional risk factors include malnutrition, chronic illness, immunosuppression, recent surgery or hospitalization, and indwelling catheters or other devices.
Clinical Presentation
The signs and symptoms of sepsis are highly variable, and a clinical diagnosis is often made before culture results are available. Although localized signs and symptoms may be present, organ hypoperfusion or shock can manifest without knowledge of causation. To diagnose sepsis, physicians must obtain historical, clinical, and laboratory findings indicative of infection and organ dysfunction (Table 212 ). Overall, the respiratory system is the most common site of sepsis, although in patients older than 65 years, the most common site is the genitourinary tract.13
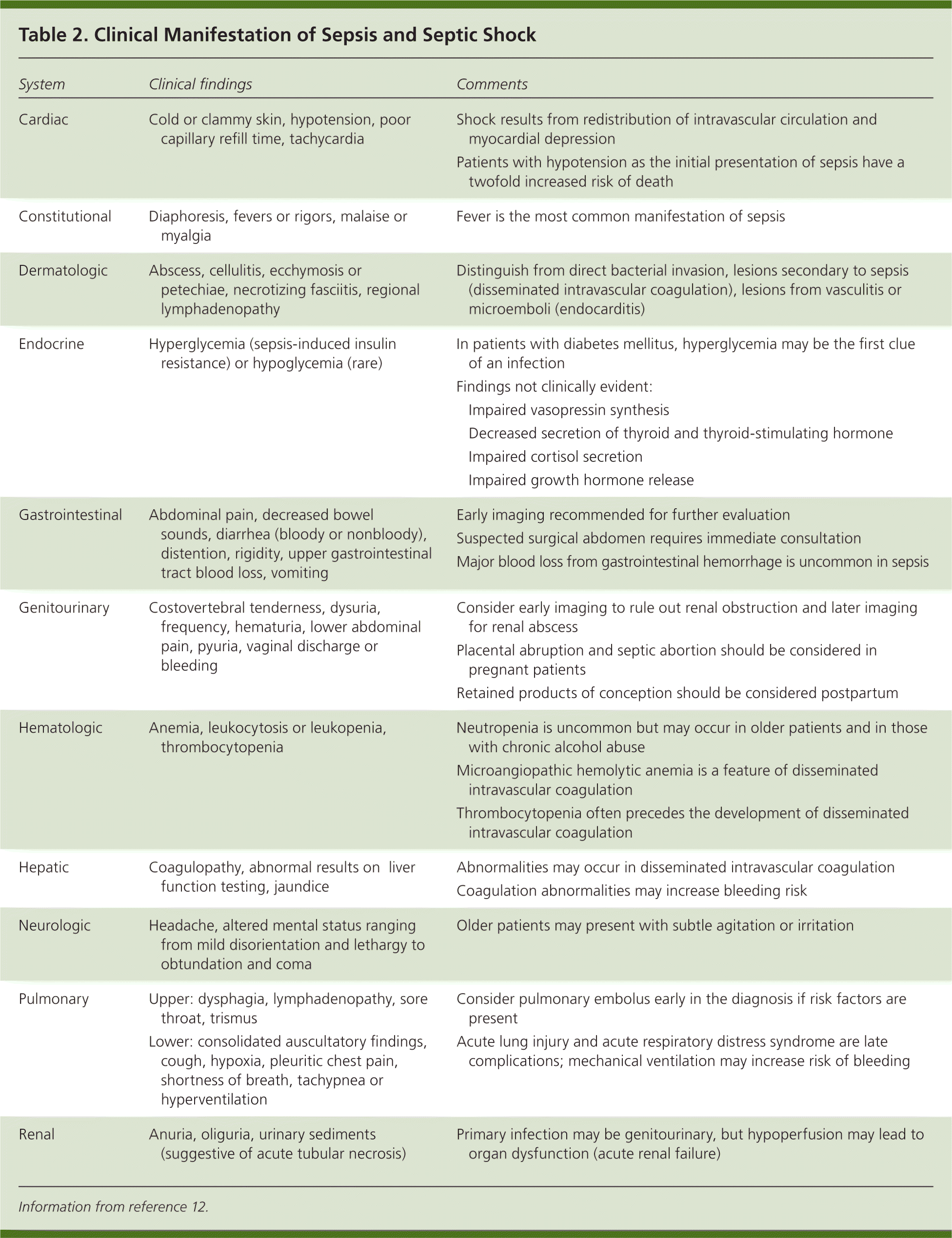
| System | Clinical findings | Comments | |
|---|---|---|---|
| Cardiac | Cold or clammy skin, hypotension, poor capillary refill time, tachycardia | Shock results from redistribution of intravascular circulation and myocardial depression | |
| Patients with hypotension as the initial presentation of sepsis have a twofold increased risk of death | |||
| Constitutional | Diaphoresis, fevers or rigors, malaise or myalgia | Fever is the most common manifestation of sepsis | |
| Dermatologic | Abscess, cellulitis, ecchymosis or petechiae, necrotizing fasciitis, regional lymphadenopathy | Distinguish from direct bacterial invasion, lesions secondary to sepsis (disseminated intravascular coagulation), lesions from vasculitis or microemboli (endocarditis) | |
| Endocrine | Hyperglycemia (sepsis-induced insulin resistance) or hypoglycemia (rare) | In patients with diabetes mellitus, hyperglycemia may be the first clue of an infection | |
| Findings not clinically evident: | |||
| Impaired vasopressin synthesis | |||
| Decreased secretion of thyroid and thyroid-stimulating hormone | |||
| Impaired cortisol secretion | |||
| Impaired growth hormone release | |||
| Gastrointestinal | Abdominal pain, decreased bowel sounds, diarrhea (bloody or nonbloody), distention, rigidity, upper gastrointestinal tract blood loss, vomiting | Early imaging recommended for further evaluation | |
| Suspected surgical abdomen requires immediate consultation | |||
| Major blood loss from gastrointestinal hemorrhage is uncommon in sepsis | |||
| Genitourinary | Costovertebral tenderness, dysuria, frequency, hematuria, lower abdominal pain, pyuria, vaginal discharge or bleeding | Consider early imaging to rule out renal obstruction and later imaging for renal abscess | |
| Placental abruption and septic abortion should be considered in pregnant patients | |||
| Retained products of conception should be considered postpartum | |||
| Hematologic | Anemia, leukocytosis or leukopenia, thrombocytopenia | Neutropenia is uncommon but may occur in older patients and in those with chronic alcohol abuse | |
| Microangiopathic hemolytic anemia is a feature of disseminated intravascular coagulation | |||
| Thrombocytopenia often precedes the development of disseminated intravascular coagulation | |||
