Summary of Recommendations and Evidence
The USPSTF recommends initiating low-dose aspirin use for the primary prevention of cardiovascular disease (CVD) and colorectal cancer (CRC) in adults aged 50 to 59 years who have a 10% or greater 10-year CVD risk, are not at increased risk for bleeding, have a life expectancy of at least 10 years, and are willing to take low-dose aspirin daily for at least 10 years (Table 1). B recommendation.
Table 1. Aspirin Use for the Primary Prevention of Cardiovascular Disease and Colorectal Cancer: Clinical Summary of the USPSTF Recommendation
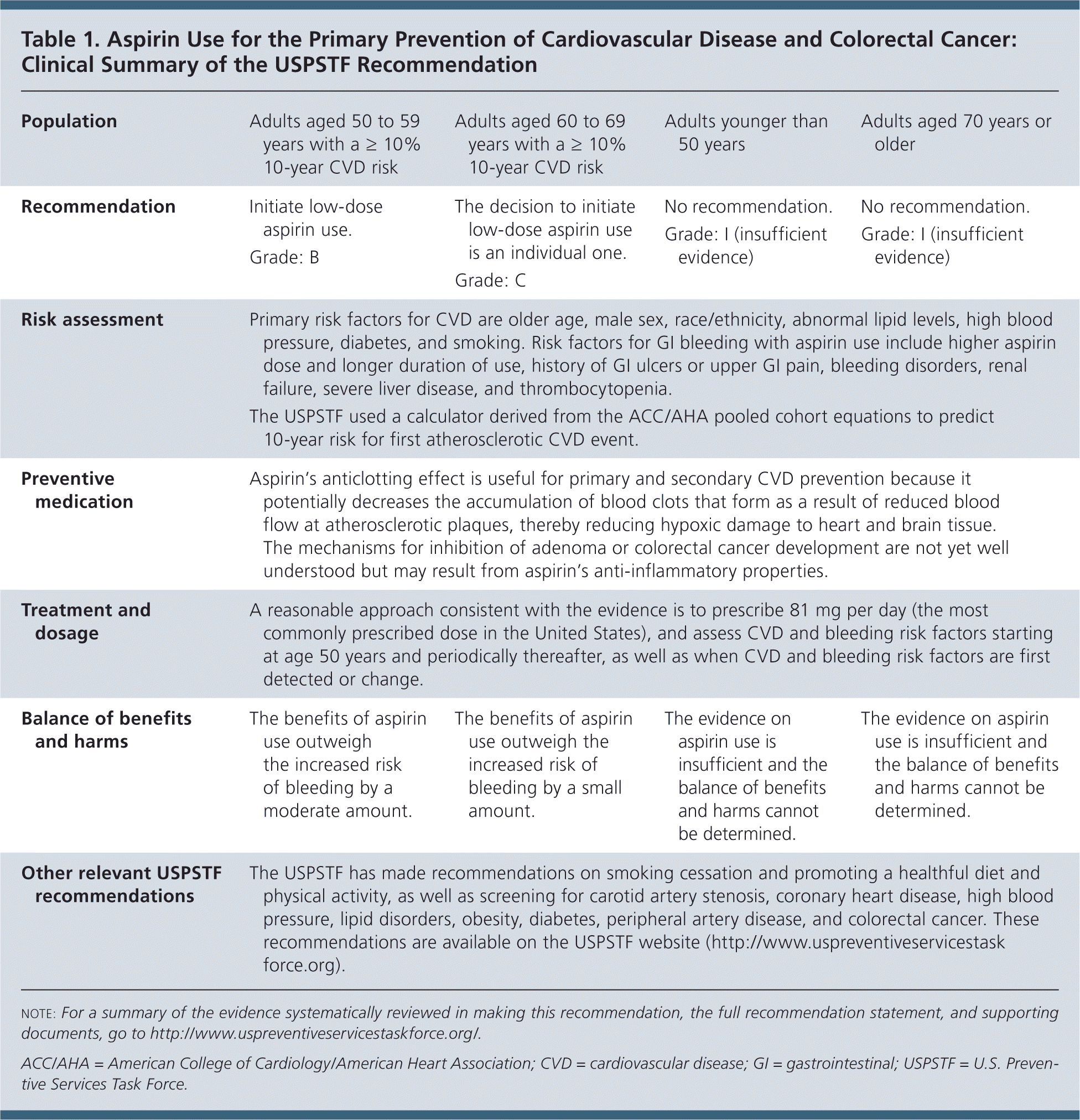
| Population | Adults aged 50 to 59 years with a ≥ 10% 10-year CVD risk | Adults aged 60 to 69 years with a ≥ 10% 10-year CVD risk | Adults younger than 50 years | Adults aged 70 years or older |
| Recommendation | Initiate low-dose aspirin use. | The decision to initiate low-dose aspirin use is an individual one. | No recommendation. | No recommendation. |
| Grade: B | Grade: C | Grade: I (insufficient evidence) | Grade: I (insufficient evidence) | |
| Risk assessment | Primary risk factors for CVD are older age, male sex, race/ethnicity, abnormal lipid levels, high blood pressure, diabetes, and smoking. Risk factors for GI bleeding with aspirin use include higher aspirin dose and longer duration of use, history of GI ulcers or upper GI pain, bleeding disorders, renal failure, severe liver disease, and thrombocytopenia. | |||
| The USPSTF used a calculator derived from the ACC/AHA pooled cohort equations to predict 10-year risk for first atherosclerotic CVD event. | ||||
| Preventive medication | Aspirin's anticlotting effect is useful for primary and secondary CVD prevention because it potentially decreases the accumulation of blood clots that form as a result of reduced blood flow at atherosclerotic plaques, thereby reducing hypoxic damage to heart and brain tissue. The mechanisms for inhibition of adenoma or colorectal cancer development are not yet well understood but may result from aspirin's anti-inflammatory properties. | |||
| Treatment and dosage | A reasonable approach consistent with the evidence is to prescribe 81 mg per day (the most commonly prescribed dose in the United States), and assess CVD and bleeding risk factors starting at age 50 years and periodically thereafter, as well as when CVD and bleeding risk factors are first detected or change. | |||
| Balance of benefits and harms | The benefits of aspirin use outweigh the increased risk of bleeding by a moderate amount. | The benefits of aspirin use outweigh the increased risk of bleeding by a small amount. | The evidence on aspirin use is insufficient and the balance of benefits and harms cannot be determined. | The evidence on aspirin use is insufficient and the balance of benefits and harms cannot be determined. |
