This is a corrected version of the article that appeared in print.
Am Fam Physician. 2013;88(7):435-440
Related editorials: Should Children with Acute Otitis Media Routinely Be Treated with Antibiotics? Yes: Routine Treatment Makes Sense for Symptomatic, Emotional, and Economic Reasons and No: Most Children Older Than Two Years Do Not Require Antibiotics
Author disclosure: No relevant financial affiliations.
Acute otitis media is diagnosed in patients with acute onset, presence of middle ear effusion, physical evidence of middle ear inflammation, and symptoms such as pain, irritability, or fever. Acute otitis media is usually a complication of eustachian tube dysfunction that occurs during a viral upper respiratory tract infection. Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis are the most common organisms isolated from middle ear fluid. Management of acute otitis media should begin with adequate analgesia. Antibiotic therapy can be deferred in children two years or older with mild symptoms. High-dose amoxicillin (80 to 90 mg per kg per day) is the antibiotic of choice for treating acute otitis media in patients who are not allergic to penicillin. Children with persistent symptoms despite 48 to 72 hours of antibiotic therapy should be reexamined, and a second-line agent, such as amoxicillin/clavulanate, should be used if appropriate. Otitis media with effusion is defined as middle ear effusion in the absence of acute symptoms. Antibiotics, decongestants, or nasal steroids do not hasten the clearance of middle ear fluid and are not recommended. Children with evidence of anatomic damage, hearing loss, or language delay should be referred to an otolaryngologist.
Otitis media is among the most common issues faced by physicians caring for children. Approximately 80% of children will have at least one episode of acute otitis media (AOM), and between 80% and 90% will have at least one episode of otitis media with effusion (OME) before school age.1,2 This review of diagnosis and treatment of otitis media is based, in part, on the University of Michigan Health System's clinical care guideline for otitis media.2
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| An AOM diagnosis requires moderate to severe bulging of the tympanic membrane, new onset of otorrhea not caused by otitis externa, or mild bulging of the tympanic membrane associated with recent onset of ear pain (less than 48 hours) or erythema. | C | 8 |
| Middle ear effusion can be detected with the combined use of otoscopy, pneumatic otoscopy, and tympanometry. | C | 9 |
| Adequate analgesia is recommended for all children with AOM. | C | 8, 15 |
