In the first editorial of this series, we described the harms of overscreening, defined as screening patients who are not in the recommended age range or ordering screening tests more often than necessary. Overscreening can exacerbate the problem of overdiagnosis, which is the topic of this editorial. Overdiagnosis occurs when a disease is diagnosed and treated, even though that disease never would have harmed the patient if left untreated. As a result, the patient is exposed to the inconvenience, worry, expense, and adverse effects of treatment, with little or no possibility of benefit. The concept of overdiagnosis has received increasing attention in the literature, especially with regard to breast and prostate cancers.
For example, an autopsy study of trauma patients in Detroit found evidence of prostate cancer in 45% of men in their 50s and nearly 70% in their 60s.1 However, the lifetime prevalence at the time was only 10% for clinically diagnosed prostate cancer and only 3% for fatal prostate cancer.2 With the introduction of the prostate-specific antigen test, the lifetime prevalence of clinically diagnosed prostate cancer has increased to more than 15%, but the risk of dying from prostate cancer has remained at just under 3%.3 Thus, most men with screen-detected prostate cancer were overdiagnosed; their prostate cancer was never going to cause symptoms or death. Although one death from prostate cancer would possibly be averted by screening approximately 1,000 men, dozens who never would have known they had prostate cancer would be harmed by the consequences of treatment.4
The same pattern is seen with breast cancer. With the introduction of population-based screening with mammography, the incidence of localized breast cancer and ductalcarcinoma in situ increased from 112 to 234 cases per 100,000 women between 1976 and 2008. If screening was effective at improving outcomes and not simply diagnosing more cases, then one would expect to see a decrease in late-stage or metastatic disease over time. However, the reduction in late-stage breast cancer during the same period was much smaller, from 102 to 94 cases per 100,000 women, suggesting that 31% of all breast cancers represent overdiagnosis.5 Using ecologic data collected with the introduction of screening programs in European countries, researchers estimated that approximately one-half of breast cancers represent overdiagnosis.6
Overdiagnosis also occurs with melanoma and thyroid cancers, diseases that have had a sharp increase in incidence without any accompanying decrease in mortality7 (Figure 18 ). This suggests that there is a reservoir of slow-growing cancers with limited malignant potential that are being detected because of increasing scrutiny and increasing numbers of biopsies.9 Autopsy data confirm that subclinical cancers occur at high frequency in thyroid, breast, and prostate cancers.7
Figure 1.
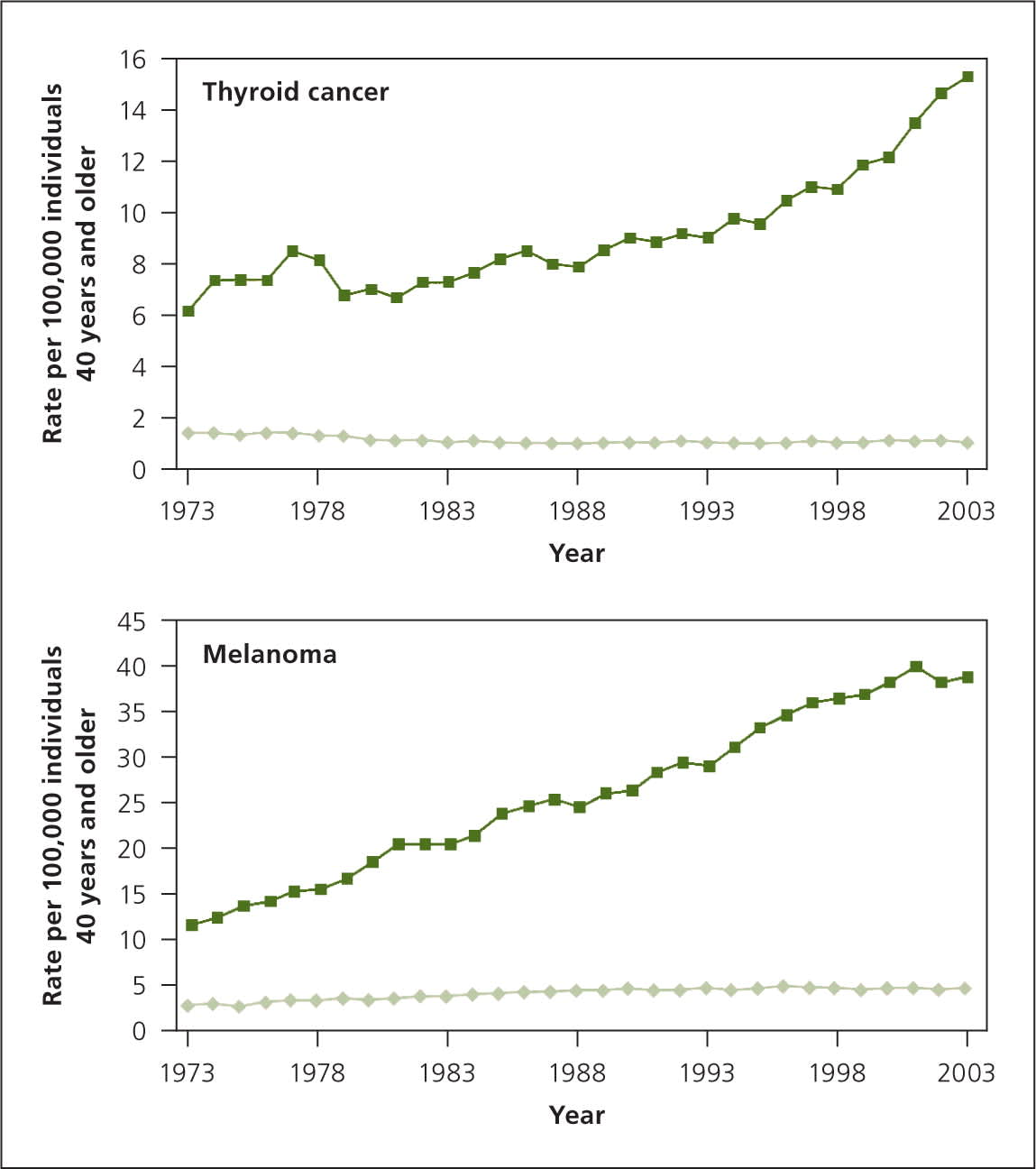
Incidence (green line) and mortality (light green line) for thyroid cancer and melanoma over time, suggesting overdiagnosis of these diseases.
Adapted with permission from Kramer BS, Croswell JM. Cancer screening: the clash of science and intuition. Annu Rev Med. 2009;60:128.
