
A more recent article on prostate cancer screening is available.
Am Fam Physician. 2015;92(8):683-688
Related editorial: Prostate Cancer Screening: The Pendulum Has Swung, and the Burden of Proof Is with Proponents
Related Medicine by the Numbers: PSA Screening for Prostate Cancer
Related letters: Current Prostate Cancer Screening Guidelines May Lead to More Deaths from the Disease and Hyperbaric Oxygen Therapy for Radiation Cystitis
Author disclosure: No relevant financial affiliations.
Among American men, prostate cancer is the most commonly diagnosed cancer and the second leading cause of cancer-related death. Although prostate-specific antigen (PSA) testing has been used to screen for prostate cancer for more than 25 years, the test has low sensitivity and specificity, and there is no clear evidence for determining what threshold warrants prostate biopsy. Only one of five randomized controlled trials of PSA screening showed an effect on prostate cancer–specific mortality, and the absolute reduction in deaths from prostate cancer was one per 781 men screened after 13 years of follow-up. None of the trials showed benefit in all-cause mortality, and screening increased prostate cancer diagnoses by about 60%. Harms of screening include adverse effects from prostate biopsy, overdiagnosis and overtreatment, and anxiety. One-half of screen-detected prostate cancers will not cause symptoms in the patient's lifetime, and 80% to 85% of men who choose observation will not die from prostate cancer within 15 years. Adverse effects of radical prostatectomy include perioperative complications, erectile dysfunction, and urinary incontinence. Radiation therapy can cause acute toxicity leading to urinary urgency, dysuria, diarrhea, and rectal pain; late toxicity includes erectile dysfunction, rectal bleeding, and urethral stricture. Despite variations across guidelines, no organization recommends routine PSA testing, and all endorse some form of shared decision-making before testing. If screening is performed, it should generally be discontinued at 70 years of age.
In 2014, about 233,000 American men were diagnosed with prostate cancer, and almost 33,000 died from it.1 Approximately one in six men (16.7%) will be diagnosed with prostate cancer during his lifetime, but less than 3% will die from the disease. Prostate cancer mainly affects older men; 60% of cases are diagnosed after 65 years of age, and 70% of men who die from prostate cancer are 75 years or older.2
| Clinical recommendations | Evidence rating | References |
|---|---|---|
| The U.S. Preventive Services Task Force and American Academy of Family Physicians recommend against prostate-specific antigen testing to screen for prostate cancer because the harms outweigh the benefits in most men. | B | 31, 32 |
| Physicians should inform patients about the harms and benefits of prostate-specific antigen testing and use shared decision making. Only men who express a clear preference for screening should be tested. | C | 10, 36, 37 |
| Prostate cancer screening should not be performed in men younger than 50 years or older than 70 years, or in men with a life expectancy of less than 10 to 15 years. | C | 10, 31, 32, 36, 37 |

| Recommendation | Sponsoring organization |
|---|---|
| Do not routinely screen for prostate cancer using a PSA test or digital rectal examination. | American Academy of Family Physicians |
| Do not routinely perform PSA-based screening for prostate cancer. | American College of Preventive Medicine |
| Do not recommend screening for prostate cancer (with the PSA test) without considering life expectancy and the risks of testing, overdiagnosis, and overtreatment. | American Geriatrics Society |
| Do not perform PSA testing for prostate cancer screening in men with no symptoms of the disease when they are expected to live less than 10 years. | American Society of Clinical Oncology |
| Offer PSA screening for prostate cancer only after engaging in shared decision making. | American Urological Association |
Because prostate cancer is the most commonly diagnosed cancer and the second leading cause of cancer-related death in American men, a large amount of effort and resources has been devoted to screening with prostate-specific antigen (PSA) testing and digital rectal examinations (DRE). PSA-based screening is controversial. Three small, low-quality randomized controlled trials (RCTs) did not find a mortality benefit.3–5 Two large European and American RCTs that had conflicting results have been used by medical societies to support current recommendations regarding prostate cancer screening.6,7
Prostate-Specific Antigen
Although PSA testing has been used to screen for prostate cancer since 1987, there is no consensus on which threshold should warrant a prostate biopsy. The most commonly used threshold of more than 4.0 ng per mL (4.0 μg per L) has an approximately 70% false-positive rate. Lowering the threshold to more than 2.5 ng per mL (2.5 μg per L) increases the false-positive rate to 80%.8 Using a biopsy threshold of more than 4.0 ng per mL, the PSA test has an overall sensitivity of 72%, specificity of 93%, and positive predictive value of 25%.9 As many as 15% of men with a PSA level less than 4.0 ng per mL will have prostate cancer on biopsy, and 15% of those cancers are high grade.10 Benign prostatic processes, such as hypertrophy or infection, can also cause PSA elevations.