| Deferring antibiotic therapy for lower-risk children with AOM should be considered. | C | 19, 20, 23 |
| High-dose amoxicillin (80 to 90 mg per kg per day in two divided doses) is the first choice for initial antibiotic therapy in children with AOM. | C | 8, 10 |
| Children with middle ear effusion and anatomic damage or evidence of hearing loss or language delay should be referred to an otolaryngologist. | C | 11 |
Etiology and Risk Factors
Usually, AOM is a complication of eustachian tube dysfunction that occurred during an acute viral upper respiratory tract infection. Bacteria can be isolated from middle ear fluid cultures in 50% to 90% of cases of AOM and OME. Streptococcus pneumoniae, Haemophilus influenzae (nontypable), and Moraxella catarrhalis are the most common organisms.3,4 H. influenzae has become the most prevalent organism among children with severe or refractory AOM following the introduction of the pneumococcal conjugate vaccine.5–7 Risk factors for AOM are listed in Table 1.8,9
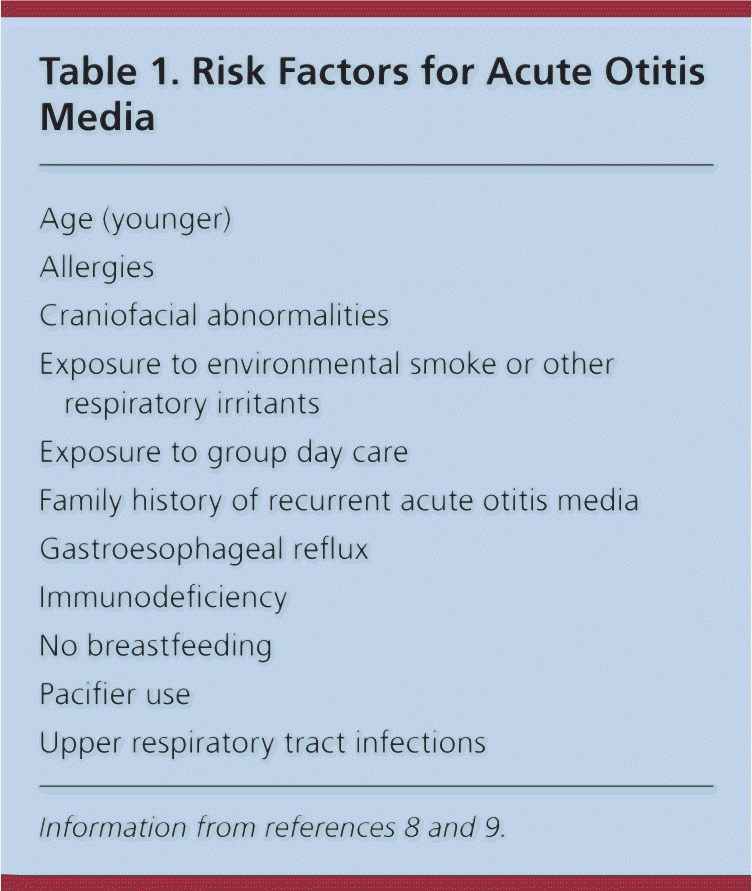
| Age (younger) |
| Allergies |
| Craniofacial abnormalities |
| Exposure to environmental smoke or other respiratory irritants |
| Exposure to group day care |
| Family history of recurrent acute otitis media |
| Gastroesophageal reflux |
| Immunodeficiency |
| No breastfeeding |
| Pacifier use |
| Upper respiratory tract infections |
Diagnosis
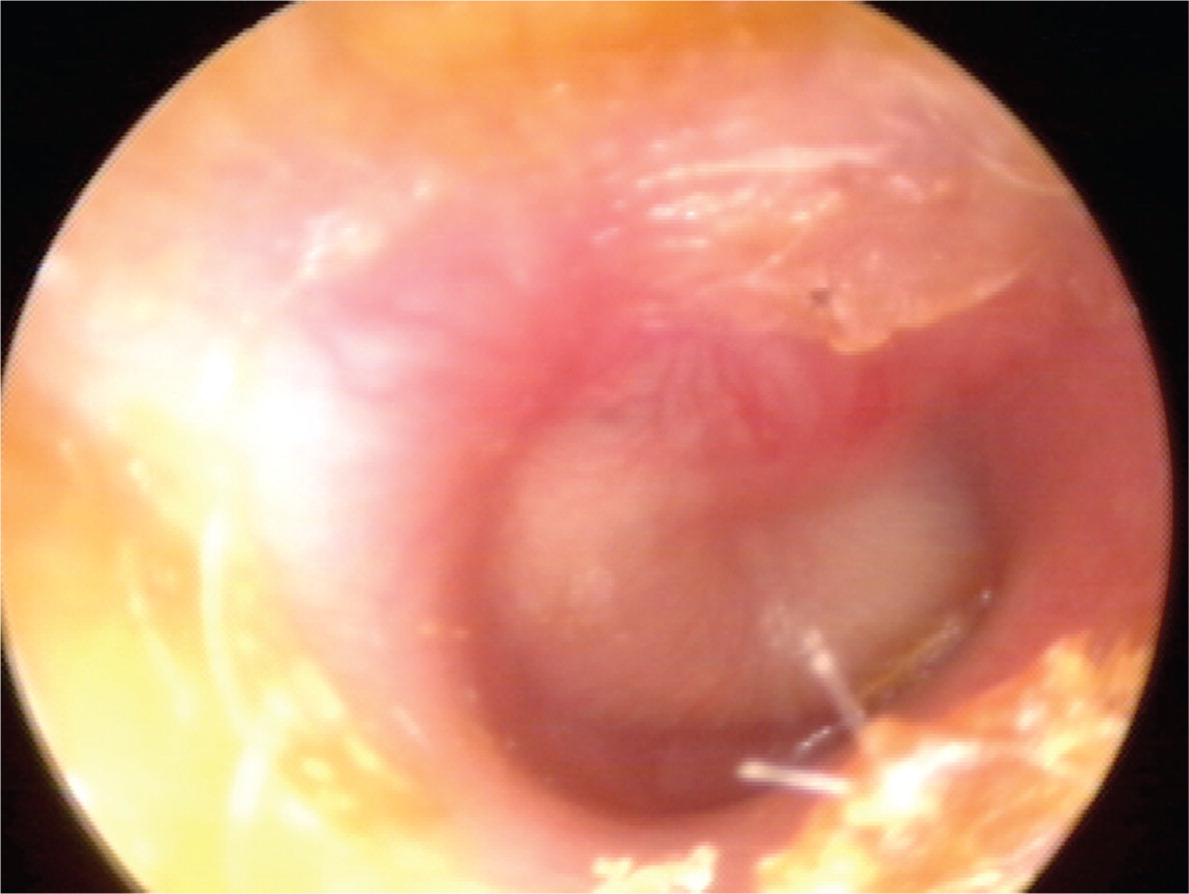
Previous diagnostic criteria for AOM were based on symptomatology without otoscopic findings of inflammation. The updated American Academy of Pediatrics guideline endorses more stringent otoscopic criteria for diagnosis.8 An AOM diagnosis requires moderate to severe bulging of the tympanic membrane (Figure 1), new onset of otorrhea not caused by otitis externa, or mild bulging of the tympanic membrane associated with recent onset of ear pain (less than 48 hours) or erythema. AOM should not be diagnosed in children who do not have objective evidence of middle ear effusion.8 An inaccurate diagnosis can lead to unnecessary treatment with antibiotics and contribute to the development of antibiotic resistance.
OME is defined as middle ear effusion in the absence of acute symptoms.10,11 If OME is suspected and the presence of effusion on otoscopy is not evident by loss of landmarks, pneumatic otoscopy, tympanometry, or both should be used.11 Pneumatic otoscopy is a useful technique for the diagnosis of AOM and OME8–12 and is 70% to 90% sensitive and specific for determining the presence of middle ear effusion. By comparison, simple otoscopy is 60% to 70% accurate.10,11 Inflammation with bulging of the tympanic membrane on otoscopy is highly predictive of AOM.7,8,12 Pneumatic otoscopy is most helpful when cerumen is removed from the external auditory canal.
