
Am Fam Physician. 2013;88(4):231-238
A more recent article on common sleep disorders in adults is available.
Related editorial: Risks Associated with Long-Term Benzodiazepine Use.
Patient Information: Handouts are available at https://familydoctor.org/familydoctor/en/diseases-conditions/insomnia.html, https://familydoctor.org/familydoctor/en/diseases-conditions/sleep-apnea.html, and https://familydoctor.org/familydoctor/en/diseases-conditions/restless-legs-syndrome.html.
Author disclosure: No relevant financial affiliations.
Sleep disorders are common and affect sleep quality and quantity, leading to increased morbidity. Patients with sleep disorders can be categorized as those who cannot sleep, those who will not sleep, those with excessive daytime sleepiness, and those with increased movements during sleep. Insomnia, defined as difficulty initiating or maintaining sleep that results in daytime impairment, is diagnosed using history findings and treated with cognitive behavior therapy, with or without sleep hypnotics. Restless legs syndrome is characterized by an urge to move the legs that worsens with rest, is relieved by movement, and often occurs in the evening or at night. Restless legs syndrome is treated based on the frequency of symptoms. Narcolepsy is characterized by excessive sleepiness, cataplexy, hypnagogic or hypnopompic hallucinations, and sleep paralysis. It is diagnosed using a sleep log or actigraphy, followed by overnight polysomnography and a multiple sleep latency test. Narcolepsy is treated with stimulants, such as modafinil; selective serotonin reuptake inhibitors; or gamma hydroxybutyric acid (sodium oxybate). Patients with snoring and witnessed apneas may have obstructive sleep apnea, which is diagnosed using overnight polysomnography. Continuous positive airway pressure is the most common and effective treatment for obstructive sleep apnea. Rapid eye movement sleep behavior disorder is characterized by increased muscle tone during rapid eye movement sleep, resulting in the patient acting out dreams with possible harmful consequences. It is diagnosed based on history and polysomnography findings, and treated with environmental safety measures and melatonin or clonazepam.
American adults average 6.9 hours of sleep each night, less than the seven to nine hours recommended by many sleep experts.1 Sleep disorders can lead to increased morbidity and higher health care costs. In controlled studies, volunteers who were restricted to four to five hours of sleep for a few days experienced worsening neurocognitive, behavioral, metabolic, and autonomic parameters.2,3 Several studies demonstrate that sleep deprivation leads to alterations in immune function with increased risk of infection, including pneumonia.4 Additionally, chronic sleep deprivation is associated with increased risk of diabetes mellitus, cardiovascular disease, cancer, and mortality.5–8
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Cognitive behavior therapy can be effectively administered by primary care physicians to treat chronic insomnia. | B | 12–14 |
| Cognitive behavior therapy is superior to pharmacotherapy in the treatment of chronic insomnia. | A | 14, 16, 20, 21 |
| Dopaminergic agonists are first-line therapy for patients with nightly, persistent symptoms of restless legs syndrome. | A | 25 |
| A sleep-wake schedule and melatonin/bright light therapy regimen are recommended for treatment of delayed sleep phase syndrome. | C | 27 |
| Continuous positive airway pressure is the most effective treatment option for obstructive sleep apnea. | C | 36 |
Patients with sleep disorders may be categorized into those who cannot sleep, those who will not sleep, those with excessive daytime sleepiness, and those with increased movements during sleep. Table 1 summarizes common sleep disorders.