| Hepatic | Coagulopathy, abnormal results on liver function testing, jaundice | Abnormalities may occur in disseminated intravascular coagulation | |
| Coagulation abnormalities may increase bleeding risk | |||
| Neurologic | Headache, altered mental status ranging from mild disorientation and lethargy to obtundation and coma | Older patients may present with subtle agitation or irritation | |
| Pulmonary | Upper: dysphagia, lymphadenopathy, sore throat, trismus | Consider pulmonary embolus early in the diagnosis if risk factors are present | |
| Lower: consolidated auscultatory findings, cough, hypoxia, pleuritic chest pain, shortness of breath, tachypnea or hyperventilation | Acute lung injury and acute respiratory distress syndrome are late complications; mechanical ventilation may increase risk of bleeding | ||
| Renal | Anuria, oliguria, urinary sediments (suggestive of acute tubular necrosis) | Primary infection may be genitourinary, but hypoperfusion may lead to organ dysfunction (acute renal failure) | |
The patient history should focus on symptoms, recent surgery, underlying disease, history of recent antibiotic use, and travel. Key components of the physical examination include vital signs (e.g., fever, hypoxia, hypotension), skin (e.g., furuncles or carbuncles, ulcers, cellulitis, intravenous sites, ecchymosis or petechiae), pulmonary system (e.g., tachypnea, hyperventilation, cough, hemoptysis, rhonchi or rales, egophony), gastrointestinal system (e.g., abdominal pain or guarding, decreased bowel sounds, diarrhea), genitourinary system (e.g., suprapubic tenderness, costovertebral tenderness), cardiovascular system (e.g., tachycardia, murmur), and neurologic system (e.g., altered mental status).
Fever is the most common manifestation of sepsis. The absence of fever and sepsis-induced hypothermia is more likely in patients at the extremes of age, debilitated patients, patients with chronic alcohol abuse, and patients with uremia.14 Hypotension is the presenting abnormality in approximately 40% of patients with sepsis.4 Withdrawal and agitation can be subtle and may be the sole presentations of sepsis in older persons. Immunocompromised patients and those with neutropenia often lack an obvious source of infection.
Early Management
Early management of sepsis requires respiratory stabilization. Supplemental oxygen should be given to all patients. Mechanical ventilation is recommended when supplemental oxygen fails to improve oxygenation, when respiratory failure is imminent, or when the airway cannot be protected. Perfusion is assessed after respiratory stabilization. Hypotension signifies inadequate tissue perfusion. Clinical signs of hypoperfusion include cold or clammy skin, altered mental status, oliguria or anuria, and lactic acidosis.
After initial respiratory stabilization, treatment consists of fluid resuscitation, vasopressor therapy, infection identification and control, prompt antibiotic administration, and the removal or drainage of the infection source (Figure 115 ). Early general surgery consultation should be obtained for suspected acute abdomen and necrotizing infections.