Tympanometry and acoustic reflectometry are valuable adjuncts to otoscopy or pneumatic otoscopy.8,10,11 Tympanometry has a sensitivity and specificity of 70% to 90% for the detection of middle ear fluid, but is dependent on patient cooperation.13 Combined with normal otoscopy findings, a normal tympanometry result may be helpful to predict absence of middle ear effusion. Acoustic reflectometry has lower sensitivity and specificity in detecting middle ear effusion and must be correlated with the clinical examination.14 Tympanocentesis is the preferred method for detecting the presence of middle ear effusion and documenting bacterial etiology,8 but is rarely performed in the primary care setting.
Management of Acute Otitis Media
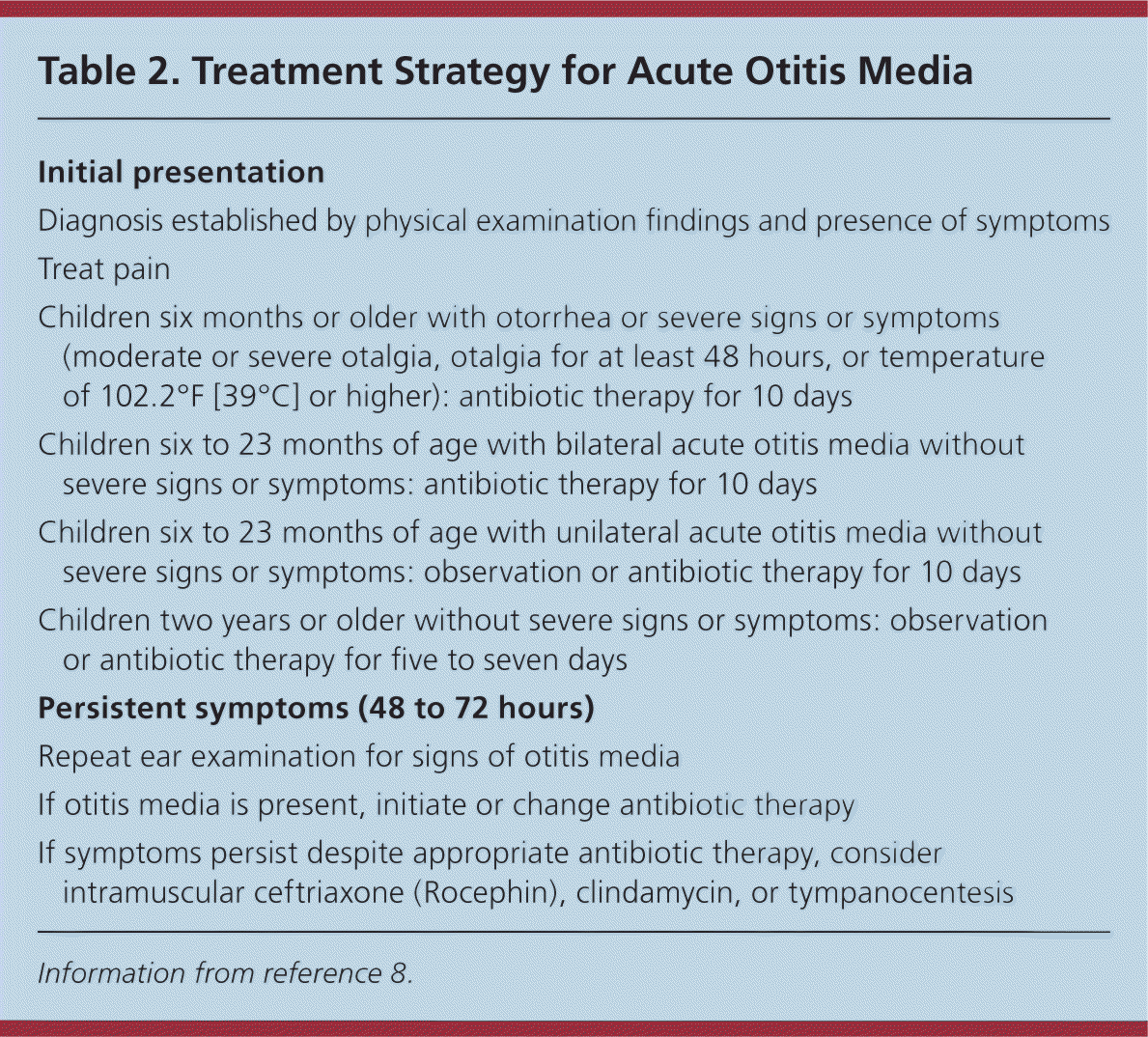
| Initial presentation |
| Diagnosis established by physical examination findings and presence of symptoms |
| Treat pain |
| Children six months or older with otorrhea or severe signs or symptoms (moderate or severe otalgia, otalgia for at least 48 hours, or temperature of 102.2°F [39°C] or higher): antibiotic therapy for 10 days |
| Children six to 23 months of age with bilateral acute otitis media without severe signs or symptoms: antibiotic therapy for 10 days |
| Children six to 23 months of age with unilateral acute otitis media without severe signs or symptoms: observation or antibiotic therapy for 10 days |
| Children two years or older without severe signs or symptoms: observation or antibiotic therapy for five to seven days |
| Persistent symptoms (48 to 72 hours) |
| Repeat ear examination for signs of otitis media |
| If otitis media is present, initiate or change antibiotic therapy |
| If symptoms persist despite appropriate antibiotic therapy, consider intramuscular ceftriaxone (Rocephin), clindamycin, or tympanocentesis |
ANALGESICS
Analgesics are recommended for symptoms of ear pain, fever, and irritability.8,15 Analgesics are particularly important at bedtime because disrupted sleep is one of the most common symptoms motivating parents to seek care.2 Ibuprofen and acetaminophen have been shown to be effective.16 Ibuprofen is preferred, given its longer duration of action and its lower toxicity in the event of overdose.2 Topical analgesics, such as benzocaine, can also be helpful.17
OBSERVATION VS. ANTIBIOTIC THERAPY
