The format of the 2002 schedule differs somewhat from the 2001 schedule; it is based in large part on the modifications developed by the Minnesota Department of Health. First, the schedule uses color, which makes the recommended ages, catch-up ages, and the preadolescent status check stand out (see accompanying schedule on page 128). Second, vaccines for children at high risk because of underlying medical conditions or environment are included under the dashed red line. These include influenza, pneumococcal polysaccharide, and hepatitis A vaccines. The third change reflects the first dose of hepatitis B vaccine.
According to the Advisory Committee on Immunization Practices, the U.S. Preventive Services Task Force, and other authorities, all pregnant women should be screened for hepatitis B surface antigen (HBsAg). Infants born to HBsAg-positive mothers should receive hepatitis B vaccine and 0.5 mL hepatitis B immune globulin (HBIG) at separate sites within 12 hours of birth to achieve maximum protection against hepatitis B. However, proper maternal screening for HBsAg, transmittal of results to the physician caring for the neonate, and vaccination has not always been done, resulting in neonates unnecessarily contracting hepatitis B with fatal or potentially fatal consequences.1 Hence, the first dose of hepatitis B is shaded beyond birth to denote that it should be given at birth unless the mother's surface antigen status is known to be negative, in which case the range from birth to two months of age is acceptable.
After the first quarter of 2002 (i.e., March 31, 2002), use in children of hepatitis B, Haemophilus influenzae type b (Hib), and diphtheria and tetanus toxoids and acellular pertussis (DTaP) vaccines that contain thimerosal will no longer be recommended. There are several reasons for this decision. First, it is a way to lessen exposure to heavy metals. Second, it is a logical extension of previous recommendations for reduction of thimerosal in vaccines.2 Third, it responds to the report from the Institute of Medicine (IOM) that recommends vaccines without thimerosal be used for children in situations where an alternative is available, as is the case for hepatitis B, Hib, and DTaP vaccines.3 There have been no known harms associated with thimerosal-containing vaccines and the IOM found that the evidence is inadequate to accept or reject a causal relationship between thimerosal-containing vaccines and neurodevelopmental disorders.
I recommend that physicians check refrigerators for supplies of hepatitis B, Hib, and DTaP vaccines that contain thimerosal. Because immediate changes in policy could lead to delays in immunizing children and because a few children have unintentionally contracted vaccine-preventable diseases following quick changes in policy in the past, the new recommendation is being phased in over several months.1 Thus, thimerosal-containing vaccines may be used through March of 2002. Both manufacturers of hepatitis B vaccine will voluntarily replace thimerosal-containing hepatitis B vaccines. Manufacturers are no longer producing childhood hepatitis B, Hib, or DTaP vaccines with thimerosal (or the amount has been reduced to trace). Inactivated poliovirus (IPV); measles, mumps, rubella (MMR); varicella vaccine; and pneumococcal conjugate vaccine never contained thimerosal. Thimerosal-containing influenza vaccine may continue to be used as substantial amounts of an alternative are not available. Useful Web sites for current information includewww.immunizationed.org, a site developed by family physician educators;www.immunize.org;www.aafp.org;www.cdc.gov/nip; andwww.immunizationinfo.org.
Recommended Childhood Immunization Schedule, United States, 2002
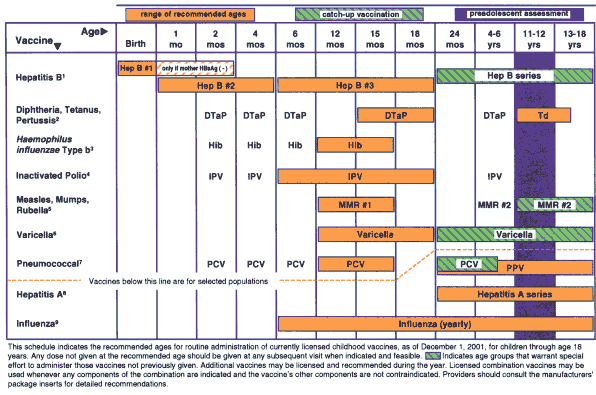
1. Hepatitis B vaccine (Hep B). All infants should receive the first dose of hepatitis B vaccine soon after birth and before hospital discharge; the first dose may also be given by age 2 months if the infant's mother is HBsAg-negative. Only monovalent hepatitis B vaccine can be used for the birth dose. Monovalent or combination vaccine containing Hep B may be used to complete the series; four doses of vaccine may be administered if combination vaccine is used. The second dose should be given at least 4 weeks after the first dose, except for Hib-containing vaccine which cannot be administered before age 6 weeks. The third dose should be given at least 16 weeks after the first dose and at least 8 weeks after the second dose. The last dose in the vaccination series (third or fourth dose) should not be administered before age 6 months.
