Am Fam Physician. 2013;88(5):312-316
Related editorial: Potential Harms of Computed Tomography: The Role of Informed Consent.
Author disclosure: No relevant financial affiliations.
Family physicians often must determine the most appropriate diagnostic tests to order for their patients. It is essential to know the types of contrast agents, their risks, contraindications, and common clinical scenarios in which contrast-enhanced computed tomography is appropriate. Many types of contrast agents can be used in computed tomography: oral, intravenous, rectal, and intrathecal. The choice of contrast agent depends on route of administration, desired tissue differentiation, and suspected diagnosis. Possible contraindications for using intravenous contrast agents during computed tomography include a history of reactions to contrast agents, pregnancy, radioactive iodine treatment for thyroid disease, metformin use, and chronic or acutely worsening renal disease. The American College of Radiology Appropriateness Criteria is a useful online resource. Clear communication between the physician and radiologist is essential for obtaining the most appropriate study at the lowest cost and risk to the patient.
Recent estimates place the number of computed tomography (CT) scans performed annually in the United States at approximately 70 million.1 Given the cost and radiation exposure, it is critical that CT is appropriate and performed with optimal technique. The decision to order contrast-enhanced CT is based on the clinical question being asked. Contrast agents are used to differentiate between organs and improve lesion detection and characterization. The type of contrast agent and route of administration can increase the diagnostic yield of the study ordered.
Types of Contrast Agents
The most common contrast agents used with CT imaging are barium- and iodine-based. The specific agent and route of administration are based on clinical indications and patient factors. Clear communication between the physician and radiologist is essential for obtaining the most appropriate study at the lowest cost and risk to the patient.
ORAL CONTRAST
Oral contrast agents are barium- or iodine-based and are used for bowel opacification. The concentration of barium determines whether it enhances the diagnosis or causes an artifact and obscures pathology. Different imaging modalities require different concentrations of contrast for optimal detection of pathology. In general, oral contrast is used for most abdominal and pelvic CT scans unless there is no suspicion of bowel pathology (e.g., noncontrast CT to detect kidney stones) or when administration would delay a diagnosis in the trauma setting.
The concentration of barium used for fluoroscopy is more than 20 times that of the typical oral contrast suspension for CT. For example, the barium concentration from an upper gastrointestinal series or an enema will produce an artifact on abdominal CT because it is significantly more concentrated than bowel contrast agents used for CT. Barium enemas are also given after abdominal CT to allow time for the less-dense barium to leave the colon. Barium suspension from fluoroscopy or CT will not produce an artifact on abdominal magnetic resonance imaging. Barium suspensions are not nephrotoxic and can be used safely in patients with renal failure.
Water-soluble, iodine-based contrast agents can also be given orally. They are used for bowel opacification and are not nephrotoxic. As with barium agents, they must be diluted for CT compared with the concentrations used in fluoroscopy. Because there is a risk of aspiration-induced pulmonary edema with concentrated iodine-based contrast agents, patients must be carefully selected. Oral contrast can be administered through a nasogastric tube to minimize the risk of aspiration.
| Clinical recommendation | Evidence rating | Reference |
|---|---|---|
| A baseline serum creatinine level should be obtained up to one month before administration of intravenous contrast agents in patients with suspected renal insufficiency. | C | 7 |