| Other relevant USPSTF recommendations | The USPSTF has made recommendations on smoking cessation and promoting a healthful diet and physical activity, as well as screening for carotid artery stenosis, coronary heart disease, high blood pressure, lipid disorders, obesity, diabetes, peripheral artery disease, and colorectal cancer. These recommendations are available on the USPSTF website (http://www.uspreventiveservicestaskforce.org). | |||
note: For a summary of the evidence systematically reviewed in making this recommendation, the full recommendation statement, and supporting documents, go to http://www.uspreventiveservicestaskforce.org/.
ACC/AHA = American College of Cardiology/American Heart Association; CVD = cardiovascular disease; GI = gastrointestinal; USPSTF = U.S. Preventive Services Task Force.
The decision to initiate low-dose aspirin use for the primary prevention of CVD and CRC in adults aged 60 to 69 years who have a 10% or greater 10-year CVD risk should be an individual one. Persons who are not at increased risk for bleeding, have a life expectancy of at least 10 years, and are willing to take low-dose aspirin daily for at least 10 years are more likely to benefit. Persons who place a higher value on the potential benefits than the potential harms may choose to initiate low-dose aspirin. C recommendation.
The current evidence is insufficient to assess the balance of benefits and harms of initiating aspirin use for the primary prevention of CVD and CRC in adults younger than 50 years. I statement.
The current evidence is insufficient to assess the balance of benefits and harms of initiating aspirin use for the primary prevention of CVD and CRC in adults aged 70 years or older. I statement.
See the Clinical Considerations section for guidance on aspirin dosage.
Rationale
IMPORTANCE
CVD and CRC are major causes of death among U.S. adults. In 2011, more than one-half of all deaths in the United States were caused by heart disease, cancer, or stroke.1,2
RECOGNITION OF RISK STATUS
The primary risk factors for CVD include older age, male sex, race/ethnicity, abnormal lipid levels, high blood pressure, diabetes, and smoking.2
The USPSTF used a calculator derived from the American College of Cardiology/American Heart Association (ACC/AHA) pooled cohort equations to predict 10-year risk for first hard atherosclerotic CVD event (defined as nonfatal myocardial infarction [MI], coronary heart disease [CHD] death, and fatal or nonfatal stroke).3 Although concerns have been raised about the equations' potential to overpredict risk and their moderate discrimination, they are the only U.S.-based, externally validated equations that report risk as a combination of cerebrovascular and CHD events.