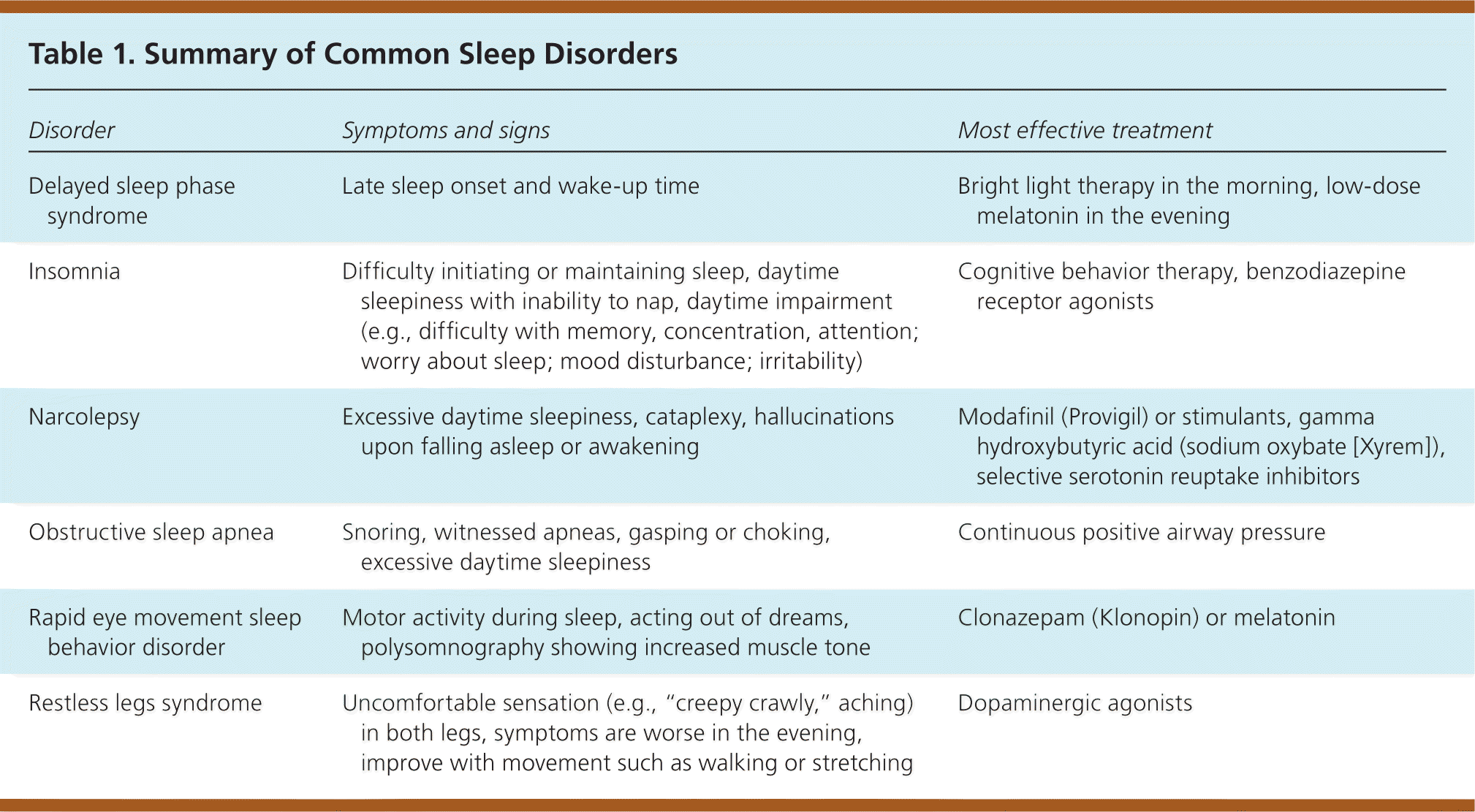
| Disorder | Symptoms and signs | Most effective treatment |
|---|---|---|
| Delayed sleep phase syndrome | Late sleep onset and wake-up time | Bright light therapy in the morning, low-dose melatonin in the evening |
| Insomnia | Difficulty initiating or maintaining sleep, daytime sleepiness with inability to nap, daytime impairment (e.g., difficulty with memory, concentration, attention; worry about sleep; mood disturbance; irritability) | Cognitive behavior therapy, benzodiazepine receptor agonists |
| Narcolepsy | Excessive daytime sleepiness, cataplexy, hallucinations upon falling asleep or awakening | Modafinil (Provigil) or stimulants, gamma hydroxybutyric acid (sodium oxybate [Xyrem]), selective serotonin reuptake inhibitors |
| Obstructive sleep apnea | Snoring, witnessed apneas, gasping or choking, excessive daytime sleepiness | Continuous positive airway pressure |
| Rapid eye movement sleep behavior disorder | Motor activity during sleep, acting out of dreams, polysomnography showing increased muscle tone | Clonazepam (Klonopin) or melatonin |
| Restless legs syndrome | Uncomfortable sensation (e.g., “creepy crawly,” aching) in both legs, symptoms are worse in the evening, improve with movement such as walking or stretching | Dopaminergic agonists |
Polysomnography
Although many sleep disorders can be diagnosed using history alone, overnight polysomnography may be useful to assess for disorders such as obstructive sleep apnea (OSA). Polysomnography monitors brain wave activity (electroencephalogram), eye movements (electro-oculogram), muscle activity (electromyogram), heart rate and rhythm (electrocardiogram), and respiration (via nasal pressure transducer and oronasal thermistor, and oxygen saturation using pulse oximetry). Table 2 lists the most common indications for polysomnography.

