
A more recent article on osteoporosis is available.
This is a corrected version of the article that appeared in print.
Am Fam Physician. 2015;92(4):261-268
Author disclosure: No relevant financial affiliations.
Osteoporosis-related fractures affect approximately one in two white women and one in five white men in their lifetime. The impact of fractures includes loss of function, significant costs, and increased mortality. The U.S. Preventive Services Task Force recommends using dual energy x-ray absorptiometry to screen all women 65 years and older, and younger women who have an increased fracture risk as determined by the FRAX Fracture Risk Assessment Tool. [corrected] Although guidelines are lacking for rescreening women who have normal bone mineral density on initial screening, intervals of at least four years appear safe. The U.S. Preventive Services Task Force found insufficient evidence to recommend screening for osteoporosis in men; other organizations recommend screening all men 70 years and older. In patients with newly diagnosed osteoporosis, suggested laboratory tests to identify secondary causes include serum 25-hydroxyvitamin D, calcium, creatinine, and thyroid-stimulating hormone. First-line treatment to prevent fractures consists of fall prevention, smoking cessation, moderation of alcohol intake, and bisphosphonate therapy. Clinicians should consider discontinuing bisphosphonate therapy after five years in women without a personal history of vertebral fractures. Raloxifene, teriparatide, and denosumab are alternative effective treatments for certain subsets of patients and for those who are unable to take or whose condition does not respond to bisphosphonates. The need for follow-up bone mineral density testing in patients receiving treatment for osteoporosis is uncertain.
More than 10 million Americans have osteoporosis, which is defined by the National Osteoporosis Foundation as a chronic, progressive disease characterized by low bone mass, microarchitecture deterioration of bone tissue, bone fragility, and a consequent increase in fracture risk.1 Roughly 50% of white women and 20% of white men have a fracture related to osteoporosis in their lifetime; although black men and women are at lower risk of osteoporosis, those with osteoporosis have similar fracture risk.1 Osteoporotic fractures are associated with increased risk of disability, nursing home placement, total health care costs, and mortality (Table 1).1–3 Osteoporosis risk increases with age, and its impact will increase as the U.S. population ages.3 Table 2 lists risk factors for osteoporosis.2
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| All women 65 years and older should be screened for osteoporosis with dual energy x-ray absorptiometry of the hip and lumbar spine. | B | 5 |