| Premedication with antihistamines and corticosteroids is recommended in patients with a history of mild to moderate reactions to intravenous contrast agents. | C | 7 |
| In patients with normal renal function, repeat measurement of serum creatinine is not recommended after outpatient administration of intravenous contrast agents. | C | 7 |
RECTAL CONTRAST
Rectal contrast can be used in patients with a suspected penetrating colonic injury.2 Rectal contrast does not always reach the cecum, so the small bowel and appendix can remain unopacified. Some centers use oral contrast to evaluate for appendicitis; some do not use bowel contrast,3 and others use rectal contrast to avoid the delay associated with oral administration.4
IV CONTRAST
Iodine-based intravenous (IV) contrast agents are used for opacification of vascular structures and solid abdominal and pelvic organs. The major families of contrast agents are ionic and nonionic. Contrast agents can be further classified as high or low osmolality, based on the iodine concentration. Most centers use nonionic contrast agents (which are generally low osmolality) for IV contrast studies.5 The rate of major reactions (e.g., anaphylaxis, death) is the same for ionic and nonionic IV contrast agents—an estimated one in 170,000 administrations—but nonionic contrast has a lower rate of minor reactions.6 Approximately 5% to 12% of patients who receive high-osmolality contrast have adverse reactions, most of which are mild or moderate.7 Use of low-osmolality contrast has been associated with a reduction in adverse effects. Children have a lower incidence of reactions to IV contrast agents, and most of these are mild (0.18% for low-osmolality agents).7,8
Risk factors for contrast reactions include multiple drug allergies and asthma. Although many radiology departments screen for shellfish allergy, there is no cross-reactivity between shellfish and iodinated contrast. However, patients with a documented anaphylactic reaction to any medication are at higher risk of a reaction to iodinated contrast.9,10
Many centers screen outpatients with suspected renal insufficiency by measuring serum creatinine one month before administration of contrast agents. Creatinine cutoffs vary among institutions, but generally range between 1.5 and 2 mg per dL (132.6 to 176.8 μmol per L) before alternative imaging strategies are considered.7 An increasing creatinine level that is still within normal limits is also concerning, and alternative imaging strategies should be considered. In patients with normal renal function, repeat measurement of serum creatinine is not recommended after outpatient administration of IV contrast agents.7
INTRATHECAL CONTRAST
Intrathecal iodinated contrast is given during myelography to evaluate spinal or basal cisternal disease and cerebrospinal fluid leaks.11 Plain radiography of the spine is then obtained under fluoroscopic guidance. CT may also be ordered for cross-sectional images of the spine with contrast in the thecal sac.
Contraindications to IV Contrast
Concerns for using IV contrast during CT include a history of reactions to contrast agents, pregnancy, treatment of thyroid disease with radioactive iodine, use of metformin (Glucophage), and chronic or acutely worsening renal disease. Contrast may also be avoided when the suspected pathology is likely to be visible on noncontrast-enhanced CT.
HISTORY OF REACTIONS TO CONTRAST AGENTS
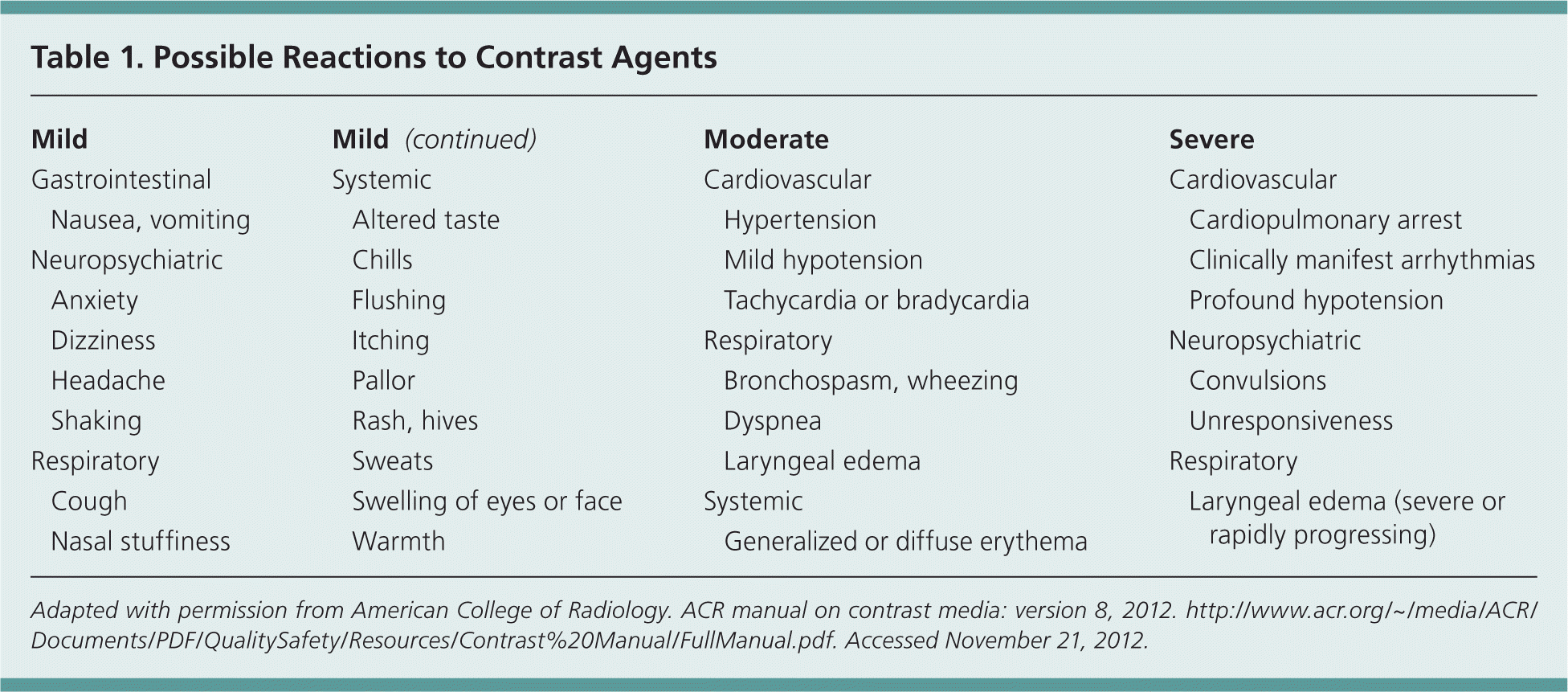
A history should be obtained to determine if the reaction was mild (which typically requires observation but not treatment), moderate (which requires prompt treatment), or severe (which requires rapid intervention and, often, hospitalization). Possible reactions are listed in Table 1.7 If a patient has had a previous minor reaction to an IV iodinated contrast agent, precontrast administration of oral or IV corticosteroids and diphenhydramine (Benadryl) may decrease their risk (Table 27 ). A history of anaphylactic reactions would preclude IV contrast except in extreme emergencies.