| Diagnosis of a sleep disorder | |
| Narcolepsy | |
| Parasomnias | |
| Periodic limb movement disorder | |
| Rapid eye movement sleep behavior disorder | |
| Sleep-related breathing disorders, such as obstructive and central sleep apnea | |
| Sleep-related seizure disorders | |
| Evaluation of sleep-related symptoms | |
| Sleep maintenance insomnia | |
| Snoring | |
| Unexplained daytime fatigue or sleepiness | |
| Treatment of sleep-related breathing disorders (i.e., using positive airway pressure titration) | |
Patients Who Cannot Sleep
INSOMNIA
Approximately 10% of the U.S. population has had insomnia that occurred every night for at least two weeks9; however, many do not discuss it with their physician. Insomnia is characterized by repeated difficulty with sleep initiation (the time it takes to fall asleep, normally less than 30 minutes; called sleep latency in sleep studies), duration (normally seven to nine hours per night for adults; also called sleep quantity), consolidation (sleep uninterrupted by arousals or awakenings), or quality that occurs despite adequate time and opportunity for sleep, resulting in daytime impairment.10 Daytime impairment may include fatigue; tiredness; difficulty with memory, concentration, and attention; worry about sleep; mood disturbances; or irritability. Insomnia is usually diagnosed with a patient history that includes evaluation for contributing psychiatric or medical conditions. Table 3 includes questions for patients that may be helpful in this assessment. Occasionally, a sleep log (eFigure A) or an actigraphy log (eFigure B) used for one to two weeks may be helpful. Overnight polysomnography is rarely needed, unless the history suggests concurrent sleep disorders or initial treatment is ineffective.11
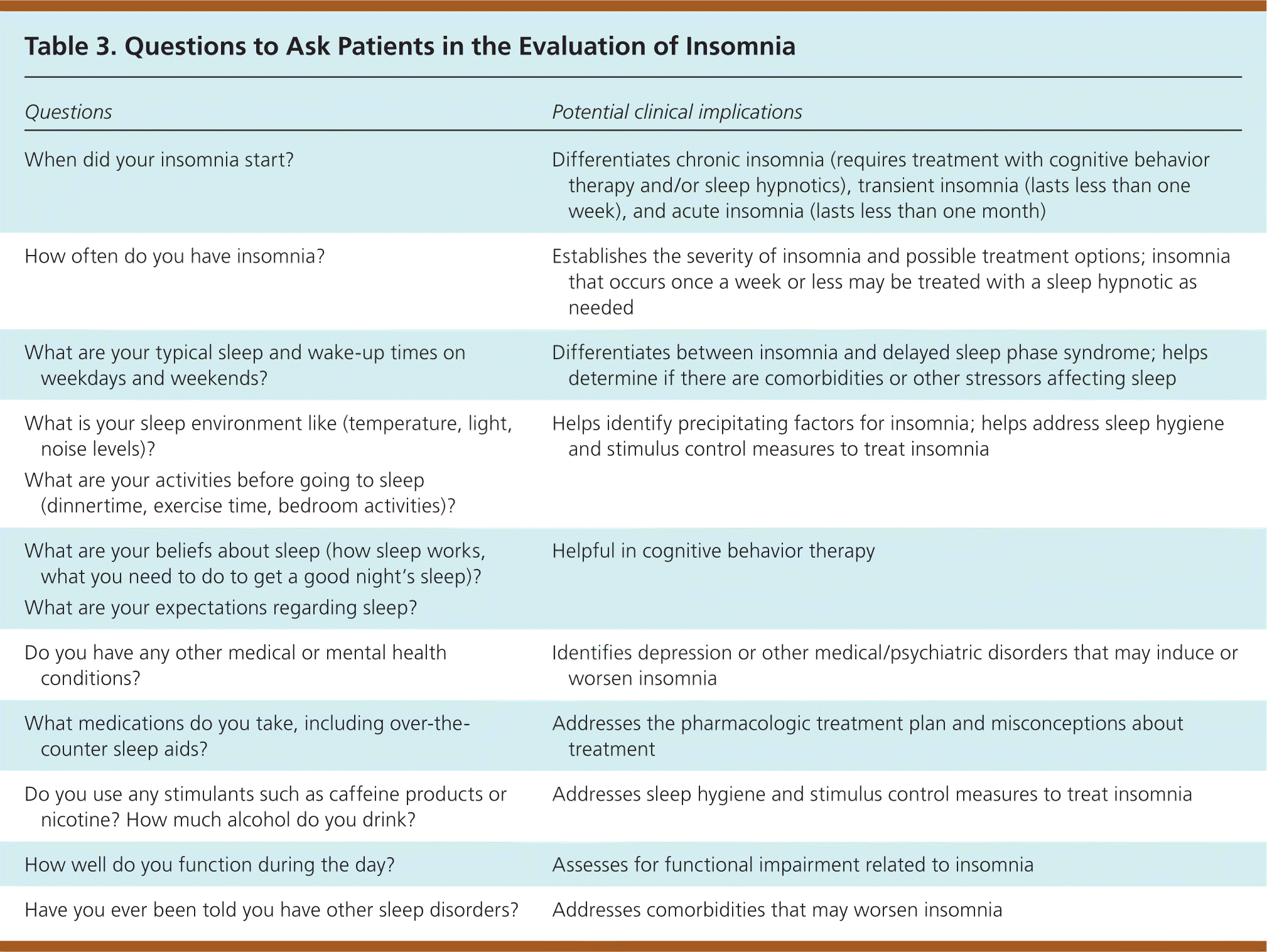
| Questions | Potential clinical implications |
|---|---|
| When did your insomnia start? | Differentiates chronic insomnia (requires treatment with cognitive behavior therapy and/or sleep hypnotics), transient insomnia (lasts less than one week), and acute insomnia (lasts less than one month) |
| How often do you have insomnia? | Establishes the severity of insomnia and possible treatment options; insomnia that occurs once a week or less may be treated with a sleep hypnotic as needed |