| Women younger than 65 years should be screened for osteoporosis if the estimated 10-year fracture risk equals or exceeds that of a 65-year-old white woman with no risk factors. | B | 1, 5 |
| The U.S. Preventive Services Task Force concludes that the current evidence is insufficient to assess the balance of benefits and harms of screening for osteoporosis in men. | C | 5 |
| A fall risk assessment should be performed and a multicomponent exercise program and smoking cessation should be recommended to decrease fracture risk in individuals 65 years and older with osteoporosis or a history of vertebral fracture. | C | 17, 20, 22 |
| Bisphosphonates should be used as first-line pharmacologic treatment for osteoporosis. | A | 16, 26 |
| In patients who cannot tolerate or whose symptoms do not improve with bisphosphonate therapy, teriparatide (Forteo) and denosumab (Prolia) are effective alternative medications to prevent osteoporotic fractures. | A | 16, 26, 44 |

| Recommendation | Sponsoring organization |
|---|---|
| Do not use dual energy x-ray absorptiometry (DEXA) to screen for osteoporosis in women younger than 65 years or in men younger than 70 years with no risk factors.* | American Academy of Family Physicians |
| Do not routinely repeat dual energy x-ray absorptiometry (DEXA) scans more often than once every two years. | American College of Rheumatology |

| Impact | Statistics |
|---|---|
| Disability (pain, disability, complications) |
|
| Mortality |
|
| Long-term nursing home care |
|
| Annualized health care costs (2002) |
|
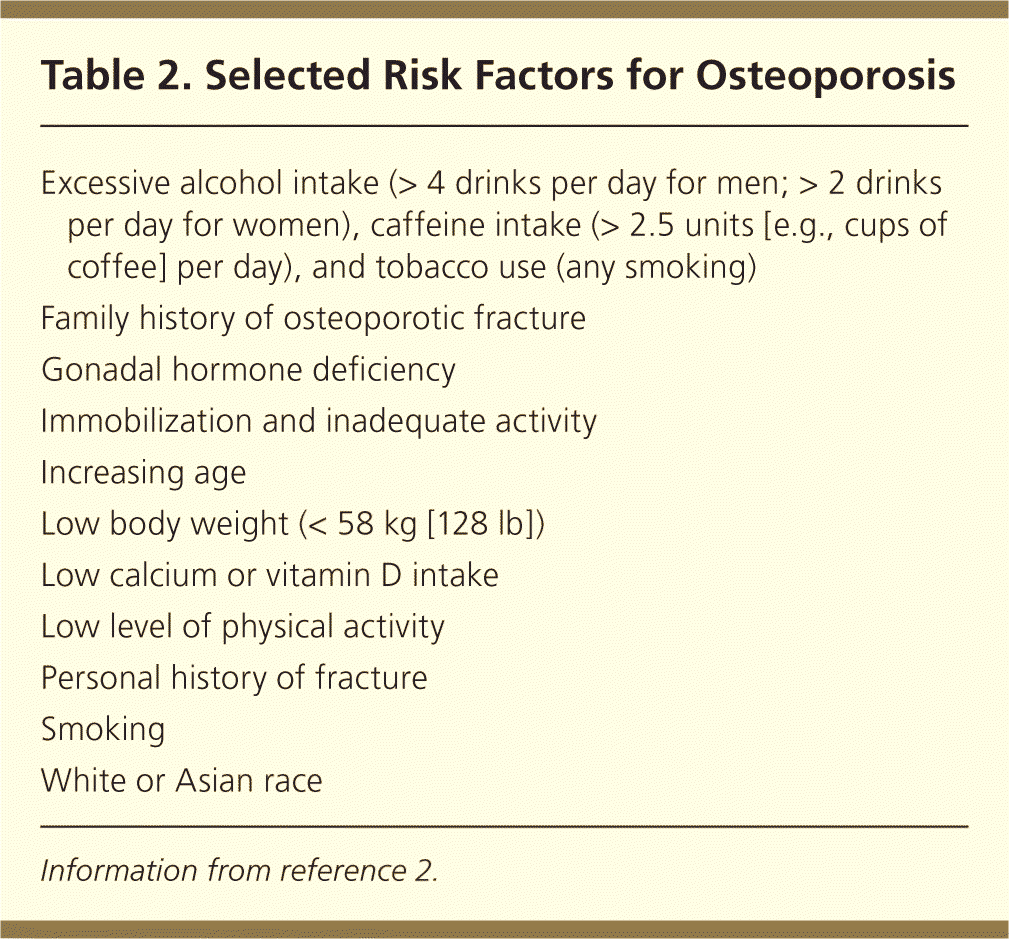
| Excessive alcohol intake (> 4 drinks per day for men; > 2 drinks per day for women), caffeine intake (> 2.5 units [e.g., cups of coffee] per day), and tobacco use (any smoking) |
| Family history of osteoporotic fracture |
| Gonadal hormone deficiency |
| Immobilization and inadequate activity |
| Increasing age |
| Low body weight (< 58 kg [128 lb]) |
| Low calcium or vitamin D intake |
| Low level of physical activity |
| Personal history of fracture |
| Smoking |
| White or Asian race |
Diagnosis
Osteoporosis is diagnosed radiographically based on bone mineral density (BMD) determinations from dual energy x-ray absorptiometry (DEXA) assessment.4 Although quantitative calcaneal ultrasonography and peripheral DEXA can also predict fracture risk, these modalities do not correlate well enough with central DEXA to be used diagnostically.1,5,6 The World Health Organization (WHO) established commonly accepted definitions of osteoporosis and osteopenia4 (Table 36 ).

| Category | Bone mass (BMD derived from DEXA measurement) |
|---|---|
| Normal | Spinal or hip BMD within 1.0 SD below the young adult female reference mean (T-score ≥ –1.0) |