Risk factors for gastrointestinal (GI) bleeding with aspirin use include higher dose and longer duration of use, history of GI ulcers or upper GI pain, bleeding disorders, renal failure, severe liver disease, and thrombocytopenia. Other factors that increase risk for GI or intracranial bleeding with low-dose aspirin use include concurrent anticoagulation or nonsteroidal anti-inflammatory drug (NSAID) use, uncontrolled hypertension, male sex, and older age.4,5
This recommendation applies to adults who are at increased CVD risk and at average risk for CRC. Persons who are at increased CVD risk and are known to be at increased risk for CRC (for example, persons with a family or personal history of CRC or familial adenomatous polyposis)6 should consult their health care provider.
BENEFITS OF ASPIRIN USE
The USPSTF found adequate evidence that aspirin use to reduce risk for cardiovascular events (nonfatal MI and stroke) in adults aged 50 to 69 years who are at increased CVD risk is of moderate benefit. The magnitude of benefit varies by age and 10-year CVD risk.
The USPSTF found adequate evidence that aspirin use reduces the incidence of CRC in adults after 5 to 10 years of use.
The USPSTF found inadequate evidence that aspirin use reduces risk for CVD events in adults who are at increased CVD risk and are younger than 50 years or older than 69 years.
HARMS OF ASPIRIN USE
The USPSTF found adequate evidence that aspirin use in adults increases the risk for GI bleeding and hemorrhagic stroke. The USPSTF determined that the harms vary but are small in adults aged 59 years or younger and small to moderate in adults aged 60 to 69 years. The USPSTF found inadequate evidence to determine the harms of aspirin use in adults aged 70 years or older.
USPSTF ASSESSMENT
In adults aged 50 to 69 years who are at increased CVD risk, the benefits of aspirin use include prevention of MI and ischemic stroke and, with long-term use, reduced incidence of CRC. Aspirin use may also result in small to moderate harms, including GI bleeding and hemorrhagic stroke.
The USPSTF concludes with moderate certainty that the benefit of aspirin use for the primary prevention of CVD events, combined with the reduced incidence of CRC, outweighs the increased risk for bleeding by a moderate amount in adults aged 50 to 59 years who have a 10-year CVD risk of 10% or greater.
The USPSTF concludes with moderate certainty that the benefit of aspirin use for the primary prevention of CVD events, combined with the reduced incidence of CRC, outweighs the increased risk for bleeding by a small amount in adults aged 60 to 69 years who have a 10-year CVD risk of 10% or greater.
The USPSTF concludes that the evidence on aspirin use in adults younger than 50 years or older than 69 years is insufficient and the balance of benefits and harms cannot be determined.
Clinical Considerations
PATIENT POPULATION UNDER CONSIDERATION
This recommendation applies to adults aged 40 years or older without known CVD (including history of MI or stroke) and without increased bleeding risk (for example, history of GI ulcers, recent bleeding, or use of medications that increase bleeding risk).
ASSESSMENT OF THE BALANCE OF BENEFITS AND HARMS
The magnitude of the health benefits of aspirin use depends on an individual's baseline CVD risk and willingness to take aspirin for a sufficient duration to obtain the benefit of reduced incidence of CRC. The magnitude of harms depends on the presence of risk factors for bleeding.
Baseline CVD Risk. The magnitude of the cardiovascular risk reduction with aspirin use depends on an individual's initial risk for CVD events. Risk assessment for CVD should include ascertainment of the following risk factors: age, sex, race/ethnicity, total cholesterol level, high-density lipoprotein cholesterol level, systolic blood pressure, hypertension treatment, diabetes, and smoking. An online version of the ACC/AHA risk calculator can be found at http://tools.acc.org/ASCVD-Risk-Estimator/.