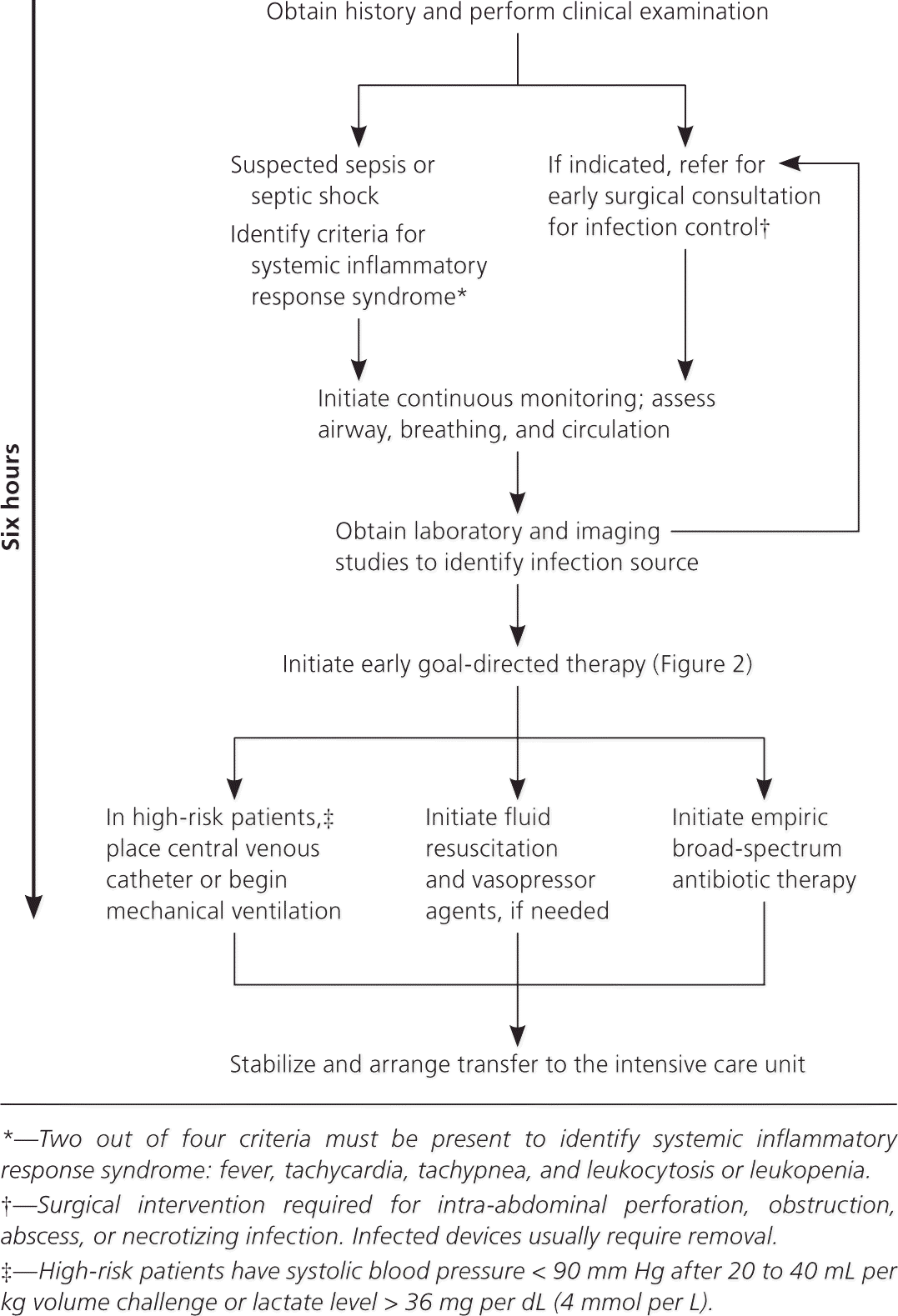
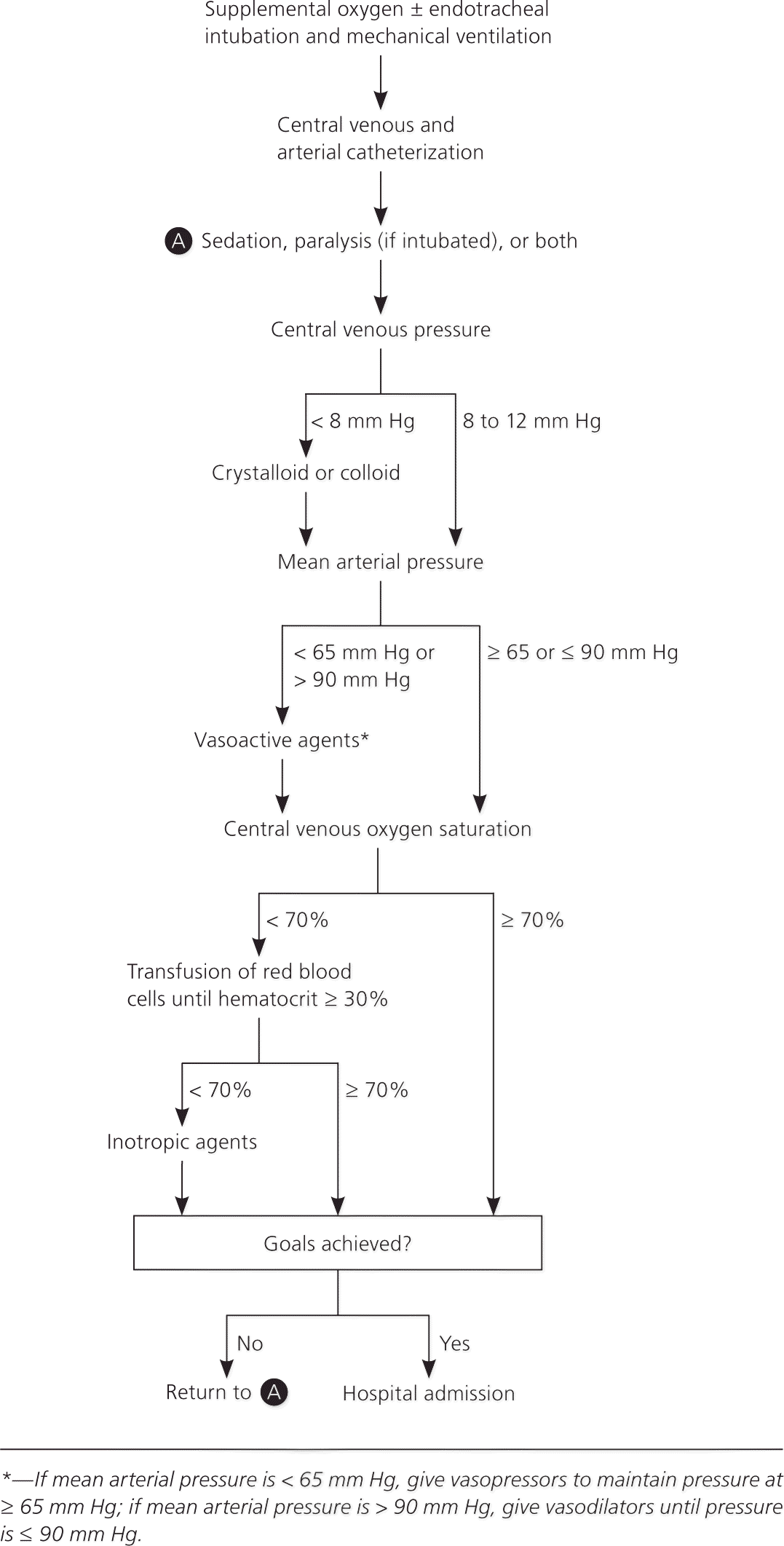
Early goal-directed therapy is a strategy implemented within six hours of suspected sepsis or septic shock. A prospective randomized controlled trial compared early goal-directed therapy with standard therapy in 263 emergency department patients with sepsis.16 Goals for both treatment arms were similar: placement of central venous catheter to maintain central venous pressure between 8 and 12 mm Hg, mean arterial blood pressure of at least 65 mm Hg, and adequate urine output. The early goal-directed therapy protocol diverged from standard management by increasing oxygen content (blood transfusions) and cardiac contractility (inotrope use) to achieve a central venous oxygen saturation greater than 70%. Patients in the early goal-directed therapy group received more intravenous fluid (5 vs. 3.5 L), inotropic support (13.7% vs. 0.8%), and packed red blood cell transfusions (64.1% vs. 18.5%) within six hours compared with the standard group. Study results demonstrated that early goal-directed therapy decreased in-hospital mortality compared with the standard therapy (30% vs. 46%). Early intervention to normalize central venous oxygen saturation and improve lactate levels increased survival rates in patients with severe sepsis (Figure 216 ).
A meta-analysis on quantitative resuscitation targeting defined hemodynamic end points showed that early resuscitation significantly decreased the mortality rate compared with late resuscitation (39% vs. 64%).17
LABORATORY AND RADIOGRAPHIC STUDIES