| What are your typical sleep and wake-up times on weekdays and weekends? | Differentiates between insomnia and delayed sleep phase syndrome; helps determine if there are comorbidities or other stressors affecting sleep |
| What is your sleep environment like (temperature, light, noise levels)? | Helps identify precipitating factors for insomnia; helps address sleep hygiene and stimulus control measures to treat insomnia |
| What are your activities before going to sleep (dinnertime, exercise time, bedroom activities)? | |
| What are your beliefs about sleep (how sleep works, what you need to do to get a good night's sleep)? | Helpful in cognitive behavior therapy |
| What are your expectations regarding sleep? | |
| Do you have any other medical or mental health conditions? | Identifies depression or other medical/psychiatric disorders that may induce or worsen insomnia |
| What medications do you take, including over-the-counter sleep aids? | Addresses the pharmacologic treatment plan and misconceptions about treatment |
| Do you use any stimulants such as caffeine products or nicotine? How much alcohol do you drink? | Addresses sleep hygiene and stimulus control measures to treat insomnia |
| How well do you function during the day? | Assesses for functional impairment related to insomnia |
| Have you ever been told you have other sleep disorders? | Addresses comorbidities that may worsen insomnia |


Cognitive behavior therapy (CBT) and hypnotic medications are treatment options for insomnia. Table 4 outlines the key points of CBT for insomnia. Primary care physicians can effectively administer CBT to treat chronic insomnia.12–14 Medication regimens approved by the U.S. Food and Drug Administration for insomnia are summarized in Table 5. Hypnotics may be associated with tolerance, dependence, and withdrawal symptoms, such as rebound insomnia, if a drug is abruptly stopped. However, studies show contradictory evidence for the development of tolerance and dependence.15 There are no comparative data on the effectiveness of short- or long-acting hypnotics for sleep onset or sleep maintenance insomnia, respectively.