CRC Prevention. Colorectal cancer prevention plays an important role in the overall health benefit of aspirin, but this benefit is not apparent until 10 years after aspirin therapy is started. Patients need to take aspirin for at least 5 to 10 years to realize this potential benefit,6,7 and persons with shorter life expectancy are less likely to benefit. Thus, aspirin use is more likely to have an effect when it is started between the ages of 50 and 59 years. Because of the time required before a reduced incidence in CRC is seen, older persons (that is, 60 years or older) are less likely to realize this benefit than adults aged 50 to 59 years.8
GI and Intracranial Bleeding. Evidence shows that risk for GI bleeding, with and without aspirin use, increases with age. For this recommendation, the USPSTF considered older age and male sex to be important risk factors for GI bleeding. Other risk factors include upper GI tract pain, GI ulcers, concurrent anticoagulation or NSAID use, and uncontrolled hypertension.4,5 NSAID therapy combined with aspirin use increases the risk for serious GI bleeding compared with aspirin use alone.9 The rate of serious bleeding among aspirin users is about 2 to 3 times greater in patients with a history of GI ulcer.10 The risk for serious GI bleeding is 2 times greater in men than in women.10 These risk factors increase the risk for bleeding substantially and should be considered in the overall decision about whether to start or continue aspirin therapy. There is no evidence that enteric-coated or buffered formulations reduce the risk for serious GI bleeding.2,4,11
Balance of Benefits and Harms. The USPSTF used a CVD microsimulation model to estimate cardiovascular event rates based on baseline risk factors and aspirin use. It used the AHA/ACC risk calculator to stratify findings of benefits and harms by 10-year CVD risk. The USPSTF also calculated estimates of CRC incidence and harms of bleeding to determine the net balance of benefits and harms across individuals with varying baseline CVD risk.8,12
Tables 2 and 3 present the USPSTF's estimated lifetime number of nonfatal MIs, ischemic strokes, and cases of CRC prevented, stratified by 10-year CVD risk level, age, and sex, among adults aged 50 to 69 years (the age range with evidence of net benefit from aspirin use). In addition, Tables 2 and 3 present the USPSTF's estimated lifetime number of GI bleeding events and hemorrhagic strokes. The USPSTF developed these estimates assuming that aspirin users are not taking NSAIDs and do not have other conditions that increase risk for GI bleeding. The USPSTF estimated life-years and quality-adjusted life-years saved as one part of its consideration of the balance of benefits and harms of these disparate clinical outcomes (see the Implementation section on the USPSTF website for more information on interpreting the results in Tables 2 and 3).
Table 2. Lifetime Events in 10,000 Men Taking Aspirin*

| CVD risk | Nonfatal MIs prevented | Nonfatal ischemic strokes prevented | CRC cases prevented | Serious GI bleeding events caused | Hemorrhagic strokes caused | Net life-years gained | QALYs gained |
|---|---|---|---|---|---|---|---|
| Aged 50 to 59 years | |||||||
| 10% | 225 | 84 | 139 | 284 | 23 | 333 | 588 |
| 15% | 267 | 86 | 121 | 260 | 28 | 395 | 644 |
| 20% | 286 | 92 | 122 | 248 | 21 | 605 | 834 |
| Aged 60 to 69 years | |||||||
| 10% | 159 | 66 | 112 | 314 | 31 | −20 | 180 |
| 15% | 186 | 80 | 104 | 298 | 24 | 96 | 309 |
| 20% | 201 | 84 | 91 | 267 | 27 | 116 | 318 |
CRC = colorectal cancer; CVD = cardiovascular disease; GI = gastrointestinal; MI = myocardial infarction; QALY = quality-adjusted life-year.
*—A complete set of results is available in the decision analysis report (see reference 28 in the original recommendation statement at http://www.uspreventiveservicestaskforce.org/).