Recommended tests include a complete blood count with differential, basic metabolic panel, measurement of lactate and liver enzyme levels, coagulation studies, and urinalysis. Suspected respiratory infections should be evaluated using chest radiography and arterial blood gas testing to assess hypoxemia and acid-base abnormalities. If disseminated intravascular coagulation is suspected, fibrin degradation products, d-dimer levels, and fibrinogen levels should be measured. Several biologic markers of sepsis such as C-reactive protein, procalcitonin, activated partial thromboplastin time, and interleukin-6 may have diagnostic and prognostic implications, although lack of standardized assays limits their practical use. Blood cultures (two peripheral and from each indwelling catheter), urine culture, stool culture (for diarrhea or recent antibiotic use), sputum culture, and skin and soft tissue culture should be obtained. However, blood culture results are negative in 50% to 65% of patients with sepsis.18 Cerebrospinal, joint, pleural, and peritoneal fluid should be evaluated as clinically indicated.
Echocardiography is recommended to diagnose endocarditis, and should be performed in patients with a heart murmur or suspected intravenous drug use. Evaluation for pulmonary embolus requires chest computed tomography (CT) or ventilation-perfusion scanning, and suspected abdominal or pelvic infection requires abdominal and pelvic CT. Evaluation of renal abscess or complicated pyelonephritis often requires renal ultrasonography or CT. Head CT should be considered for patients presenting with altered mental status to rule out intracranial hemorrhage, abscess, or malignancy.