| Low bone mass (osteopenia) | Spinal or hip BMD between 1.0 and 2.5 SDs below the young adult female reference mean (T-score < –1.0 and > –2.5) |
| Osteoporosis | Spinal or hip BMD ≥ 2.5 SDs below the young adult female reference mean (T-score ≤ –2.5) |
| Severe/established osteoporosis | BMD ≥ 2.5 SDs below the young adult female reference mean and the presence of one or more fragility fractures |
The WHO criteria should not be applied to men younger than 50 years, children, or premenopausal women. For these groups, the International Society for Clinical Densitometry recommends use of the z score (age and sex norms). Z scores of −2.0 or less are below the expected range for age. Osteoporosis in men younger than 50 years cannot be diagnosed based on BMD assessment alone.7
Screening
Published osteoporosis screening guidelines vary greatly (eTable A). The U.S. Preventive Services Task Force (USPSTF) recommends screening all women 65 years and older.5 DEXA of the hip and lumbar spine is the preferred assessment method. The USPSTF also advises screening women younger than 65 years whose 10-year fracture risk is greater than or equal to that of a 65-year-old white woman without additional risk factors.5 The FRAX Fracture Risk Assessment Tool (http://www.shef.ac.uk/FRAX/) was used by the USPSTF as a method of determining increased fracture risk for these women. [corrected] Although guidelines for rescreening women with normal initial screening results are lacking, recent evidence suggests that intervals of at least four years appear safe.8,9
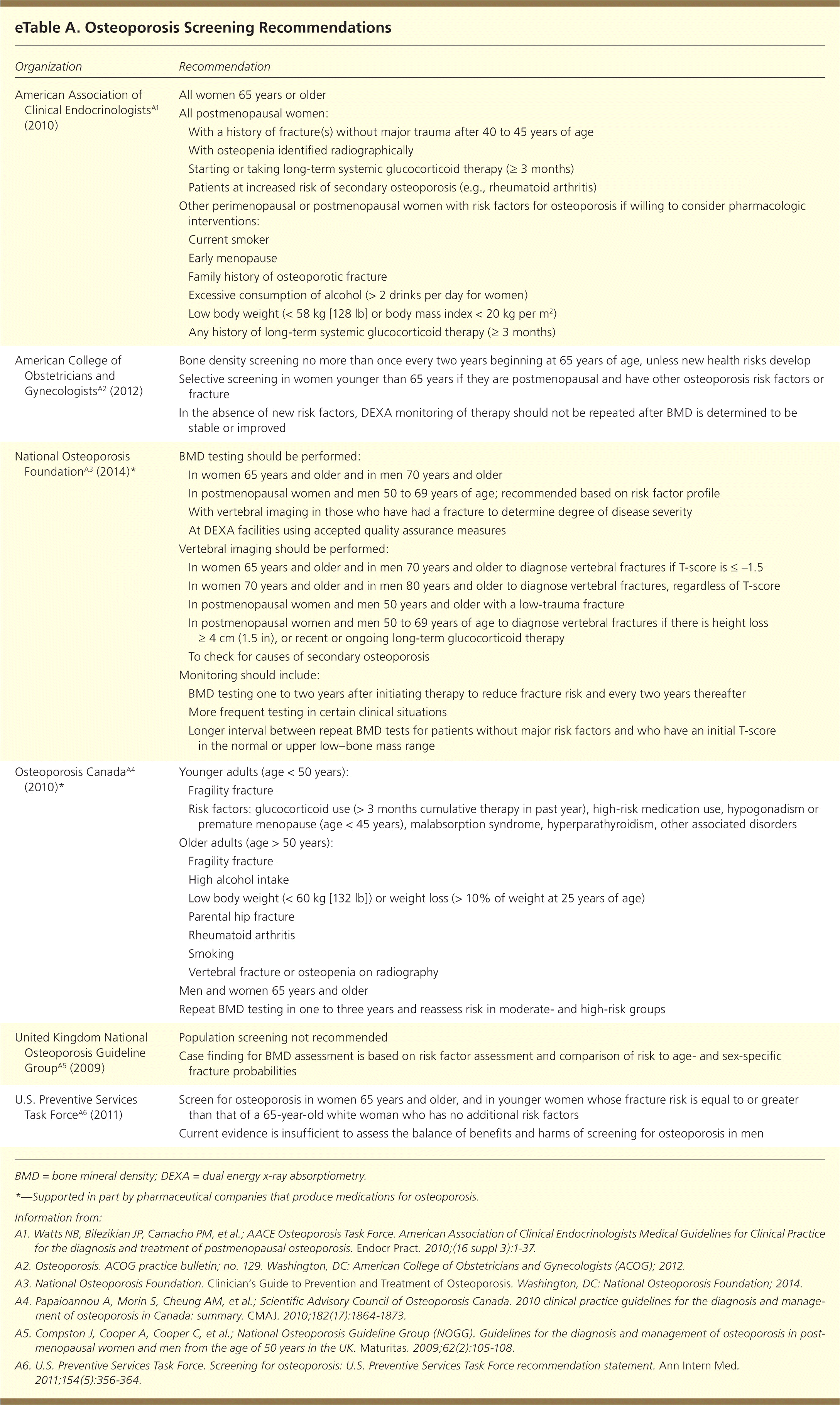
| Organization | Recommendation | |
|---|---|---|
| American Association of Clinical EndocrinologistsA1 (2010) | All women 65 years or older | |
| All postmenopausal women: | ||
| With a history of fracture(s) without major trauma after 40 to 45 years of age | ||
| With osteopenia identified radiographically | ||
| Starting or taking long-term systemic glucocorticoid therapy (≥ 3 months) | ||
| Patients at increased risk of secondary osteoporosis (e.g., rheumatoid arthritis) | ||
| Other perimenopausal or postmenopausal women with risk factors for osteoporosis if willing to consider pharmacologic interventions: | ||
| Current smoker | ||
| Early menopause | ||
| Family history of osteoporotic fracture | ||
| Excessive consumption of alcohol (> 2 drinks per day for women) | ||
| Low body weight (< 58 kg [128 lb] or body mass index < 20 kg per m2) | ||
| Any history of long-term systemic glucocorticoid therapy (≥ 3 months) | ||
| American College of Obstetricians and GynecologistsA2 (2012) | Bone density screening no more than once every two years beginning at 65 years of age, unless new health risks develop | |
| Selective screening in women younger than 65 years if they are postmenopausal and have other osteoporosis risk factors or fracture | ||