Antibiotic-resistant bacteria remain a major public health challenge. A widely endorsed strategy for improving the management of AOM involves deferring antibiotic therapy in patients least likely to benefit from antibiotics.18 Antibiotics should be routinely prescribed for children with AOM who are six months or older with severe signs or symptoms (i.e., moderate or severe otalgia, otalgia for at least 48 hours, or temperature of 102.2°F [39°C] or higher), and for children younger than two years with bilateral AOM regardless of additional signs or symptoms.8
Among children with mild symptoms, observation may be an option in those six to 23 months of age with unilateral AOM, or in those two years or older with bilateral or unilateral AOM.8,10,19 A large prospective study of this strategy found that two out of three children will recover without antibiotics.20 Recently, the American Academy of Family Physicians recommended not prescribing antibiotics for otitis media in children two to 12 years of age with nonsevere symptoms if observation is a reasonable option.21,22 If observation is chosen, a mechanism must be in place to ensure appropriate treatment if symptoms persist for more than 48 to 72 hours. Strategies include a scheduled follow-up visit or providing patients with a backup antibiotic prescription to be filled only if symptoms persist.8,20,23
ANTIBIOTIC SELECTION
[ corrected] Table 3 summarizes the antibiotic options for children with AOM.8 High-dose amoxicillin should be the initial treatment in the absence of a known allergy.8,10,24 The advantages of amoxicillin include low cost, acceptable taste, safety, effectiveness, and a narrow microbiologic spectrum. Children who have taken amoxicillin in the past 30 days, who have conjunctivitis, or who need coverage for β-lactamase–positive organisms should be treated with high-dose amoxicillin/clavulanate (Augmentin).8
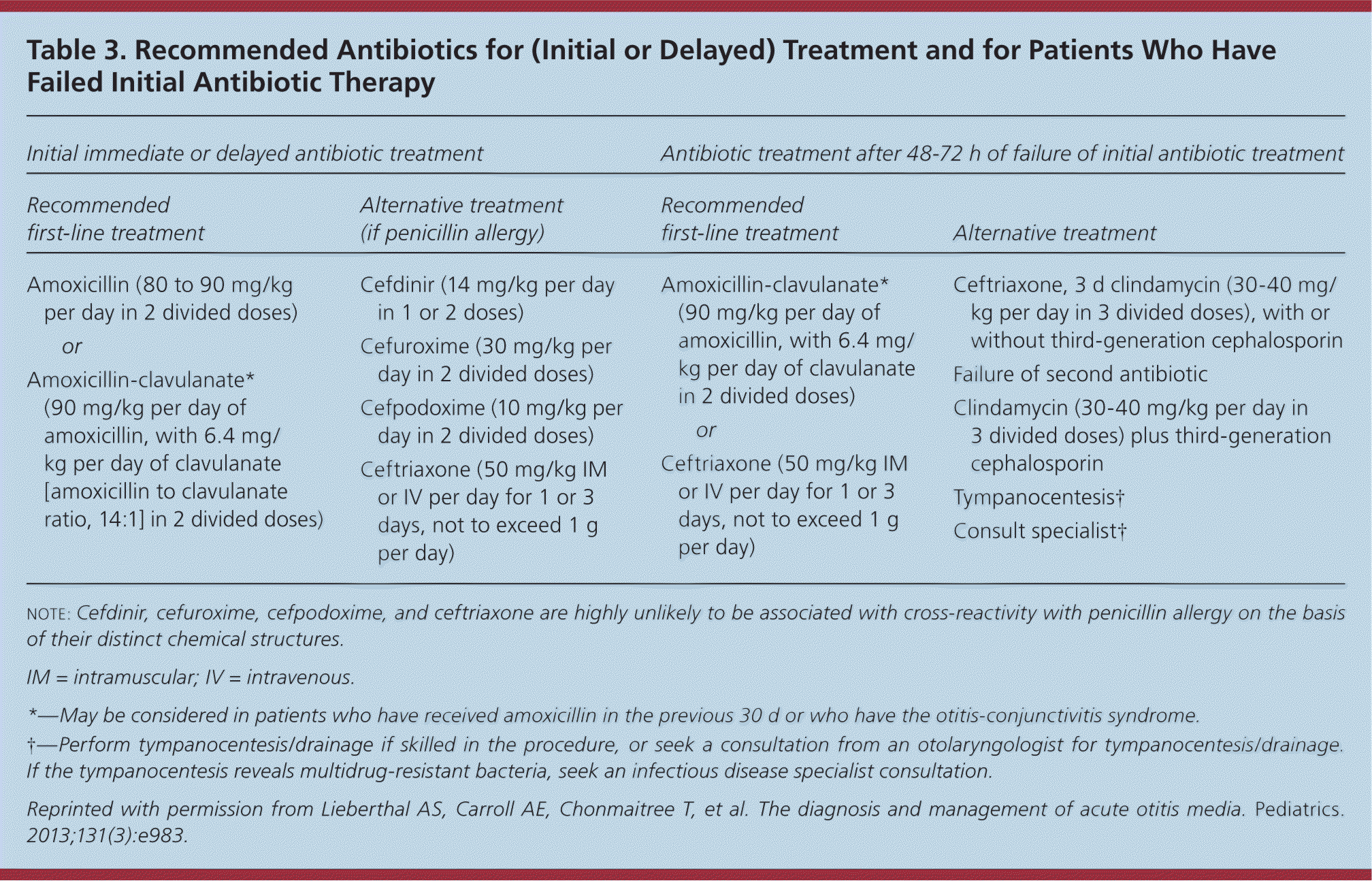
| Initial immediate or delayed antibiotic treatment | Antibiotic treatment after 48–72 h of failure of initial antibiotic treatment | ||||
|---|---|---|---|---|---|
| Recommended first-line treatment | Alternative treatment (if penicillin allergy) | Recommended first-line treatment | Alternative treatment | ||
|
|
| |||
Oral cephalosporins, such as cefuroxime (Ceftin), may be used in children who are allergic to penicillin. Recent research indicates that the degree of cross reactivity between penicillin and second- and third-generation cephalosporins is low (less than 10% to 15%), and avoidance is no longer recommended.25 Because of their broad-spectrum coverage, third-generation cephalosporins in particular may have an increased risk of selection of resistant bacteria in the community.26 High-dose azithromycin (Zithromax; 30 mg per kg, single dose) appears to be more effective than the commonly used five-day course, and has a similar cure rate as high-dose amoxicillin/clavulanate.8,27,28 However, excessive use of azithromycin is associated with increased resistance, and routine use is not recommended.8 Trimethoprim/sulfamethoxazole is no longer effective for the treatment of AOM due to evidence of S. pneumoniae resistance.29