| Mild | |
| Gastrointestinal | |
| Nausea, vomiting | |
| Neuropsychiatric | |
| Anxiety | |
| Dizziness | |
| Headache | |
| Shaking | |
| Respiratory | |
| Cough | |
| Nasal stuffiness | |
| Systemic | |
| Altered taste | |
| Chills | |
| Flushing | |
| Itching | |
| Pallor | |
| Rash, hives | |
| Sweats | |
| Swelling of eyes or face | |
| Warmth | |
| Moderate | |
| Cardiovascular | |
| Hypertension | |
| Mild hypotension | |
| Tachycardia or bradycardia | |
| Respiratory | |
| Bronchospasm, wheezing | |
| Dyspnea | |
| Laryngeal edema | |
| Systemic | |
| Generalized or diffuse erythema | |
| Severe | |
| Cardiovascular | |
| Cardiopulmonary arrest | |
| Clinically manifest arrhythmias | |
| Profound hypotension | |
| Neuropsychiatric | |
| Convulsions | |
| Unresponsiveness | |
| Respiratory | |
| Laryngeal edema (severe or rapidly progressing) | |

| Methylprednisolone (Medrol), 32 mg orally 12 and 2 hours before contrast administration; plus diphenhydramine (Benadryl), 50 mg intravenously, intramuscularly, or orally 1 hour before contrast administration |
| Prednisone, 50 mg orally 13, 7, and 1 hour before contrast administration; plus diphenhydramine, 50 mg intravenously, intramuscularly, or orally 1 hour before contrast administration |
PREGNANCY
Iodinated contrast crosses the human placenta. No mutagenic or teratogenic effects have been shown with nonionic, low-osmolality contrast in animal studies. The American College of Radiology recommends using IV iodinated contrast in pregnant women when the information needed affects the care of the patient and fetus and cannot be obtained without contrast, and when the referring physician thinks that imaging should not wait until after the pregnancy.7
THYROID DISEASE
Iodinated contrast media can saturate the thyroid gland and significantly reduce uptake of iodine 131, rendering the treatment ineffective. Iodinated contrast should be avoided for two months before administration of iodine 131. However, if contrast must be administered within two months of iodine 131 treatment, consultation with an endocrinologist should be considered.7 Administration of iodinated contrast may provoke thyrotoxicosis, although this is rare.12
METFORMIN USE
Approximately 90% of absorbed metformin is excreted by the kidneys within 24 hours. The risk of developing metabolic acidosis with metformin use is rare, but is higher in persons with chronic renal disease or transient impairment of renal function. There is no direct interaction between metformin and IV radiologic contrast agents. However, IV radiologic contrast may cause transient alteration in renal function, which could impair metformin clearance, leading to a higher risk of metabolic acidosis. The U.S. Food and Drug Administration advises that metformin should be withheld at the time of IV contrast administration and for 48 hours afterward, and resumed only after reevaluation of renal status (i.e., return to baseline serum creatinine level).13
Lactic acidosis has never been documented in patients with normal renal function who are receiving metformin. The American College of Radiology proposes a three-tiered risk assessment for patients receiving metformin in whom IV contrast is to be administered (Table 3).7 Many institutions have their own protocols for metformin administration with IV contrast, so physicians should be aware of the standard of care in their community.