| Cognitive therapy | |
| Focuses on changing false beliefs and attitudes about sleep (e.g., everyone needs at least 8 hours of sleep for good health) | |
| Sleep hygiene education | |
| No pets in the bedroom | |
| No caffeine consumption after 4 p.m. | |
| Keep bedroom cool and conducive to sleep | |
| No watching the bedroom clock | |
| No nicotine use, especially in the evening | |
| No exercising within 2 to 3 hours before bedtime | |
| Sleep restriction | |
| Time in bed can be reduced by estimating the actual total time that the patient is sleeping (e.g., if the patient is in bed for 8 hours but sleeps for 5.5 hours, time in bed could be reduced to 5.5 hours); time in bed usually should not be reduced to less than 5 hours | |
| After sleep efficiency (ratio of time sleeping to time in bed) reaches 90%, the time in bed can be increased by 15 minutes every week | |
| Stimulus control | |
| Go to bed only when sleepy | |
| Use the bedroom only for sleep and sex | |
| Go to another room if unable to fall asleep within 15 to 20 minutes | |
| Read or engage in other quiet activities and return to bed only when sleepy | |
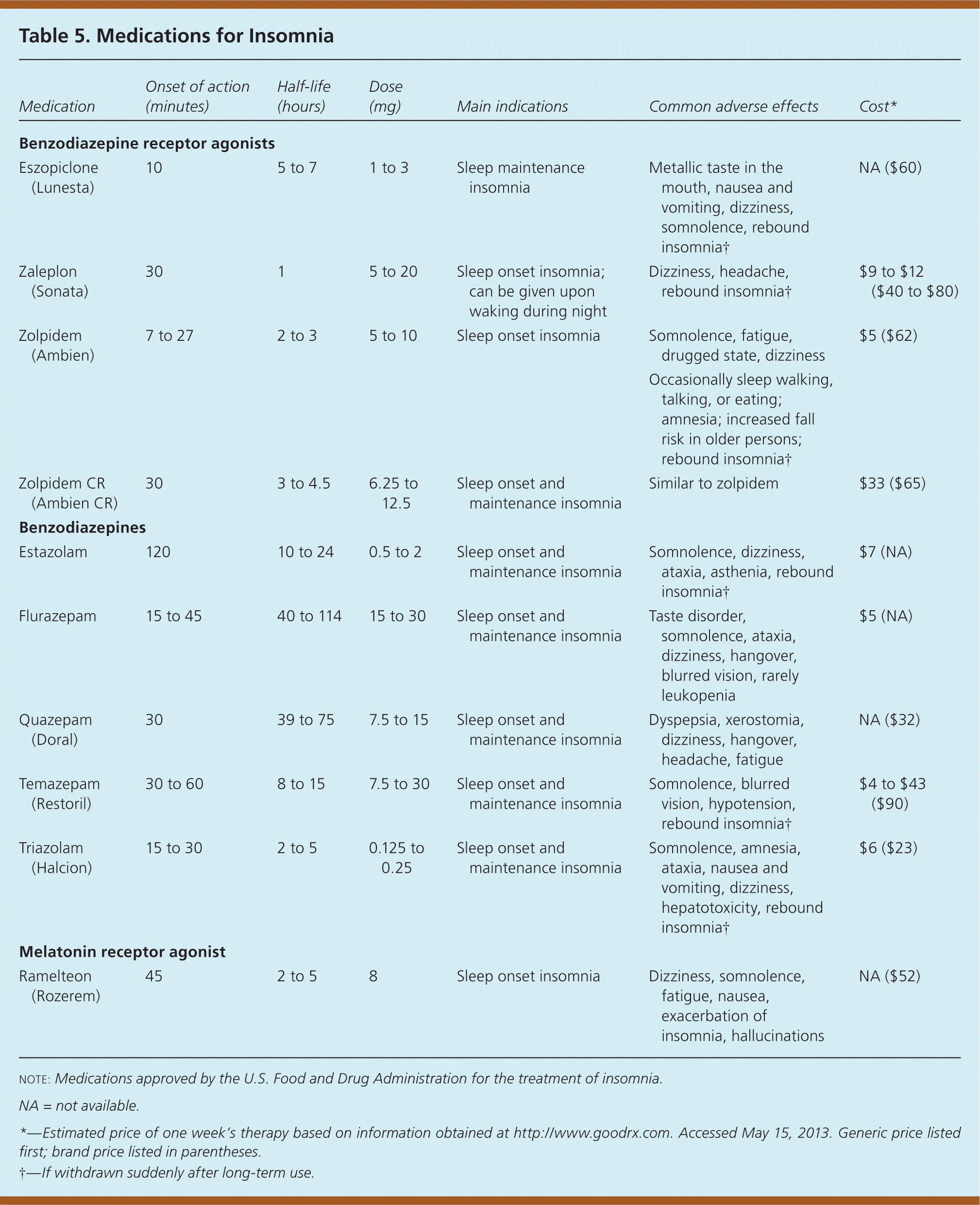
| Medication | Onset of action (minutes) | Half-life (hours) | Dose (mg) | Main indications | Common adverse effects | Cost* |
|---|---|---|---|---|---|---|
| Benzodiazepine receptor agonists | ||||||
| Eszopiclone (Lunesta) | 10 | 5 to 7 | 1 to 3 | Sleep maintenance insomnia | Metallic taste in the mouth, nausea and vomiting, dizziness, somnolence, rebound insomnia† | NA ($60) |
| Zaleplon (Sonata) | 30 | 1 | 5 to 20 | Sleep onset insomnia; can be given upon waking during night | Dizziness, headache, rebound insomnia† | $9 to $12 ($40 to $80) |
| Zolpidem (Ambien) | 7 to 27 | 2 to 3 | 5 to 10 | Sleep onset insomnia | Somnolence, fatigue, drugged state, dizziness | $5 ($62) |
| Occasionally sleep walking, talking, or eating; amnesia; increased fall risk in older persons; rebound insomnia† | ||||||
| Zolpidem CR (Ambien CR) | 30 | 3 to 4.5 | 6.25 to 12.5 | Sleep onset and maintenance insomnia | Similar to zolpidem | $33 ($65) |
| Benzodiazepines | ||||||
| Estazolam | 120 | 10 to 24 | 0.5 to 2 | Sleep onset and maintenance insomnia | Somnolence, dizziness, ataxia, asthenia, rebound insomnia† | $7 (NA) |
| Flurazepam | 15 to 45 | 40 to 114 | 15 to 30 | Sleep onset and maintenance insomnia | Taste disorder, somnolence, ataxia, dizziness, hangover, blurred vision, rarely leukopenia | $5 (NA) |
| Quazepam (Doral) | 30 | 39 to 75 | 7.5 to 15 | Sleep onset and maintenance insomnia | Dyspepsia, xerostomia, dizziness, hangover, headache, fatigue | NA ($32) |
| Temazepam (Restoril) | 30 to 60 | 8 to 15 | 7.5 to 30 | Sleep onset and maintenance insomnia | Somnolence, blurred vision, hypotension, rebound insomnia† | $4 to $43 ($90) |
| Triazolam (Halcion) | 15 to 30 | 2 to 5 | 0.125 to 0.25 | Sleep onset and maintenance insomnia | Somnolence, amnesia, ataxia, nausea and vomiting, dizziness, hepatotoxicity, rebound insomnia† | $6 ($23) |
| Melatonin receptor agonist | ||||||