Intramuscular or intravenous ceftriaxone (Rocephin) should be reserved for episodes of treatment failure or when a serious comorbid bacterial infection is suspected.2 One dose of ceftriaxone may be used in children who cannot tolerate oral antibiotics because it has been shown to have similar effectiveness as high-dose amoxicillin.30,31 A three-day course of ceftriaxone is superior to a one-day course in the treatment of nonresponsive AOM caused by penicillin-resistant S. pneumoniae.31 Although some children will likely benefit from intramuscular ceftriaxone, overuse of this agent may significantly increase high-level penicillin resistance in the community.2 High-level penicillin-resistant pneumococci are also resistant to first- and third-generation cephalosporins.
Antibiotic therapy for AOM is often associated with diarrhea.8,10,32 Probiotics and yogurts containing active cultures reduce the incidence of diarrhea and should be suggested for children receiving antibiotics for AOM.32 There is no compelling evidence to support the use of complementary and alternative treatments in AOM.8
PERSISTENT OR RECURRENT AOM
Children with persistent, significant AOM symptoms despite at least 48 to 72 hours of antibiotic therapy should be reexamined.8 If a bulging, inflamed tympanic membrane is observed, therapy should be changed to a second-line agent.2 For children initially on amoxicillin, high-dose amoxicillin/clavulanate is recommended.8,10,28
For children with an amoxicillin allergy who do not improve with an oral cephalosporin, intramuscular ceftriaxone, clindamycin, or tympanocentesis may be considered.4,8 If symptoms recur more than one month after the initial diagnosis of AOM, a new and unrelated episode of AOM should be assumed.10 For children with recurrent AOM (i.e., three or more episodes in six months, or four episodes within 12 months with at least one episode during the preceding six months) with middle ear effusion, tympanostomy tubes may be considered to reduce the need for systemic antibiotics in favor of observation, or topical antibiotics for tube otorrhea.8,10 However, tympanostomy tubes may increase the risk of long-term tympanic membrane abnormalities and reduced hearing compared with medical therapy.33 Other strategies may help prevent recurrence (Table 4).34–37

| Check for undiagnosed allergies leading to chronic rhinorrhea |