| Category | Risk assessment findings | Action |
|---|---|---|
| 1 | Normal renal function and no comorbid disorder* | Metformin (Glucophage) can be continued when contrast is administered; serum creatinine does not need to be measured |
| 2 | Normal renal function and at least one comorbid disorder* | Metformin should be discontinued when contrast is administered; if the patient remains clinically stable and has no new intercurrent risk factors for renal impairment, metformin may be resumed in 48 hours without repeating serum creatinine measurement |
| 3 | Known renal dysfunction | Metformin should be discontinued when contrast is administered; resume only after careful reevaluation and monitoring of renal status |
RENAL FAILURE
Iodinated contrast agents can cause reversible acute renal failure. Risk factors include chronic kidney disease, diabetes mellitus, heart failure, older age, anemia, left ventricular systolic dysfunction, and contrast volume. Recent studies suggest that a combination of hydration, sodium bicarbonate, N-acetylcysteine, and decreased contrast volume may reduce this risk in high-risk populations.14,15
The question of whether this risk has been overstated has been raised in the medical literature. One study showed similar increases in serum creatinine levels between inpatient populations who received IV iodinated contrast and those who did not.16
Diagnostic Indications: Noncontrast-Enhanced CT vs. IV Contrast
Noncontrast-enhanced CT is used in patients with head trauma and acute stroke. Unenhanced CT is also used in patients with spine and extremity trauma. High-resolution CT, which is used to evaluate diffuse lung disease, does not use IV contrast.17 Noncontrast imaging of the abdomen is routinely done to screen for renal stones in patients with flank pain.18 Common clinical scenarios in which noncontrast-enhanced CT is appropriate are summarized in Table 4,19 and common clinical scenarios in which contrast enhancement is recommended are summarized in Table 5.19 Indications for selection of imaging studies for specific clinical scenarios can be searched using the American College of Radiology Appropriateness Criteria at http://www.acr.org/ac.19

| Clinical scenario | Protocol / comments |
|---|---|
| Acute stroke (suspected) | Noncontrast-enhanced CT of the head is the preferred initial study if performed within three hours of acute symptom onset; contrast-enhanced CT should be obtained for patients with symptoms lasting longer than three hours; contrast-enhanced CT combined with CT angiography of the neck may be needed for follow-up |
| Closed head injury | Spinal CT if spinal injury is suspected |
| Diffuse lung disease/chronic dyspnea | Thin section high-resolution CT without contrast |
| Extremity soft tissue swelling, infection, or trauma | Contrast is necessary if vascular involvement or injury is suspected |
| Kidney stone (suspected) | Specify renal stone protocol |
| Spinal trauma (suspected) | Scan suspected area of trauma in cervical, thoracic, or lumbar spine |

| Clinical scenario | Protocol / comments |
|---|---|
| Acute appendicitis | Abdominal and pelvic CT; oral or rectal contrast agent based on institutional preference |
| Cancer staging | Protocols vary depending on cancer type and stage |
| Diverticulitis; suspected complications of inflammatory bowel disease | Intravenous contrast agent for diverticulitis; oral and/or rectal contrast agent can be administered to visualize bowel |
| Pancreatitis | Noncontrast-enhanced CT is sensitive for calcifications (chronic pancreatitis); contrast-enhanced CT is best for evolving pancreatitis or pancreatic pseudocyst |
| Pulmonary embolism (suspected) | Many centers now include venous phase CT of the pelvis and lower extremities in combination with CT angiography of the lung |
When to Order CT with and Without Contrast
Disease processes that involve calcifications may benefit from noncontrast-enhanced images because contrast may mask the appearance of calcifications. Typically, CT focusing on vascular disease (e.g., aneurysm, dissection) or renal or pancreatic pathology may include noncontrast-enhanced images to identify calcifications.20 Contrast-enhanced studies would be performed on the same day, in the same setting.
Data Sources: We used the term radiologic contrast to search the following: PubMed Clinical Queries (systematic reviews); the OVID database (all evidence-based medicine reviews; Cochrane Database of Systematic Reviews, ACP Journal Club, Database of Abstracts of Reviews of Effects, Cochrane Central Trial Registry, Cochrane Methodology Register, Health Technology Assessment, and NHS Economic Effectiveness Database); Dynamed; and the U.S. Preventive Services Task Force and Agency for Healthcare Research and Quality clinical guidelines and evidence reports. Search dates: November 2009 and April 27, 2010.