INITIAL FLUID RESUSCITATION
The highest priority in early sepsis is establishing vascular access and initiating fluid resuscitation. Hypovolemia, myocardial depression, and hypoperfusion (termed “distributive shock”) result in hypotension, which is the most important event leading to increased morbidity and mortality in patients with sepsis. Intravenous fluid should be initiated immediately when sepsis is recognized.9,16,17 Delaying fluid resuscitation can worsen tissue hypoxia, leading to multiple organ dysfunction. Isotonic crystalloid is the most common resuscitative fluid used; however, the use of colloid fluid showed no differences in pulmonary edema, mortality, or length of hospitalization compared with isotonic crystalloid.19,20
Dosing intravenous fluid is often empiric; however, most physicians use central venous pressure measurements to guide initial fluid therapy. Beyond the first 12 hours of sepsis, central venous pressures poorly predict hemodynamic response to fluid challenges.21 Thereafter, the use of cardiac output monitoring systems to assess changes in arterial waveform is more accurate in predicting fluid responsiveness.22 The initial resuscitative goal is to optimize intravascular volume to achieve a central venous pressure of 8 to 12 mm Hg, and 12 to 15 mm Hg when patients are mechanically ventilated.16,23 Fluid challenge therapy should begin with a bolus of 20 mL per kg over 30 minutes or less. The degree of hypovolemia will guide further challenges and should be in increments of 500 mL. The average fluid resuscitation volume in two recognized sepsis trials was 5 L in six hours and 6.3 L in 12 hours.16,24 Response to fluid resuscitation and continued rates of administration beyond the first 12 hours should be assessed by blood pressure response, tissue perfusion, and urine output. Optimal survival occurs with a positive fluid balance (i.e., intravenous fluid input minus urine output) of 3 to 4 L at 12 hours.24
VASOPRESSOR THERAPY
Vasopressor support is recommended when intravenous fluid administration fails to restore adequate mean arterial pressure and organ perfusion. The mean arterial pressure should be maintained at greater than 65 mm Hg. Dopamine and norepinephrine are the first-line vasopressor agents recommended in the treatment of septic shock.9,23,25 A randomized controlled trial (n = 1,679) compared dopamine with norepinephrine and demonstrated no clinically significant difference in 28-day mortality between the groups, although arrhythmia events were more common in the dopamine group.26 Most experts favor using norepinephrine over dopamine in persons with septic shock. The addition of low-dose vasopressin (Pitressin) to norepinephrine may be synergistic but does not improve 28-day mortality rates in persons with severe sepsis compared with norepinephrine alone.27
The use of low-dose dopamine to maintain renal function or treat renal dysfunction in clinical practice is not recommended. A meta-analysis failed to show benefit in mortality reduction, prevention of acute renal failure, or the need for dialysis in critically ill patients.28
ANTIBIOTIC THERAPY
Early appropriate antibiotic therapy is associated with improved clinical outcomes. Consensus guidelines recommend antibiotic therapy within one hour of suspected sepsis.11,23 In septic shock, the initiation of antibiotic therapy within one hour increases survival; with each hour antibiotic therapy is delayed, survival decreases by about 8%.29