| Eliminate bottle propping and pacifiers34 |
| Eliminate exposure to passive smoke35 |
| Routinely immunize with the pneumococcal conjugate and influenza vaccines36 |
| Use xylitol gum in appropriate children (two pieces, five times a day after meals and chewed for at least five minutes)37 |
Probiotics, particularly in infants, have been suggested to reduce the incidence of infections during the first year of life. Although available evidence has not demonstrated that probiotics prevent respiratory infections,38 probiotics do not cause adverse effects and need not be discouraged. Antibiotic prophylaxis is not recommended.8
Management of OME
Management of OME is summarized in Table 5.11 Two rare complications of OME are transient hearing loss potentially associated with language delay, and chronic anatomic injury to the tympanic membrane requiring reconstructive surgery.11 Children should be screened for speech delay at all visits. If a developmental delay is apparent or middle ear structures appear abnormal, the child should be referred to an otolaryngologist.11 Antibiotics, decongestants, and nasal steroids do not hasten the clearance of middle ear fluid and are not recommended.11,39

| Evaluate tympanic membranes at every well-child and sick visit if feasible; perform pneumatic otoscopy or tympanometry when possible (consider removing cerumen) |
| If transient effusion is likely, reevaluate at three-month intervals, including screening for language delay; if there is no anatomic damage or evidence of developmental or behavioral complications, continue to observe at three- to six-month intervals; if complications are suspected, refer to an otolaryngologist |
| For effusion that appears to be associated with anatomic damage, such as adhesive otitis media or retraction pockets, reevaluate in four to six weeks; if abnormality persists, refer to an otolaryngologist |
| Antibiotics, decongestants, and nasal steroids are not indicated |
Tympanostomy Tube Placement
Tympanostomy tubes are appropriate for children six months to 12 years of age who have had bilateral OME for three months or longer with documented hearing difficulties, or for children with recurrent AOM who have evidence of middle ear effusion at the time of assessment for tube candidacy. Tubes are not indicated in children with a single episode of OME of less than three months' duration, or in children with recurrent AOM who do not have middle ear effusion in either ear at the time of assessment for tube candidacy. Children with chronic OME who did not receive tubes should be reevaluated every three to six months until the effusion is no longer present, hearing loss is detected, or structural abnormalities of the tympanic membrane or middle ear are suspected.40