Table 3. Lifetime Events in 10,000 Women Taking Aspirin*
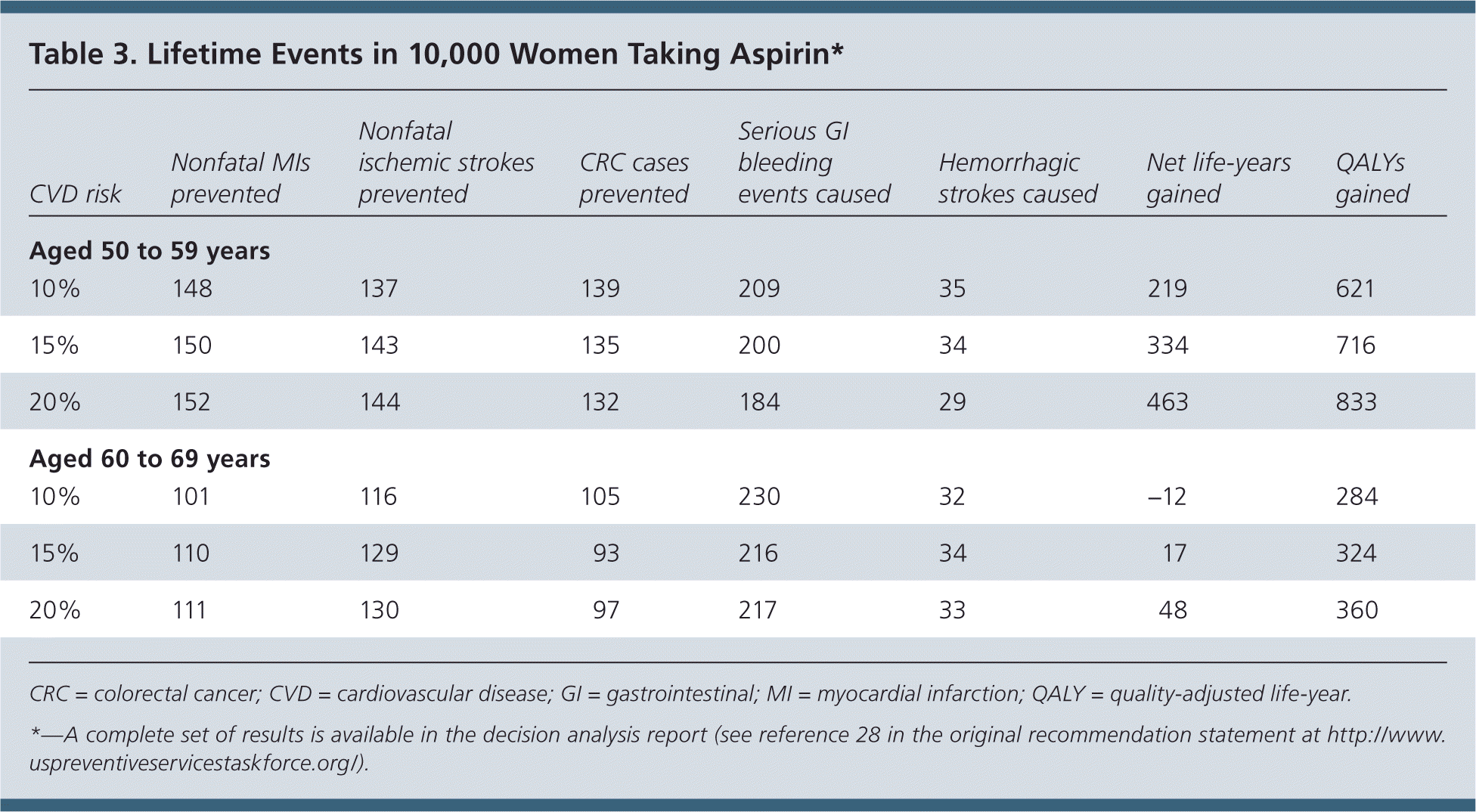
| CVD risk | Nonfatal MIs prevented | Nonfatal ischemic strokes prevented | CRC cases prevented | Serious GI bleeding events caused | Hemorrhagic strokes caused | Net life-years gained | QALYs gained |
|---|---|---|---|---|---|---|---|
| Aged 50 to 59 years | |||||||
| 10% | 148 | 137 | 139 | 209 | 35 | 219 | 621 |
| 15% | 150 | 143 | 135 | 200 | 34 | 334 | 716 |
| 20% | 152 | 144 | 132 | 184 | 29 | 463 | 833 |
| Aged 60 to 69 years | |||||||
| 10% | 101 | 116 | 105 | 230 | 32 | −12 | 284 |
| 15% | 110 | 129 | 93 | 216 | 34 | 17 | 324 |
| 20% | 111 | 130 | 97 | 217 | 33 | 48 | 360 |
CRC = colorectal cancer; CVD = cardiovascular disease; GI = gastrointestinal; MI = myocardial infarction; QALY = quality-adjusted life-year.
*—A complete set of results is available in the decision analysis report (see reference 28 in the original recommendation statement at http://www.uspreventiveservicestaskforce.org/).