Empiric antibiotic therapy should be based on the most likely source, clinical context (community- vs. hospital-acquired sepsis), recent antibiotic use, and local resistance patterns (Table 330 ). Empiric antibiotic therapy should be narrowed or redirected when the causative organism has been identified, thereby reducing the risk of resistance or superinfection.
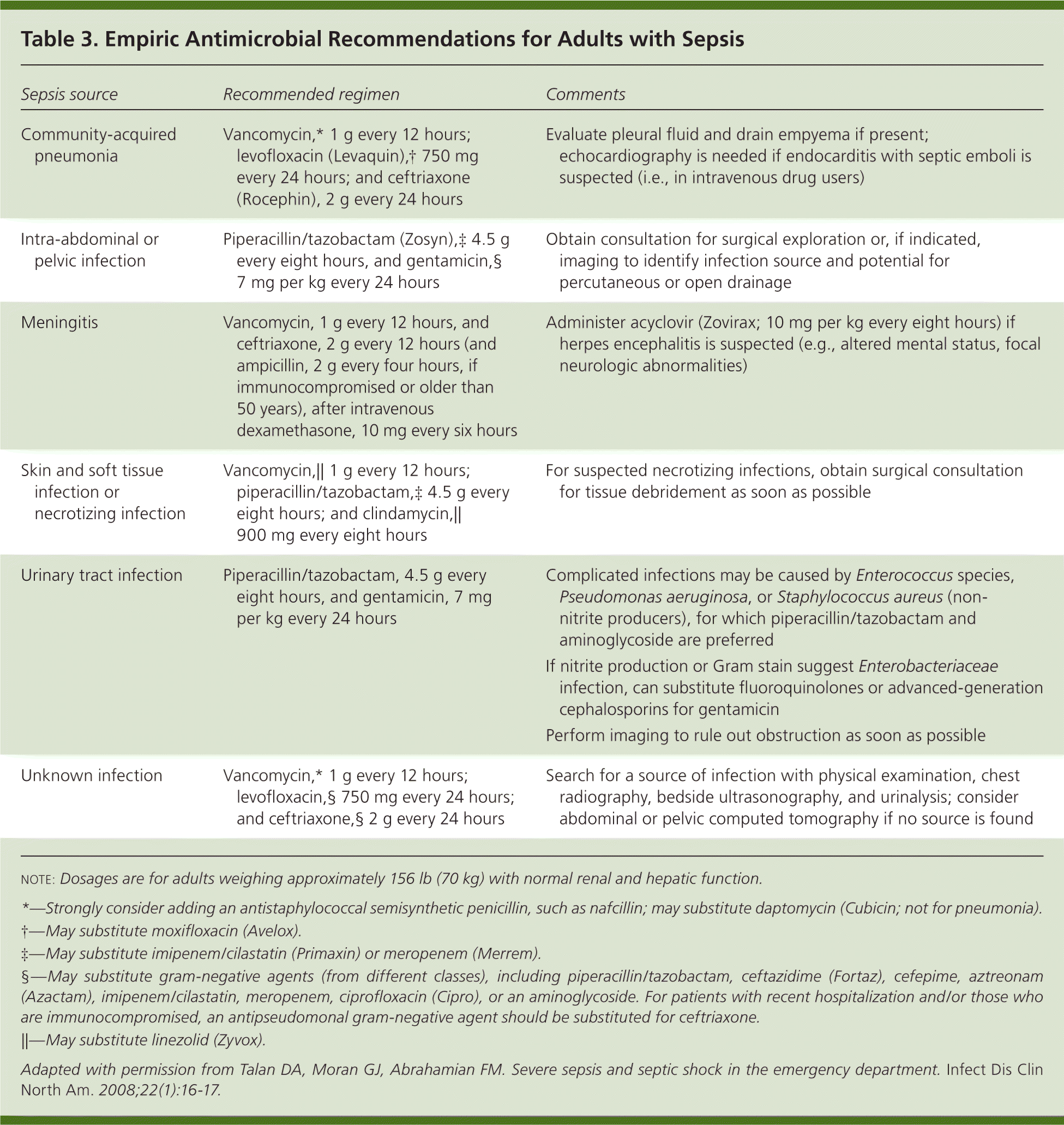
| Sepsis source | Recommended regimen | Comments |
|---|---|---|
| Community-acquired pneumonia | Vancomycin,* 1 g every 12 hours; levofloxacin (Levaquin),† 750 mg every 24 hours; and ceftriaxone (Rocephin), 2 g every 24 hours | Evaluate pleural fluid and drain empyema if present; echocardiography is needed if endocarditis with septic emboli is suspected (i.e., in intravenous drug users) |
| Intra-abdominal or pelvic infection | Piperacillin/tazobactam (Zosyn),‡ 4.5 g every eight hours, and gentamicin,§ 7 mg per kg every 24 hours | Obtain consultation for surgical exploration or, if indicated, imaging to identify infection source and potential for percutaneous or open drainage |
| Meningitis | Vancomycin, 1 g every 12 hours, and ceftriaxone, 2 g every 12 hours (and ampicillin, 2 g every four hours, if immunocompromised or older than 50 years), after intravenous dexamethasone, 10 mg every six hours | Administer acyclovir (Zovirax; 10 mg per kg every eight hours) if herpes encephalitis is suspected (e.g., altered mental status, focal neurologic abnormalities) |
| Skin and soft tissue infection or necrotizing infection | Vancomycin,|| 1 g every 12 hours; piperacillin/tazobactam,‡ 4.5 g every eight hours; and clindamycin,|| 900 mg every eight hours | For suspected necrotizing infections, obtain surgical consultation for tissue debridement as soon as possible |
| Urinary tract infection | Piperacillin/tazobactam, 4.5 g every eight hours, and gentamicin, 7 mg per kg every 24 hours | Complicated infections may be caused by Enterococcus species, Pseudomonas aeruginosa, or Staphylococcus aureus (non-nitrite producers), for which piperacillin/tazobactam and aminoglycoside are preferred |
| If nitrite production or Gram stain suggest Enterobacteriaceae infection, can substitute fluoroquinolones or advanced-generation cephalosporins for gentamicin | ||
| Perform imaging to rule out obstruction as soon as possible | ||
| Unknown infection | Vancomycin,* 1 g every 12 hours; levofloxacin,§ 750 mg every 24 hours; and ceftriaxone,§ 2 g every 24 hours | Search for a source of infection with physical examination, chest radiography, bedside ultrasonography, and urinalysis; consider abdominal or pelvic computed tomography if no source is found |
A 2010 meta-analysis compared combination antibiotic therapy with monotherapy, and found that combination therapy demonstrated survival benefit and improved clinical response in patients who had septic shock.31 However, combination antibiotic therapy did not improve sepsis survival compared with monotherapy in hemodynamically stable patients.31 When there is a risk of Pseudomonas aeruginosa bacteremia, antipseudomonal combination therapy is recommended.9
Later Management