Children with tympanostomy tubes who present with acute uncomplicated otorrhea should be treated with topical antibiotics and not oral antibiotics. Routine, prophylactic water precautions such as ear plugs, headbands, or avoidance of swimming are not necessary for children with tympanostomy tubes.40
Special Populations
INFANTS EIGHT WEEKS OR YOUNGER
Young infants are at increased risk of severe sequelae from suppurative AOM. Middle ear pathogens found in neonates younger than two weeks include group B streptococcus, gram-negative enteric bacteria, and Chlamydia trachomatis.41 Febrile neonates younger than two weeks with apparent AOM should have a full sepsis workup, which is indicated for any febrile neonate.41 Empiric amoxicillin is acceptable for infants older than two weeks with upper respiratory tract infection and AOM who are otherwise healthy.42
ADULTS
There is little published information to guide the management of otitis media in adults. Adults with new-onset unilateral, recurrent AOM (greater than two episodes per year) or persistent OME (greater than six weeks) should receive additional evaluation to rule out a serious underlying condition, such as mechanical obstruction, which in rare cases is caused by nasopharyngeal carcinoma. Isolated AOM or transient OME may be caused by eustachian tube dysfunction from a viral upper respiratory tract infection; however, adults with recurrent AOM or persistent OME should be referred to an otolaryngologist.
Data Sources: We reviewed the updated Agency for Healthcare Research and Quality Evidence Report on the management of acute otitis media, which included a systematic review of the literature through July 2010. We searched Medline for literature published since July 1, 2010, using the keywords human, English language, guidelines, controlled trials, and cohort studies. Searches were performed using the following terms: otitis media with effusion or serous effusion, recurrent otitis media, acute otitis media, otitis media infants 0–4 weeks, otitis media adults, otitis media and screening for speech delay, probiotic bacteria after antibiotics. Search dates: October 2011 and August 14, 2013.
EDITOR'S NOTE: This article is based, in part, on an institution-wide guideline developed at the University of Michigan. As part of the guideline development process, authors of this article, including representatives from primary and specialty care, convened to review current literature and make recommendations for diagnosis and treatment of otitis media and otitis media with effusion in primary care.