| In the absence of new risk factors, DEXA monitoring of therapy should not be repeated after BMD is determined to be stable or improved | ||
| National Osteoporosis FoundationA3 (2014)* | BMD testing should be performed: | |
| In women 65 years and older and in men 70 years and older | ||
| In postmenopausal women and men 50 to 69 years of age; recommended based on risk factor profile | ||
| With vertebral imaging in those who have had a fracture to determine degree of disease severity | ||
| At DEXA facilities using accepted quality assurance measures | ||
| Vertebral imaging should be performed: | ||
| In women 65 years and older and in men 70 years and older to diagnose vertebral fractures if T-score is ≤ –1.5 | ||
| In women 70 years and older and in men 80 years and older to diagnose vertebral fractures, regardless of T-score | ||
| In postmenopausal women and men 50 years and older with a low-trauma fracture | ||
| In postmenopausal women and men 50 to 69 years of age to diagnose vertebral fractures if there is height loss ≥ 4 cm (1.5 in), or recent or ongoing long-term glucocorticoid therapy | ||
| To check for causes of secondary osteoporosis | ||
| Monitoring should include: | ||
| BMD testing one to two years after initiating therapy to reduce fracture risk and every two years thereafter | ||
| More frequent testing in certain clinical situations | ||
| Longer interval between repeat BMD tests for patients without major risk factors and who have an initial T-score in the normal or upper low–bone mass range | ||
| Osteoporosis CanadaA4 (2010)* | Younger adults (age < 50 years): | |
| Fragility fracture | ||
| Risk factors: glucocorticoid use (> 3 months cumulative therapy in past year), high-risk medication use, hypogonadism or premature menopause (age < 45 years), malabsorption syndrome, hyperparathyroidism, other associated disorders | ||
| Older adults (age > 50 years): | ||
| Fragility fracture | ||
| High alcohol intake | ||
| Low body weight (< 60 kg [132 lb]) or weight loss (> 10% of weight at 25 years of age) | ||
| Parental hip fracture | ||
| Rheumatoid arthritis | ||
| Smoking | ||
| Vertebral fracture or osteopenia on radiography | ||
| Men and women 65 years and older | ||
| Repeat BMD testing in one to three years and reassess risk in moderate- and high-risk groups | ||
| United Kingdom National Osteoporosis Guideline GroupA5 (2009) | Population screening not recommended | |
| Case finding for BMD assessment is based on risk factor assessment and comparison of risk to age- and sex-specific fracture probabilities | ||
| U.S. Preventive Services Task ForceA6 (2011) | Screen for osteoporosis in women 65 years and older, and in younger women whose fracture risk is equal to or greater than that of a 65-year-old white woman who has no additional risk factors | |