| Ramelteon (Rozerem) | 45 | 2 to 5 | 8 | Sleep onset insomnia | Dizziness, somnolence, fatigue, nausea, exacerbation of insomnia, hallucinations | NA ($52) |
Although antihistamines, such as diphenhydramine (Benadryl), are widely used for insomnia, evidence on effectiveness and safety is very limited.16,17 In large clinical trials, melatonin had only minimal effect on initial sleep latency with little effect on total sleep time.18 Similarly, there are limited data on the effectiveness of sedating antidepressants, such as trazodone, for insomnia. However, they may be considered in patients with comorbid depression or if treatment with hypnotics fails.16,19
A meta-analysis showed similar short-term effectiveness with hypnotics used alone and with CBT, whereas CBT alone resulted in greater reduction in sleep latency.14 Similarly, other studies have shown that CBT combined with pharmacotherapy is superior to pharmacotherapy alone, and improvement with CBT alone is maintained at 10 to 24 months of follow-up.20,21 In some studies, pharmacotherapy performed slightly or no better than placebo on various outcome measures.20,21
Patients with insomnia who do not respond to medications and CBT should be referred to a sleep center for further testing and treatment.
RESTLESS LEGS SYNDROME
Restless legs syndrome is a neurologic disorder that affects between 2.5% and 15% of the U.S. general population.22 The condition is characterized by four essential features: (1) the intense urge to move the legs, usually accompanied or caused by uncomfortable sensations (e.g., “creepy crawly,” aching) in the legs; (2) symptoms that begin or worsen during periods of rest or inactivity; (3) symptoms that are partially or totally relieved by movements such as walking or stretching; and (4) symptoms that are worse or only occur in the evening or at night.10
The diagnosis of restless legs syndrome is based on history findings. Because the condition can be a symptom of iron deficiency,23–25 a ferritin measurement may be warranted, even in the absence of anemia. A ferritin level less than 50 ng per mL (112 pmol per L) is associated with restless legs syndrome.26 Restless legs syndrome is familial in about 50% of patients, although it may be idiopathic or secondary to chronic renal failure, antidepressant use, pregnancy, or neuropathy. Most patients with restless legs syndrome have periodic limb movements during sleep, which are characterized by periodic episodes of repetitive, stereotypic movements that predominantly occur in the lower extremities. Patients' bed partners may notice these movements.
Pharmacologic treatment of restless legs syndrome depends on the frequency of symptoms (Table 6).25 Dopaminergic agonists are first-line therapy for patients with nightly, persistent symptoms. Common adverse effects of dopaminergic agonists include insomnia, nasal congestion, swelling of the extremities, and daytime sleepiness. There have also been reports of increased tendency toward compulsive behaviors, such as gambling, in patients taking these medications. In patients with iron deficiency, iron supplementation may improve or resolve symptoms of restless legs syndrome.