Infants born to HBsAg-positive mothers should receive hepatitis B vaccine and 0.5 mL hepatitis B immune globulin (HBIG) within 12 hours of birth at separate sites. The second dose is recommended at age 1–2 months and the vaccination series should be completed (third or fourth dose) at age 6 months.
Infants born to mothers whose HBsAg status is unknown should receive the first dose of the hepatitis B vaccine series within 12 hours of birth. Maternal blood should be drawn at the time of delivery to determine the mother's HBsAg status; if the HBsAg test is positive, the infant should receive HBIG as soon as possible (no later than age 1 week).
2. Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP). The fourth dose of DTaP may be administered as early as age 12 months, provided 6 months have elapsed since the third dose and the child is unlikely to return at age 15–18 months.Tetanus and diphtheria toxoids (Td) is recommended at age 11–12 years if at least 5 years have elapsed since the last dose of tetanus and diphtheria toxoid-containing vaccine. Subsequent routine Td boosters are recommended every 10 years.
3. Haemophilus influenzae type b (Hib) conjugate vaccine. Three Hib conjugate vaccines are licensed for infant use. If PRP-OMP (PedvaxHIB or ComVax [Merck] is administered at ages 2 and 4 months, a dose at age 6 months is not required. DTaP/Hib combination products should not be used for primary immunization in infants at ages 2, 4, or 6 months, but can be used as boosters following any Hib vaccine.
4. Inactivated polio vaccine (IPV). An all-IPV schedule is recommended for routine childhood polio vaccination in the United States. All children should receive four doses of IPV at ages 2 months, 4 months, 6–18 months, and 4–6 years.
5. Measles, mumps, and rubella vaccine (MMR). The second dose of MMR is recommended routinely at age 4–6 years but may be administered during any visit, provided at least 4 weeks have elapsed since the first dose and that both doses are administered beginning at or after age 12 months. Those who have not previously received the second dose should complete the schedule by the 11–12-year-old visit.
6. Varicella vaccine. Varicella vaccine is recommended at any visit at or after age 12 months for susceptible children, i.e., those who lack a reliable history of chickenpox. Susceptible persons aged 13 years should receive two doses, given at least 4 weeks apart.
7. Pneumococcal vaccine. The heptavalent pneumococcal conjugate vaccine (PCV) is recommended for all children age 2–23 months. It is also recommended for certain children age 24–59 months. Pneumococcal polysaccharide vaccine (PPV) is recommended in addition to PCV for certain high-risk groups. See MMWR 2000;49(RR-9):1–35.
8. Hepatitis A vaccine. Hepatitis A vaccine is recommended for use in selected states and regions, and for certain high-risk groups; consult your local public health authority. See MMWR 1999;48(RR-12):1–37.
9. Influenza vaccine. Influenza vaccine is recommended annually for children age 6 months with certain risk factors (including, but not limited to, asthma, cardiac disease, sickle cell disease, HIV, diabetes; see MMWR 2001;50(RR-4):1–44, and can be administered to all others wishing to obtain immunity. Children aged 12 years should receive vaccine in a dosage appropriate for their age (0.25 mL if age 6–35 months or 0.5 mL if aged 3 years). Children aged 8 years who are receiving influenza vaccine for the first time should receive two doses separated by at least 4 weeks.
For additional information about vaccines, vaccine supply, and contraindications for immunization, please visit the National Immunization Program Web site at www.cdc.gov/nip or call the National Immunization Hotline at 800-232-2522 (English) or 800-232-0233 (Spanish).
Approved by the Advisory Committee on Immunization Practices (www.cdc.gov/nip/acip), the American Academy of Pediatrics (www.aap.org), and the American Academy of Family Physicians (www.aap.org).