| Current evidence is insufficient to assess the balance of benefits and harms of screening for osteoporosis in men | ||
The USPSTF found insufficient evidence to recommend routine screening for osteoporosis in men.5 Men with a minimal trauma fracture who are older than 50 years or those with secondary causes associated with bone loss could be considered for screening. The National Osteoporosis Foundation also recommends screening all men 70 years and older, based on the assumption that this group has a similar osteoporotic fracture risk and treatment effectiveness as 65-year-old white women.1
Evaluation for Secondary Osteoporosis
Primary osteoporosis is related to aging and loss of gonadal function. Secondary osteoporosis is caused by other health conditions (Table 4).2 Up to 30% of osteoporosis cases in postmenopausal women are estimated to be from a secondary cause.10 The estimate climbs to greater than 50% in men, premenopausal women, and perimenopausal women if vitamin D deficiency is included as a secondary cause.11–13 In addition to performing a history and physical examination, expert consensus suggests a basic laboratory evaluation for all newly diagnosed patients to determine if there are contraindications for certain osteoporosis medications and to identify the more common secondary causes. The most commonly recommended laboratory tests include serum 25-hydroxyvitamin D, calcium, creatinine, and thyroid-stimulating hormone levels.1,14
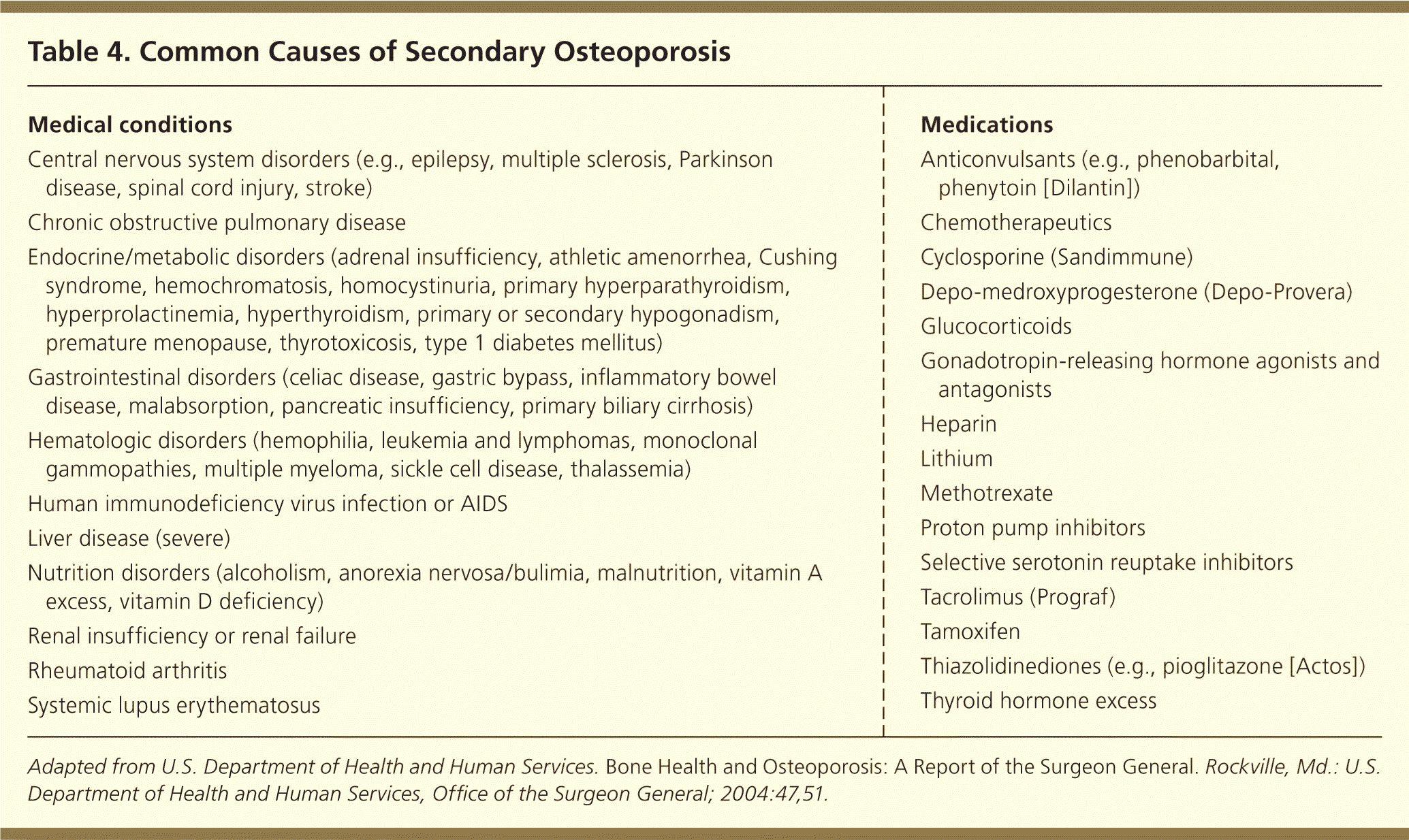
| Medical conditions |
| Central nervous system disorders (e.g., epilepsy, multiple sclerosis, Parkinson disease, spinal cord injury, stroke) |
| Chronic obstructive pulmonary disease |
| Endocrine/metabolic disorders (adrenal insufficiency, athletic amenorrhea, Cushing syndrome, hemochromatosis, homocystinuria, primary hyperparathyroidism, hyperprolactinemia, hyperthyroidism, primary or secondary hypogonadism, premature menopause, thyrotoxicosis, type 1 diabetes mellitus) |
| Gastrointestinal disorders (celiac disease, gastric bypass, inflammatory bowel disease, malabsorption, pancreatic insufficiency, primary biliary cirrhosis) |
| Hematologic disorders (hemophilia, leukemia and lymphomas, monoclonal gammopathies, multiple myeloma, sickle cell disease, thalassemia) |
| Human immunodeficiency virus infection or AIDS |
| Liver disease (severe) |
| Nutrition disorders (alcoholism, anorexia nervosa/bulimia, malnutrition, vitamin A excess, vitamin D deficiency) |
| Renal insufficiency or renal failure |
| Rheumatoid arthritis |
| Systemic lupus erythematosus |
| Medications |
| Anticonvulsants (e.g., phenobarbital, phenytoin [Dilantin]) |
| Chemotherapeutics |
| Cyclosporine (Sandimmune) |
| Depo-medroxyprogesterone (Depo-Provera) |
| Glucocorticoids |
