
Am Fam Physician. 2016;93(4):307-314
Author disclosure: No relevant financial affiliations.
Conjugated estrogens/bazedoxifene (Duavee) combines conjugated estrogen with bazedoxifene, a selective estrogen receptor modulator. Bazedoxifene stimulates estrogen receptors in bone and has antagonistic effects in the breast and uterus.1 Conjugated estrogens/bazedoxifene is labeled for the treatment of moderate to severe vasomotor symptoms associated with menopause and prevention of postmenopausal osteoporosis.2
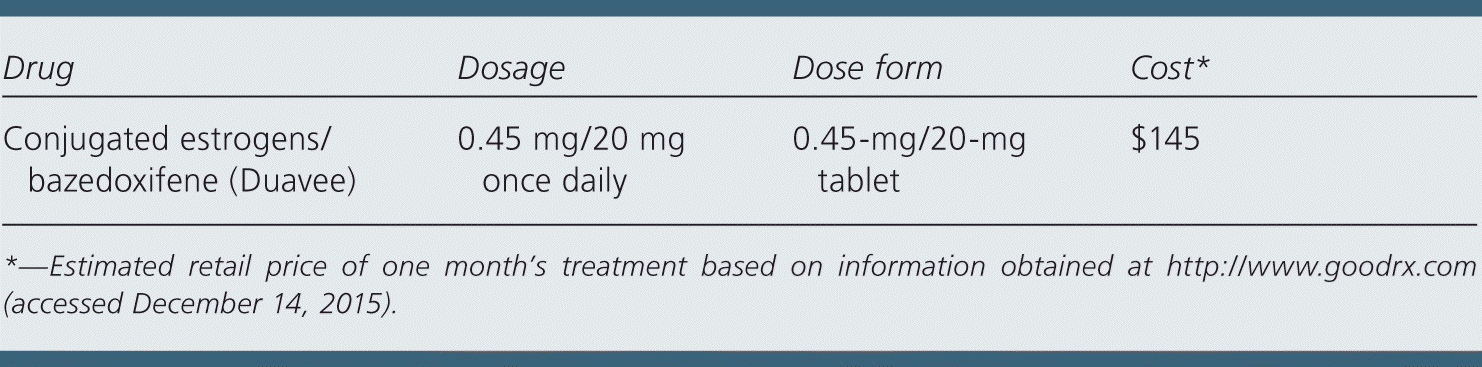
| Drug | Dosage | Dose form | Cost* |
|---|---|---|---|
| Conjugated estrogens/bazedoxifene (Duavee) | 0.45 mg/20 mg once daily | 0.45-mg/20-mg tablet | $145 |
SAFETY
As with other estrogen therapies, conjugated estrogens/bazedoxifene includes a boxed warning of increased risk of dementia in women older than 65 years, endometrial cancer, stroke, and deep venous thrombosis.2 However, in studies of 7,487 patients, there was no increase in endometrial hyperplasia. There is not enough research to directly evaluate the effect of conjugated estrogens/bazedoxifene on thrombotic or cardiovascular events.1,3,4 Because of established risks of estrogen agonists/antagonists, conjugated estrogens/bazedoxifene should not be used with other estrogen preparations or in patients with abnormal uterine bleeding, breast cancer, estrogen-dependent neoplasia, venous or arterial thromboembolism, liver disease, or thrombophilic disorders. It has not been studied in patients with renal impairment, obesity (body mass index greater than 34 kg per m2), or in women older than 75 years. Conjugated estrogens/bazedoxifene is a U.S. Food and Drug Administration pregnancy category X drug and should not be used by lactating women.2
TOLERABILITY
Conjugated estrogens/bazedoxifene is generally well tolerated. About one in 12 patients (8%) will stop taking the medication in the first year because of adverse effects.4,5 When compared with conjugated estrogens/medroxyprogesterone, conjugated estrogens/bazedoxifene results in less vaginal bleeding, and in head-to-head trials it has lower dropout rates due to adverse effects (8% vs. 13%; P < .05).4,5
EFFECTIVENESS
Conjugated estrogens/bazedoxifene reduces the number and severity of hot flashes, decreases pain with intercourse, and reduces vaginal dryness vs. placebo.1,3,6 Also, use of conjugated estrogens/bazedoxifene results in statistically significant improvements in sexual functioning, menopause-related quality of life, sleep quality, and satisfaction with treatment.7–9 Improvements in sleep quality and quality of life are similar to those seen with conjugated estrogens/medroxyprogesterone.9
For the prevention of postmenopausal osteoporosis, conjugated estrogens/bazedoxifene will maintain or slightly increase bone mineral density in the lumbar spine and hip. Its effect on vertebral, hip, or overall fracture rate is not known.4,5,10 Although conjugated estrogens/bazedoxifene does not affect breast density, studies have not examined its use for the prevention of breast cancer.11
PRICE
A one-month supply of conjugated estrogens/bazedoxifene costs approximately $145. In comparison, a one-month supply of conjugated estrogens/medroxyprogesterone costs approximately $148. Cyclical combined hormone replacement with separate tablets of estradiol and medroxyprogesterone costs about $10 per month.
SIMPLICITY
Conjugated estrogens/bazedoxifene is taken once daily without regard to meals.
Bottom Line
Conjugated estrogens/bazedoxifene is effective for the treatment of menopausal symptoms and may be better tolerated than conjugated estrogens/medroxyprogesterone. It will maintain bone mineral density in the lumbar spine and hip, but its effect on fractures is not known. Cardiovascular safety beyond two years is not known.