Patients should be referred to a sleep clinic if symptoms of restless legs syndrome cannot be controlled or if augmentation symptoms (e.g., symptoms begin progressively earlier in the day or develop in the arms and trunk) occur despite appropriate treatment.

| Symptoms | First choice | Alternative choice | ||
|---|---|---|---|---|
| Occasional | Carbidopa/levodopa (Sinemet), 10 to 25 mg/100 mg at bedtime, as needed | Opiates
| ||
| Frequent | Dopaminergic agonist
| Opiates
| ||
| Nightly | Dopaminergic agonist
| Gabapentin (Neurontin) and pregabalin (Lyrica)
| ||
| Painful | Gabapentin and pregabalin
| Dopaminergic agonists
| ||
Patients Who Will Not Sleep
DELAYED SLEEP PHASE SYNDROME
Delayed sleep phase syndrome is a circadian rhythm disorder characterized by late sleep onset in association with a late wake-up time, resulting in excessive daytime sleepiness, insomnia, and daytime functional impairments.10 Delayed sleep phase syndrome may be genetic and/or socially reinforced (e.g., caused by Internet and computer use, watching television, or social activity at night). A sleep log and actigraphy can confirm the diagnosis if it is not evident by the history.
Treatment involves a combination of melatonin (0.3 to 3 mg given five hours before bed) and bright light exposure upon awakening. The patient's sleep-wake schedule and melatonin/bright light therapy regimen are incrementally advanced (i.e., moved earlier) every five to seven days until a desired sleep-wake schedule is reached.27
Patients with Excessive Daytime Sleepiness
It is difficult to differentiate daytime sleepiness from fatigue. Detailed questioning and use of the Epworth Sleepiness Scale28 (available at https://www.aafp.org/afp/2009/0301/p391.html#afp20090301p391-f1) may be helpful, but patients may underreport their symptoms. Although patients with certain sleep disorders, such as narcolepsy and OSA, present with excessive daytime sleepiness, it is important to rule out insufficient sleep before diagnosing pathologic causes of sleepiness.
NARCOLEPSY
Narcolepsy is a chronic, debilitating condition with a prevalence of around 0.02%.29 Clinical features of narcolepsy usually begin in the teens or twenties, although diagnosis may be established years later. Onset after 50 years of age is unusual. The classic tetrad of narcolepsy is excessive sleepiness; cataplexy; hallucinations upon falling asleep (hypnagogic) and/or upon awakening (hypnopompic); and sleep paralysis (generalized, transient inability to move or speak during sleep-wake transitions).
Cataplexy is the sudden decrease or loss of voluntary muscle tone following an emotional trigger—usually laughter, but sometimes anger or surprise. It can manifest as jaw dropping, head nodding, arms dropping to the side, knees sagging, or the patient collapsing to the floor. These attacks may last from a few seconds to a few minutes, and the patient's conscious awareness is preserved. The presence of cataplexy is highly specific for narcolepsy.
Referral to a sleep clinic is indicated if narcolepsy is suspected. The typical sequence of testing for suspected narcolepsy is one to two weeks of a sleep log or actigraphy to document sleep duration, followed by polysomnography to evaluate for other sleep disorders and document adequate sleep time, and concluding the next day with a multiple sleep latency test. The multiple sleep latency test is a daytime nap test to objectively assess for sleepiness and for onset of rapid eye movement (REM) sleep during naps. The combination of a mean sleep latency of less than eight minutes plus at least two naps with early onset REM sleep supports a diagnosis of narcolepsy.10
Cataplexy, sleep paralysis, and hypnagogic hallucinations can be treated with REM-suppressing antidepressants, such as venlafaxine (Effexor) or other selective serotonin reuptake inhibitors. Sleepiness may be managed with adequate sleep hygiene and scheduled daytime naps. Otherwise, modafinil (Provigil; 200 to 800 mg daily) or stimulants such as methylphenidate (Ritalin; 10 to 100 mg daily) or dextroamphetamine (5 to 60 mg daily) can be used. The U.S. Food and Drug Administration has approved gamma hydroxybutyric acid (sodium oxybate [Xyrem]) for use in patients with narcolepsy. Sodium oxybate is usually administered twice per night because of its short half-life, and is effective for both daytime sleepiness and cataplexy.