| Gonadotropin-releasing hormone agonists and antagonists |
| Heparin |
| Lithium |
| Methotrexate |
| Proton pump inhibitors |
| Selective serotonin reuptake inhibitors |
| Tacrolimus (Prograf) |
| Tamoxifen |
| Thiazolidinediones (e.g., pioglitazone [Actos]) |
| Thyroid hormone excess |
Treatment
The National Osteoporosis Foundation recommends treatment of postmenopausal women and men with a personal history of hip or vertebral fracture, a T-score of −2.5 or less, or a combination of low bone mass (T-score between −1 and −2.5) and a 10-year probability of hip fracture of at least 3% or any major fracture of at least 20% as calculated by the FRAX Fracture Risk Assessment Tool.1 [corrected] The WHO recommendations are less specific, stating that persons with or at risk of osteoporosis should be considered for treatment.15 Randomized controlled trials of treatment have shown reduction of fractures for only two groups: those with a T-score of less than –2.5 and those who have already experienced a hip or vertebral fracture.16
NONPHARMACOLOGIC TREATMENT
Fall prevention is a priority for patients with osteoporosis because falls are more closely associated with fracture risk than is BMD.17 The USPSTF recommends exercise or physical therapy and vitamin D supplementation to prevent falls in community-dwelling adults 65 years or older who are at increased risk of falls.18 A multicomponent exercise program that consists of weight-bearing resistance and balance training should be recommended. Aerobic exercise programs that do not incorporate strength and balance training should be avoided because of the association with increased fracture risk.19 A thorough assessment of a patient's risks of falling and mitigation of those risk factors have strong evidence of effectiveness in fall prevention.20 A Cochrane review suggested that hip protectors decrease fracture risk.21
Dietary modifications may have a role in optimizing bone health. Consuming more than 2.5 units of caffeine daily (1 unit = one cup of coffee or two cups of tea) may increase fracture risk.24 Diets with adequate protein intake are necessary for optimal bone health, but the proper amount or source (plant vs. animal) remains controversial. A balanced diet consisting of vitamin D, calcium, protein, vegetables, and fruits is recommended; mononutrient dietary supplementation is unlikely to be helpful.24 Table 5 shows a comparison of nonpharmacologic therapies.17–25

| Intervention | Comments |
|---|---|
| Alcohol moderation | ≤ 4 drinks per day for men or ≤ 2 drinks per day for women |
| Decreased caffeine intake | ≤ 2.5 cups of coffee or ≤ 5 cups of tea per day |
| Multicomponent exercise with strength and balance training | — |
| Multifactorial falls risk assessment | — |
| Smoking cessation | — |