Overdiagnosis is not limited to cancer. Improved technologies that allow us to identify smaller and smaller abnormalities increase the likelihood of overdiagnosis. For example, in the eight years following the introduction of the multidetector computed tomographic scanners in 1998, the incidence of pulmonary embolism increased from 62.1 to 112.3 per 100,000 adults. Unfortunately, mortality from pulmonary embolism stayed virtually the same during that period, declining only slightly from 12.3 to 11.9 per 100,000 adults. That is because we are now detecting and treating increasingly small subsegmental emboli that are unlikely to be lethal, while still exposing these patients to the substantial risks, costs, and inconvenience of anticoagulation.10 Overdiagnosis by itself causes only minor harms, such as anxiety, but it usually leads to overtreatment, which may be more harmful. We will address overtreatment in the next editorial in this series.
Overdiagnosis creates a self-affirming positive cycle, with improvement in some survival statistics that encourages more screening and overdiagnosis. Use of five-year survival as a measure of screening effectiveness is particularly misleading—if you include a lot of patients with overdiagnosed cancers that would never be harmful, obviously the five-year survival would be inflated. However, knowing that 30% of breast cancers are overdiagnosed and would have never been harmful is not helpful to the individual woman who has a diagnosis of early-stage invasive breast cancer. We do not have good tools for many cancers, including breast cancer, to help us distinguish the localized cancers that are likely to progress from those that are indolent and unlikely to harm the patient. Progress is being made in the management of localized prostate cancers, with good evidence that patients with a prostate-specific antigen level of less than 10 ng per mL (10 mcg per L) and those with a low-grade tumor do just as well with watchful waiting.11
A working group from the National Cancer Institute recommends more research to develop molecular diagnostic tests and biomarkers that will help us distinguish over-diagnosed disease from disease requiring treatment.12 Another recommendation from this group is to rethink what we call low-grade and premalignant neoplasms, by removing the word cancer from their names and reclassifying them as “indolent lesions of epithelial origin.”12 It is also important that organizations creating practice guidelines consider overdiagnosis when balancing benefits and harms, as the U.S. Preventive Services Task Force does. Communicating that prolonging survival from the time of diagnosis is not the same as delaying death from that disease is an important message for patients. Finally, shared decision-making tools for clinicians and patients will help patients understand the benefits and harms of different screening and treatment strategies, including watchful waiting, for these indolent lesions of epithelial origin.
editor's note: Dr. Ebell is deputy editor for evidence-based medicine for AFP. Drs. Ebell and Herzstein are members of the USPSTF. This article is their own work and does not necessarily represent the views or policies of the USPSTF.