OBSTRUCTIVE SLEEP APNEA
OSA affects 4% of men and 2% of women,30 with a higher prevalence in older persons. It is characterized by partial (hypopnea) or complete (apnea) closure of the upper airway despite ongoing respiratory efforts. OSA leads to excessive daytime sleepiness, cognitive impairment, occupational accidents, and motor vehicle crashes. Evidence suggests that OSA also causes hypertension.31,32 Treatment with continuous positive airway pressure (CPAP) decreases blood pressure in patients with OSA,33 especially those with severe OSA and daytime sleepiness.34
Factors that should prompt evaluation for OSA include excessive daytime sleepiness, obesity, treatment-refractory hypertension, need for bariatric surgery, atrial fibrillation, congestive heart failure, stroke, nocturnal cardiac dysrhythmias, type 2 diabetes mellitus, and pulmonary hypertension. If OSA is suspected, a referral for polysomnography is indicated. An apnea-hypopnea index (number of apneas and hypopneas per hour of sleep) of five per hour or more establishes an OSA diagnosis. An apnea-hypopnea index of five to 15 per hour indicates mild disease; 15 to 30 per hour, moderate disease; and greater than 30 per hour, severe disease.
Portable home monitoring devices may be used as an alternative to polysomnography for diagnosing OSA in patients with high pretest probability of moderate to severe OSA.35 Portable home monitoring devices are less expensive than polysomnography and have the added advantage of patients being in their normal sleeping environment. Portable home monitoring devices may not be appropriate in patients who have congestive heart failure or chronic obstructive pulmonary disease because of lower accuracy, or in those with comorbid sleep disorders such as parasomnia.35
The most effective treatment for OSA is CPAP,36 which serves as a pneumatic splint to the upper airway. Autotitrating CPAP is another option that automatically adjusts the pressure within a set range in response to apneas, hypopneas, snoring, or flow limitation. Patients who cannot tolerate CPAP may be treated with bilevel positive airway pressure. Compliance with CPAP therapy is challenging for many patients. Other treatment options include weight loss, positional therapy, surgical approaches, oral appliances, and Provent therapy (a nasal device that has an expiratory valve to create positive end-expiratory pressure to keep the upper airway open). Choosing one of these therapies depends on OSA severity, patient preference, and tolerance.
Patients with Increased Movements During Sleep
REM SLEEP BEHAVIOR DISORDER
REM sleep behavior disorder is characterized by increased muscle tone during REM sleep, resulting in the acting out of dreams with possible harmful consequences to the patient or bed partner. The disorder is associated with parkinsonian syndromes and other α-synucleinopathies. It may also be secondary to other neurologic disorders (e.g., spinocerebellar ataxia), sleep disorders (e.g., severe OSA, narcolepsy), or use of medications (e.g., selective serotonin reuptake inhibitors). REM sleep behavior disorder is more common in men than in women. It is often suspected based on the bed partner observing the patient flailing the limbs, making vocalizations, and falling out of bed during sleep. REM sleep behavior disorder is usually diagnosed using polysomnography, which shows abnormally high muscle tone during REM sleep.
Environmental safety measures, such as removing sharp or fragile objects from the vicinity of the bed, should be taken to prevent injury. Melatonin has been shown to be effective with gradually increasing dosages until adequate response is obtained (add 3 mg at bedtime every two weeks to a maximum of 12 mg per night). Clonazepam (Klonopin), 0.5 to 2 mg at bedtime, is also effective in more than 90% of cases.37
PERIODIC LIMB MOVEMENTS
Periodic limb movements are characterized by periodic episodes of repetitive and stereotypic limb movements during sleep, predominantly in the lower extremities. If these movements are associated with sleep disturbance that results in daytime fatigue or sleepiness, it is then called periodic limb movement disorder. The disorder may be diagnosed only after other causes of daytime symptoms are excluded. Most patients with restless legs syndrome have periodic limb movements. These movements have also been associated with REM sleep behavior disorder and narcolepsy.
The diagnosis of periodic limb movement disorder is made using polysomnography when the periodic limb movement index is more than 15 per hour in adults and more than five per hour in children. The movements alone do not require treatment, but periodic limb movement disorder can be treated with pharmacologic agents used for restless legs syndrome.
Data Sources: A PubMed search was completed in clinical queries using the key terms sleep disorders, approach, and management. The search included meta-analyses, randomized controlled trials, clinical trials, guidelines, and reviews published after January 2000. We also searched Clinical Evidence, the Cochrane database, Essential Evidence Plus, the Institute for Clinical Systems Improvement, the National Guideline Clearinghouse database, and Embase. We reviewed practice guidelines from the American Academy of Sleep Medicine and the International Classification of Sleep Disorders, 2nd edition. Search dates: January through July 2012.