| Sunlight/ultraviolet exposure | 30 minutes per day, 5 days per week |
| Vitamin D supplementation | 800 IU per day |
PHARMACOLOGIC TREATMENT
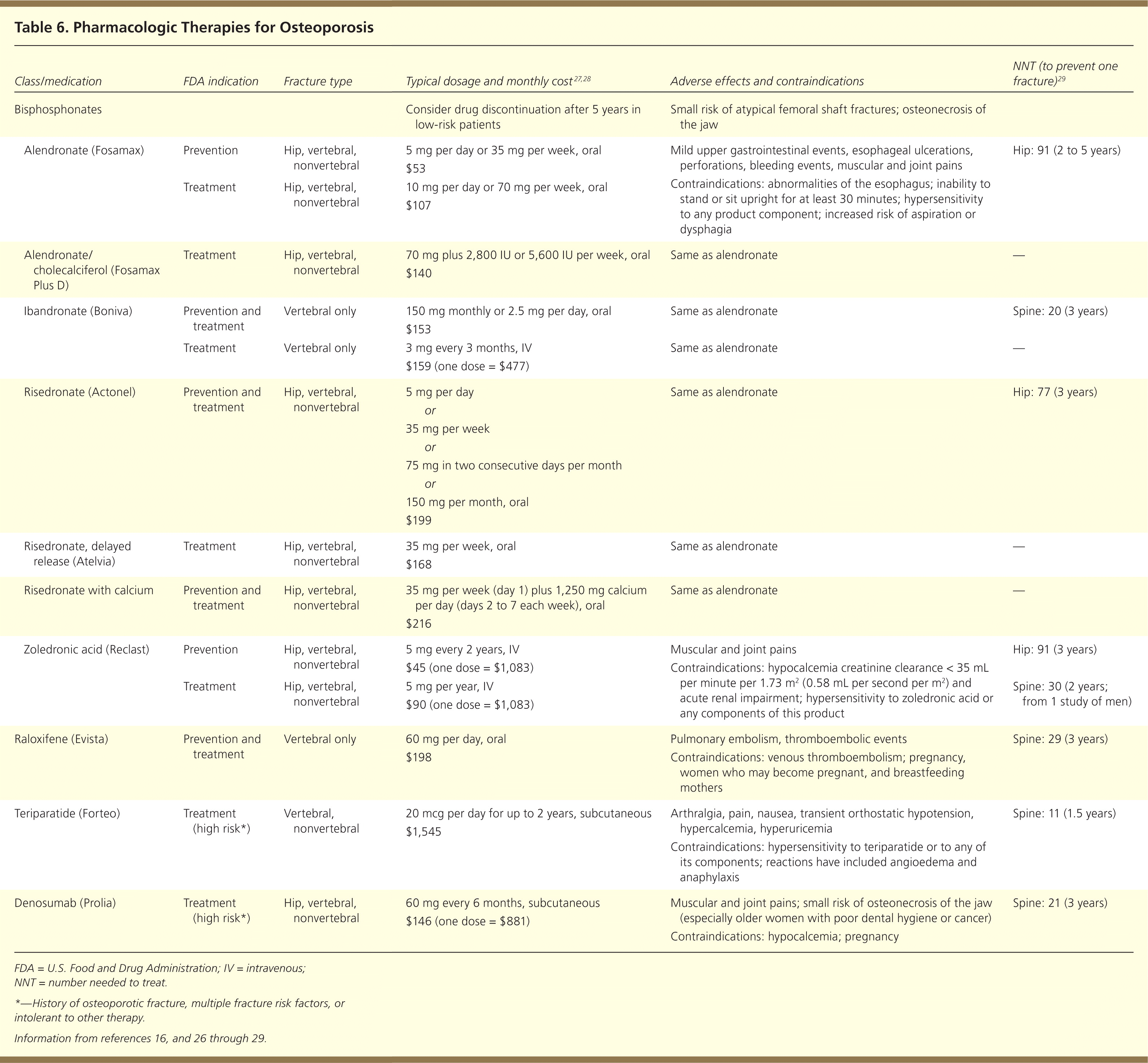
| Class/medication | FDA indication | Fracture type | Typical dosage and monthly cost27,28 | Adverse effects and contraindications | NNT (to prevent one fracture)29 | ||
|---|---|---|---|---|---|---|---|
| Bisphosphonates | Consider drug discontinuation after 5 years in low-risk patients | Small risk of atypical femoral shaft fractures; osteonecrosis of the jaw | |||||
| Alendronate (Fosamax) | Prevention | Hip, vertebral, nonvertebral |
|
| Hip: 91 (2 to 5 years) | ||
| Treatment | Hip, vertebral, nonvertebral |
| |||||
| Alendronate/cholecalciferol (Fosamax Plus D) | Treatment | Hip, vertebral, nonvertebral |
| Same as alendronate | — | ||
| Ibandronate (Boniva) | Prevention and treatment | Vertebral only |
| Same as alendronate | Spine: 20 (3 years) | ||
| Treatment | Vertebral only |
| Same as alendronate | — | |||
| Risedronate (Actonel) | Prevention and treatment | Hip, vertebral, nonvertebral |
| Same as alendronate | Hip: 77 (3 years) | ||
| Risedronate, delayed release (Atelvia) | Treatment | Hip, vertebral, nonvertebral |
| Same as alendronate | — | ||
| Risedronate with calcium | Prevention and treatment | Hip, vertebral, nonvertebral |
| Same as alendronate | — | ||
| Zoledronic acid (Reclast) | Prevention | Hip, vertebral, nonvertebral |
|
|
| ||
| Treatment | Hip, vertebral, nonvertebral |
| |||||
| Raloxifene (Evista) | Prevention and treatment | Vertebral only |
|
| Spine: 29 (3 years) | ||
| Teriparatide (Forteo) | Treatment (high risk*) | Vertebral, nonvertebral |
|
| Spine: 11 (1.5 years) | ||
| Denosumab (Prolia) | Treatment (high risk*) | Hip, vertebral, nonvertebral |
| Muscular and joint pains; small risk of osteonecrosis of the jaw (especially older women with poor dental hygiene or cancer) Contraindications: hypocalcemia; pregnancy | Spine: 21 (3 years) | ||
Bisphosphonates. Oral bisphosphonates inhibit osteoclastic activity and are antiresorptive agents. They are considered first-line pharmacologic therapy. Randomized clinical trials demonstrate a reduction of vertebral and hip fractures with alendronate (Fosamax) and risedronate (Actonel).16,26 Alendronate and risedronate also decrease vertebral fractures in men30,31 and in patients with glucocorticoid-induced osteoporosis.32,33 Daily and intermittent use of ibandronate (Boniva) have demonstrated effectiveness in reducing fractures of the spine only.34 Weekly and monthly dosing formulations improve adherence.35 Oral bisphosphonates should be taken only with water and a wait of at least 30 minutes before reclining or ingesting other medication or food. This decreases upper gastrointestinal adverse effects and allows for appropriate absorption.