Many strategies have been suggested to improve the diagnostic performance of the PSA test, such as free PSA, PSA velocity, and PSA density. However, none of these strategies has been evaluated in an RCT.
Digital Rectal Examination
There are limited data on the value of DRE alone or in combination with PSA testing. Some RCTs evaluated DRE with PSA, but none evaluated DRE alone. Even in patients with elevated PSA levels, DRE did not influence the chance of detecting prostate cancer after eight years of follow-up.11 Harms from DRE include discomfort and rectal bleeding.10
Benefits of Early Detection and Treatment
Potential benefits of early detection of prostate cancer include decreased prostate cancer–specific mortality and metastatic disease and an increased chance of finding localized disease.8 Possible benefits have to be balanced with the lack of reduction in overall mortality and the significant harms from overdiagnosis and overtreatment.
The European Randomised Study of Screening for Prostate Cancer (ERSPC), including more than 160,000 men 55 to 69 years of age from seven European countries, is the only screening trial to find a reduction in prostate cancer–specific mortality after 13 years of follow-up. The absolute reduction in prostate cancer–specific mortality was 1.28 deaths per 1,000 men (number needed to screen = 781). To prevent one death from prostate cancer, 27 additional prostate cancers would need to be diagnosed and treated.6,8
The Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial (PLCO), including 76,693 men 55 to 74 years of age from the United States, found no difference in prostate cancer–specific mortality in all participants or within subgroups (defined by age, comorbidity, or screening before entering the trial) after 13 years of follow-up.7 The main limitation of the PLCO is that 52% of the control group had at least one PSA test; however, significantly more prostate cancers were still detected in the group assigned to annual screening.12
A Cochrane review of five RCTs concluded that PSA screening does not decrease prostate cancer–specific or overall mortality. Screening increased the diagnosis of localized prostate cancer and decreased the diagnosis of metastatic prostate cancer.13
The Prostate Cancer Intervention Versus Observation Trial (PIVOT), the only RCT of surgical treatment for localized prostate cancer conducted in the PSA screening era, found that surgery did not decrease prostate cancer–specific or overall mortality compared with observation. Posttrial subgroup analysis suggested an overall mortality benefit from surgery in men with a PSA level of more than 10 ng per mL (10 μg per L) at the time of cancer diagnosis. Surgical treatment also decreased the incidence of bony metastases.14
Harms of Screening and Subsequent Testing
Prostate cancer screening is associated with high rates of overdiagnosis and overtreatment, which lead to psychological and physical harms. Screening for prostate cancer can result in an increase in prostate cancer diagnoses of up to 63%.15
One-third of men who undergo prostate biopsy require physician follow-up for problems related to the procedure, which patients have classified as moderate to severe.18,19 These include pain and fever (3% to 5%), hematuria (22%), hematospermia (27% to 50%), and hospitalization (3%).18,20 Biopsy misses prostate cancer in 10% of cases, and repeat biopsy is needed in up to 31% of men who have an initial negative biopsy result.21,22
Most hospitalizations after prostate biopsy are related to infections, such as urosepsis or prostatitis. Although the overall rate for hospital admission after a prostate biopsy is 3%, one study found that the rate of hospitalization for Medicare patients is nearly 7%.23 Biopsy-related mortality is less than 1%, and most deaths within 30 days of PSA screening and associated procedures are because of comorbid conditions.