Improvement in survival remains contingent on early recognition and management of severe sepsis and septic shock. Table 4 lists additional interventions designed to improve survival.16,23,26,32–37 Therapeutic advancements have led to the development of guidelines that call for implementation of a six-hour resuscitation bundle.15,23,38 This includes initiation of central venous pressure and central venous oxygen saturation monitoring within two hours; broad-spectrum antibiotics administered within four hours, although within one hour is preferred; early goal-directed therapy completed within six hours; and monitoring of lactate clearance. Patients presenting to the emergency department with sepsis who complete the resuscitation bundle before intensive care unit transfer have lower in-hospital mortality rates compared with patients whose resuscitative bundles were not completed (21% vs. 40%).39
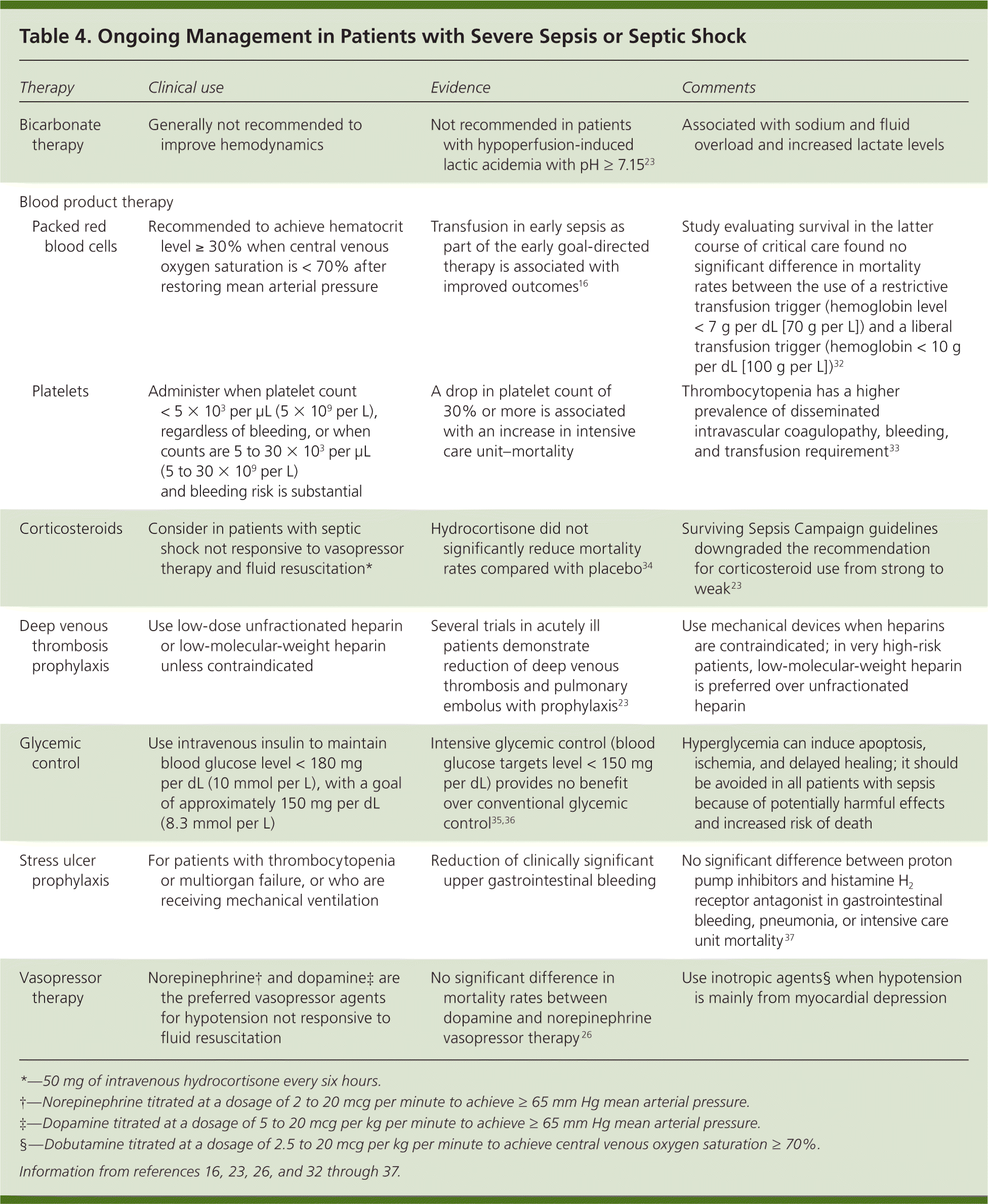
| Therapy | Clinical use | Evidence | Comments | |
|---|---|---|---|---|
| Bicarbonate therapy | Generally not recommended to improve hemodynamics | Not recommended in patients with hypoperfusion-induced lactic acidemia with pH ≥ 7.1523 | Associated with sodium and fluid overload and increased lactate levels | |
| Blood product therapy | ||||
| Packed red blood cells | Recommended to achieve hematocrit level ≥ 30% when central venous oxygen saturation is < 70% after restoring mean arterial pressure | Transfusion in early sepsis as part of the early goal-directed therapy is associated with improved outcomes16 | Study evaluating survival in the latter course of critical care found no significant difference in mortality rates between the use of a restrictive transfusion trigger (hemoglobin level < 7 g per dL [70 g per L]) and a liberal transfusion trigger (hemoglobin < 10 g per dL [100 g per L])32 | |
| Platelets | Administer when platelet count < 5 × 103 per μL (5 × 109 per L), regardless of bleeding, or when counts are 5 to 30 × 103 per μL (5 to 30 × 109 per L) and bleeding risk is substantial | A drop in platelet count of 30% or more is associated with an increase in intensive care unit–mortality | Thrombocytopenia has a higher prevalence of disseminated intravascular coagulopathy, bleeding, and transfusion requirement33 | |
| Corticosteroids | Consider in patients with septic shock not responsive to vasopressor therapy and fluid resuscitation* | Hydrocortisone did not significantly reduce mortality rates compared with placebo34 | Surviving Sepsis Campaign guidelines downgraded the recommendation for corticosteroid use from strong to weak23 | |
| Deep venous thrombosis prophylaxis | Use low-dose unfractionated heparinor low-molecular-weight heparin unless contraindicated | Several trials in acutely ill patients demonstrate reduction of deep venous thrombosis and pulmonary embolus with prophylaxis23 | Use mechanical devices when heparins are contraindicated; in very high-risk patients, low-molecular-weight heparin is preferred over unfractionated heparin | |
| Glycemic control | Use intravenous insulin to maintain blood glucose level < 180 mg per dL (10 mmol per L), with a goal of approximately 150 mg per dL (8.3 mmol per L) | Intensive glycemic control (blood glucose targets level < 150 mg per dL) provides no benefit over conventional glycemic control35,36 | Hyperglycemia can induce apoptosis, ischemia, and delayed healing; it should be avoided in all patients with sepsis because of potentially harmful effects and increased risk of death | |