The intravenous bisphosphonates approved by the U.S. Food and Drug Administration for the treatment of postmenopausal osteoporosis are zoledronic acid (Reclast), 5 mg yearly (shown to decrease vertebral and hip fractures),16,26,36 and ibandronate, 3 mg every three months.37 Although these medications are expensive, they are useful for high-risk patients who are unable to tolerate or adhere to oral therapy.
The optimal length of oral bisphosphonate therapy is unknown. One study found that women who take alendronate for five years followed by five years of placebo have no increased incidence of nonvertebral or hip fractures compared with women who take alendronate for 10 years. There is, however, an increase in vertebral fractures.38 Osteonecrosis of the jaw and atypical femoral fractures are rare complications of bisphosphonate therapy that are associated with longer duration of use.39,40 Clinicians should consider discontinuing bisphosphonate therapy after five years in women without a personal history of vertebral fractures.
Raloxifene. Raloxifene, a selective estrogen receptor modulator, is approved for treating postmenopausal osteoporosis, and is effective at reducing vertebral fractures only.16,26 Raloxifene is commonly associated with increased vasomotor symptoms. It is associated with an increased risk of venous thromboembolism and a decreased risk of invasive breast cancer.16 The best candidates for raloxifene are postmenopausal women with osteoporosis who are unable to tolerate bisphosphonates, have no vasomotor symptoms or history of venous thromboembolism, and have a high breast cancer risk score.16,27 Bazedoxifene is a selective estrogen receptor modulator more recently approved for use in the United States for the prevention of osteoporosis as part of a combination therapy with conjugated estrogen (Duavee).
Calcitonin. Calcitonin nasal spray is an antiresorptive agent approved for the treatment of postmenopausal osteoporosis. It has been shown to decrease the occurrence of vertebral compression fractures only.16,26 Although calcitonin has modest analgesic properties in the setting of acute and chronic vertebral compression fracture, it is not considered first-line treatment for osteoporosis because more effective medications are available.16,41 There have also been reports of increased cancer rates associated with use of calcitonin.42
Teriparatide. Teriparatide is a recombinant human parathyroid hormone with bone anabolic activity. In a dosage of 20 mcg per day given subcutaneously for up to two years, teriparatide decreases vertebral and nonvertebral fractures.16,26 Teriparatide is approved for the treatment of postmenopausal women with severe bone loss, men with osteoporosis who have high risk of fracture, and individuals whose condition has not improved with bisphosphonate therapy. One study suggests that it is advisable to follow teriparatide therapy with bisphosphonate therapy to maintain BMD gains.43
Denosumab. Denosumab is a human monoclonal antibody that inhibits the formation and activity of osteoclasts by blocking receptor activator of nuclear factor kappa B ligand. In a dose of 60 mg given subcutaneously every six months for three years, it significantly increased BMD in postmenopausal women compared with weekly dosing of alendronate.44 Denosumab has been shown to decrease hip, vertebral, and nonvertebral fractures compared with low doses of calcium and vitamin D. It appears to be a reasonable alternative for persons whose condition does not improve with bisphosphonates. Renal insufficiency is a listed caution, but denosumab appears to be safe for patients with chronic kidney disease stages 1 to 3.45
Hormone Therapy. The Women's Health Initiative study confirmed that estrogen, with or without progesterone, slightly reduced the risk of hip and vertebral fractures; however, this benefit did not outweigh the increased risk of stroke, venous thromboembolism, coronary heart disease, and breast cancer, even for women at high risk of fracture.46 Lower doses of conjugated equine estrogens and estradiol have been shown to improve BMD, but a reduced risk of fracture has not been demonstrated and the safety is unknown.47
Combination Therapy. There has been no demonstrated effectiveness of combination therapy in reducing fractures. Although research continues, there is currently a limited role for combination therapy beyond clinical trials.
FOLLOW-UP
After initiation of treatment, the need for follow-up bone density testing is uncertain. A decrease in BMD could suggest treatment nonadherence, inadequate calcium or vitamin D intake, an unidentified secondary cause of osteoporosis, or treatment failure.48 However, a single-institution study found that although follow-up DEXA scanning for patients with osteoporosis was performed often, this rarely led to changes in treatment, even in patients found to have decreased BMD.49
Data Sources: We reviewed all cited references from the original 2009 review article, then performed a PubMed search using the following key words: osteoporosis, osteopenia, screening, diagnosis, treatment, prevention, secondary, and vitamin D. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Additional searches included Essential Evidence Plus, the U.S. Preventive Services Task Force, the Institute for Clinical Systems Improvement, the National Guideline Clearinghouse, the Cochrane Database of Systematic Reviews, and the National Osteoporosis Foundation website. Search dates: April and July 2014, and May 2015.
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of Defense, the U.S. Army Medical Corps, or the U.S. Army at large.
note: This review updates a previous article on this topic by Sweet, Sweet, Jeremiah, and Galazka.29