Overall, the USPSTF determined that the greatest net benefit to be gained is by adults aged 50 to 59 years whose 10-year CVD risk is 10% or greater. The USPSTF recommends that persons in this age and risk group start taking aspirin. Adults aged 60 to 69 years may also benefit from starting aspirin use, although the net benefit is smaller due to the increased risk for GI bleeding and decreased benefit in CRC prevention in this age group.8
Further, the decision about the level of CVD risk at which the potential benefits outweigh potential harms is an individual one. Some adults may decide that avoiding an MI or a stroke is very important and that having a GI bleeding event is not as significant. They may decide to take aspirin at a lower CVD risk level than those who are more concerned about GI bleeding. Adults who have a high likelihood of benefit with little potential for harm should be encouraged to consider aspirin use. Conversely, adults who have little potential for benefit or are at high risk for GI bleeding should be discouraged from it.
TREATMENT AND DOSAGE
The optimal dose of aspirin to prevent CVD events is not known. Primary prevention trials have demonstrated benefits with various regimens, including doses of 75 and 100 mg per day and 100 and 325 mg every other day. A dose of 75 mg per day seems as effective as higher doses. The risk for GI bleeding may increase with the dosage. A pragmatic approach consistent with the evidence is to prescribe 81 mg per day, which is the most commonly prescribed dose in the United States.
Although the optimal timing and frequency of discussions about aspirin therapy are unknown, a reasonable approach may be to assess CVD and bleeding risk factors starting at age 50 years and periodically thereafter, as well as when CVD and bleeding risk factors are first detected or change.
SUGGESTIONS FOR PRACTICE REGARDING THE I STATEMENTS
Potential Preventable Burden. Evidence from primary prevention trials on the benefits of initiating aspirin use in adults younger than 50 years is limited. The potential benefit is probably lower than in adults aged 50 to 69 years because the risk for CVD events is lower (only a small percentage of adults younger than 50 years have a 10-year CVD risk ≥ 10%).8 Adults younger than 50 years who have an increased 10-year CVD risk may gain significant benefit from aspirin use; how much benefit is uncertain.
Evidence on the benefits and harms of initiating aspirin use in older adults is limited. Many adults aged 70 years or older are at increased risk for CVD because of their age. They have a high incidence of MI and stroke; thus, the potential benefit of aspirin could be substantial.
Potential Harms. The relationship between older age and GI bleeding is well established; thus, the potential harms for adults older than 70 years are significant. The complexity of risk factors, medication use, and concomitant illness make it difficult to assess the balance of benefits and harms of initiating aspirin use in this age group. In addition, aspirin use in adults older than 70 years results in smaller reductions in the incidence of CRC compared with younger adults.
Current Practice. Nearly 40% of U.S. adults older than 50 years use aspirin for the primary or secondary prevention of CVD.5 A study of National Health and Nutrition Examination Survey data assessed how common aspirin use is for the primary prevention of CVD and whether physicians recommend it or patients start it on their own. Among patients who were eligible for aspirin therapy and were at increased CHD risk (> 10% 10-year risk), about 41% were told by a physician to take aspirin. Among patients aged 65 years or older who were told by a physician to take aspirin, 80% adhered to the recommendation.13
USEFUL RESOURCES
The USPSTF has made other recommendations on CVD prevention, including smoking cessation and promoting a healthful diet and physical activity, as well as screening for carotid artery stenosis, CHD, high blood pressure, lipid disorders, obesity, diabetes, and peripheral artery disease. In addition, it has made recommendations on screening for CRC. These recommendations are available on the USPSTF website (http://www.uspreventiveservicestaskforce.org).
ADDITIONAL APPROACHES TO PREVENTION
Million Hearts (http://www.millionhearts.hhs.gov) is a national initiative to prevent 1 million heart attacks and strokes by 2017. It aims to prevent heart disease and stroke by improving access to effective care, improving the quality of care for the “ABCS” (aspirin when appropriate, blood pressure control, cholesterol management, and smoking cessation), focusing clinical attention on the prevention of heart attack and stroke, and activating the public to lead a heart-healthy lifestyle.
The Community Preventive Services Task Force recommends several intervention strategies to prevent CVD for communities and health care organizations (available at http://www.thecommunityguide.org/cvd/). For health care systems, it recommends introducing clinical decision-support systems to implement clinical guidelines at the point of care. For insurers and payers, it recommends reducing out-of-pocket costs to patients for medications to control high blood pressure and high cholesterol. For clinicians and health care organizations, it recommends incorporating multidisciplinary team-based care to improve blood pressure control, including patients, primary care providers, and other professionals (such as nurses, pharmacists, dietitians, social workers, and community health workers).