| Stress ulcer prophylaxis | For patients with thrombocytopenia or multiorgan failure, or who are receiving mechanical ventilation | Reduction of clinically significant upper gastrointestinal bleeding | No significant difference between proton pump inhibitors and histamine H2 receptor antagonist in gastrointestinal bleeding, pneumonia, or intensive care unit mortality37 | |
| Vasopressor therapy | Norepinephrine† and dopamine‡ are the preferred vasopressor agents for hypotension not responsive to fluid resuscitation | No significant difference in mortality rates between dopamine and norepinephrine vasopressor therapy 26 | Use inotropic agents§ when hypotension is mainly from myocardial depression | |
In addition to early goal-directed therapy, other therapies have been reported to improve outcomes. In randomized controlled trials, reductions in mortality in patients with sepsis were achieved with low-dose corticosteroid therapy, conventional (as opposed to intensive) glycemic control (i.e., serum glucose levels less than 180 mg per dL [10 mmol per L]), and low tidal volumes in ventilated patients.16,23,34–36,40 Blood product therapy with packed red blood cells is recommended in patients with a hemoglobin level less than 7 g per dL (70 g per L) for a target level of 7 to 9 g per dL (70 to 90 g per L), although the early goal-directed therapy protocol uses a target hematocrit level of 30% or greater in patients with low central venous oxygen saturation (less than 70%).16,23 Drotrecogin alfa (Xigris), an initially promising therapy for severe sepsis and septic shock in adults, was withdrawn from the market on October 25, 2011, after failing to demonstrate survival benefit.41
Future Directions
Surveys of medical centers instituting early goal-directed therapy sepsis programs demonstrated a 45% relative risk reduction in mortality rates.42 Prospective studies show that the use of standardized hospital order sets hastens fluid resuscitation and appropriate antibiotic therapy, resulting in lower 28-day mortality rates (48% vs. 30%).43 To overcome clinician and institutional barriers to early treatment protocols, some tertiary medical centers have collaborated with small rural hospitals to provide telemedicine as an advisory support mechanism in the management of sepsis.44
Prognosis
Patients who survive sepsis, regardless of severity, have higher mortality rates after discharge. One-year mortality rates from severe sepsis after hospital discharge range from 7% to 43%. Survivors of sepsis also have an increased incidence of posttraumatic stress disorder, cognitive dysfunction, physical disability, and persistent pulmonary dysfunction.45
Data Sources: The author searched OvidSP, PrimeAnswers, Trip Database, Institute for Healthcare Improvement, Essential Evidence Plus, National Guideline Clearinghouse, Agency for Healthcare Research and Quality, Cochrane Library, ClinicalTrials.gov, UpToDate, and the Surviving Sepsis Campaign. Key words included: sepsis, severe sepsis, septic shock, Surviving Sepsis Campaign, pathophysiology, antimicrobial, early goal-directed therapy, fluid resuscitation, central venous oxygen saturation, mortality, epidemiology, outcome assessment, quality of life, long-term follow-up, systemic inflammatory response syndrome, glycemic control, drotrecogin alfa activated, corticosteroids, vasopressor agents, dopamine, norepinephrine, vasopressin, inotropic therapy, stress ulcer prophylaxis, sepsis care bundles, and acute renal failure. Search dates: August 2011 and January 2012.