Harms of Treating Cancers Detected by Screening
One-half of screening-detected prostate cancers will not cause symptoms in the patient's lifetime, and 80% to 85% of men who choose observation will not die from prostate cancer within 15 years.14 In men who have low-risk disease, defined as a PSA level of less than 10 ng per mL, stage T1 or T2a cancer, and a Gleason score of less than 6, the 15-year prostate cancer mortality rate is less than 5%.14,24,25 Nonetheless, in the United States, approximately 90% of men diagnosed with prostate cancer decide to undergo curative treatment.26,27
Treatments aimed at curing the cancer include radical prostatectomy, external beam radiotherapy, and brachytherapy. Short-term adverse effects of radical prostatectomy include perioperative bleeding that may require blood transfusion, which occurs in up to 20% of patients. Complications arise within 30 days of radical prostatectomy in 22% of patients.28 The main long-term complications of prostatectomy are urinary incontinence (20% of patients) and erectile dysfunction (up to 50% of patients).14,25,29
If external beam radiotherapy or brachytherapy is used, acute toxicity can present in up to 50% of patients as urinary urgency, dysuria, diarrhea, bleeding, or rectal pain. Late toxicity includes complications that present after six months of ending treatment. These complications include erectile dysfunction (up to 50% of patients) and less common problems such as frequent bowel movement, rectal bleeding, and urethral stricture.30 Serious complications associated with prostate cancer treatment include cardiovascular events (2 out of 1,000 patients), deep venous thrombosis and pulmonary embolism (1 out of 1,000), and death (less than 1 out of 1,000).31
Screening Recommendations
The U.S. Preventive Services Task Force (USPSTF)31 and the American Academy of Family Physicians32 recommend against PSA-based screening for prostate cancer. The USPSTF concludes that the harms of screening and unnecessary treatment outweigh the limited benefits.31 One year after the draft recommendation from the USPSTF, there was a 28% drop in the diagnosis of low-, intermediate-, and high-risk prostate cancer in the United States without a change in the diagnosis of nonlocalized prostate cancer.33 The American Academy of Family Physicians and the American College of Preventive Medicine include routine prostate cancer screening using the PSA test or DRE on their Choosing Wisely lists of tests physicians and patients should question.34,35
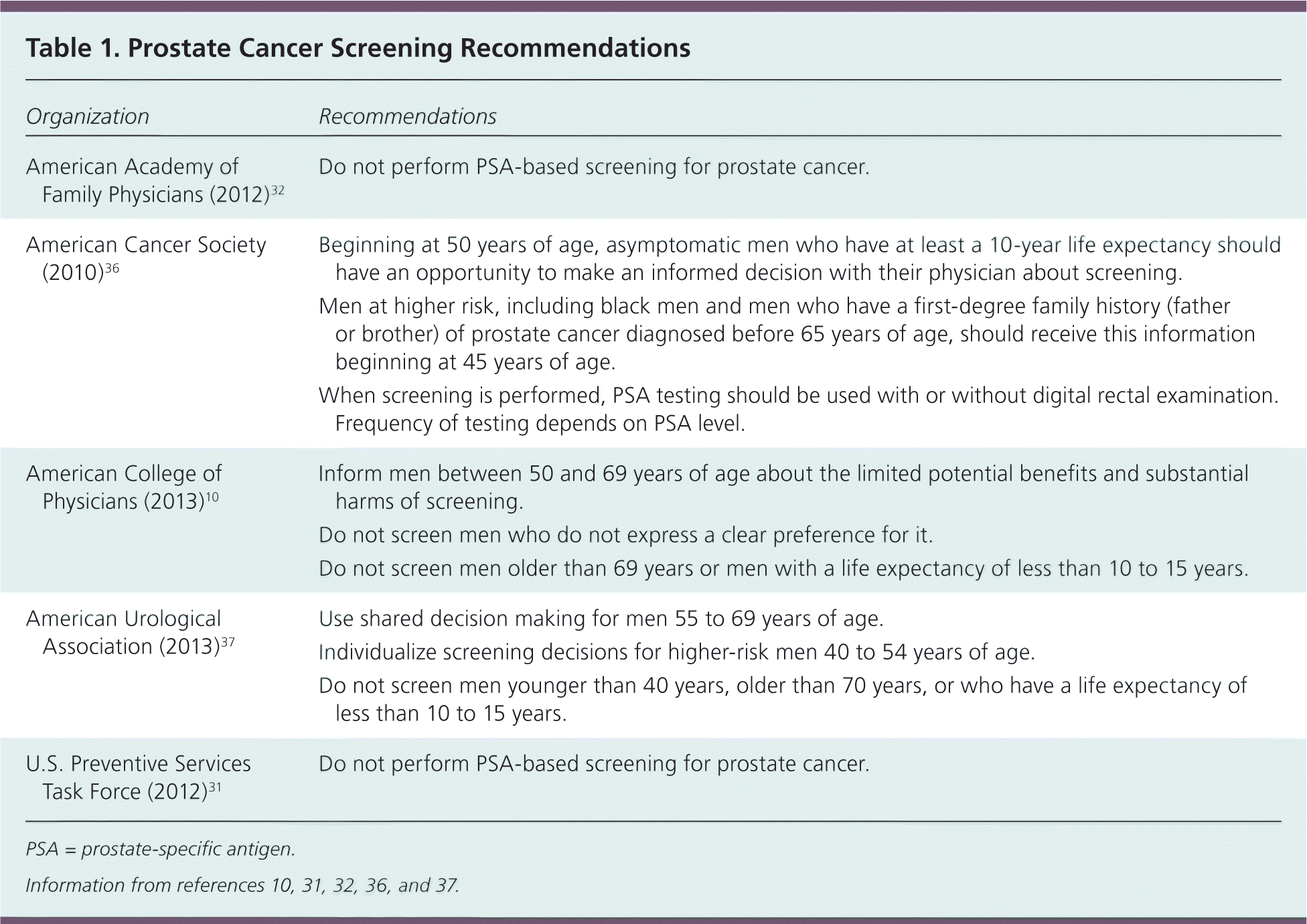
The American College of Physicians, American Cancer Society, and American Urological Association recommend discussing the benefits and harms of screening every one or two years with appropriate men and using a shared decision-making approach.10,36,37 The American College of Physicians recommends screening only men who express a clear preference for it.10 The American Urological Association argues that although the probability that an individual will avoid death from prostate cancer by undergoing PSA screening is low, this small effect could be very meaningful at the population level.37 Proponents of screening also argue that the mortality benefit from PSA screening shown in the ERSPC trial would probably continue to improve after longer follow-up.38 Strategies that could improve the net benefit of PSA screening include offering the PSA every other year rather than annually and using a threshold higher than 4 ng per mL for prostate biopsy.39 Specific screening recommendations from varying organizations are summarized in Table 1.10,31,32,36,37
| Organization | Recommendations |
|---|---|
|
|
|
|
|
|
|
|
|
|
In 2012, about 37% of American men older than 50 years were screened with a PSA test. Although all major guidelines recommend against screening men older than 70 years, the screening rate was more than 45% in men 70 to 79 years of age, and more than 35% in men older than 80 years. Additionally, there was a marked geographic variation in self-reported screening rates, from nearly 60% of men in Hawaii to less than 25% of men in New Hampshire.40
Shared Decision Making
Factors that physicians should discuss as part of shared decision making include the limitations of the PSA test, mortality benefit in absolute terms, options after an abnormal PSA test result, harms of screening, life expectancy, comorbid conditions, family history of prostate cancer, and the patient's goals and wishes. Table 2 provides a list of key discussion points about prostate cancer screening.6,10

| Prostate cancer screening with the PSA test is optional because the chance of harm from screening is greater than the chance of benefit for most men. |
| The potential benefit of prostate cancer screening corresponds to preventing, at most, one death caused by prostate cancer per 1,000 men screened, although 37 men would be diagnosed with prostate cancer unnecessarily, after 11 years of follow-up.6 |
| Harms of screening include anxiety related to abnormal PSA test results or a cancer diagnosis. Prostate biopsy can cause pain and bleeding, and there is a small risk of hospitalization. Treatment of prostate cancer is associated with a small risk of surgery-related death, erectile dysfunction, urinary incontinence, and other complications. |
| Men who choose to have a PSA test increase their chances of a prostate cancer diagnosis, and most prostate cancers are slow growing and do not cause death. |
| The PSA test does not distinguish between aggressive cancer and slow-growing cancer. PSA levels can be elevated because of benign conditions, such as prostate enlargement and infection. Also, 15% of patients with prostate cancer have a normal PSA level.10 |
Because shared decision making is difficult to achieve in a short office visit,41 patient decision aids can be used during the discussion or provided to patients before the office visit. A Cochrane review found that using these decision aids resulted in a 13% decrease in PSA screening.42 A decision aid from the American Cancer Society is available at http://www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-024618.pdf.
Data Sources: PubMed and Medline searches were completed using the key terms prostate cancer, prostate specific antigen, treatment, and screening. The searches included meta-analyses, randomized controlled trials, clinical trials, and reviews. We also searched the Cochrane database, Essential Evidence Plus, the National Guideline Clearinghouse database, and DynaMed. Search dates: April 7, 2014; August 30, 2014; and July 22, 2015.
The authors thank Dr. Paul Misch for his assistance in editing the manuscript.
