
Am Fam Physician. 2002;66(5):817-820
Echinococcus tapeworms are parasites that infect dog species worldwide and occasionally are transmitted to humans. Infection occurs most commonly in persons who raise sheep or cattle, and who have contact with dogs. The tapeworm eggs are typically ingested during play with dogs or through consumption of garden vegetables or water contaminated by dog feces. The most common sites of tapeworm cyst formation within the body are the liver and lungs, although any internal organ or bone can be infected. The tapeworm infection is generally asymptomatic for 10 to 20 years, until the cyst grows large enough to cause problems. Untreated infection can be fatal. The possibility of this uncommon infection is most often considered when cysts are found on imaging studies of affected organs. Serologic testing has variable sensitivity, but it can be helpful. Surgical removal of cysts is the usual treatment, often with perioperative use of anthelmintic medication to prevent recurrence of disease caused by later growth of undetected daughter cysts.
Echinococcosis (also known as hydatid disease) is an important public health problem in many parts of the world, especially in rural areas where sheep and cattle are raised.1,2 Echinococcus tapeworms are parasitic organisms with a two-stage life cycle.3,4 In the adult phase, the tapeworm lives in the intestines of the dog who is the so-called “definitive” host. A great number of other animals, especially sheep or cattle and, occasionally, humans, can become infected with the larval stage of the worm and are called “intermediate” hosts. Intermediate hosts become infected when they ingest food or water contaminated by dog feces. Most human cases occur in areas where dogs and livestock are raised together.1–3,5
The liver and lungs are the most common sites for cyst formation in humans, although any organ can be involved. Symptom presentation is varied and nonspecific. The diagnosis is most often arrived at when radiographic studies show space-occupying lesions of internal organs, bone, or brain.5
Epidemiology
Human infection, often acquired in childhood during play with infected dogs, is most common in sheep-raising areas, especially in Russia and China, but also in Europe,6 South Africa, Australia,7 New Zealand, the Middle East,8 and South America.1,9,10 Most of the new human cases that are diagnosed in the United States each year occur in members of well-defined risk groups, such as sheep farmers in the West, some Native American tribes in the Southwest,9,11 and immigrants who acquired their infection abroad.1,2,4 A northern strain of echinococcosis, with a life cycle in caribou, moose, and wolves, infects Native Inuit and Aleut populations in Alaska and Canada.4
Clinical Presentation
In humans, echinococcosis can be a serious disease, with a mortality rate of 4 to 5 percent.12 Cyst size and location greatly influence the outcome. Most hydatid cysts develop in the liver, and most of them are asymptomatic (approximately 60 percent) at the time of diagnosis.3 Cysts grow from 1 to 5 cm per year.2,13
The lung is the next most frequent site of cyst location, followed by the kidneys, muscle, spleen, soft tissue, brain, and bone. Symptoms are generally caused by a mechanical obstruction within the affected organ.3,13 Cysts may be symptomless for 10 to 20 years until they become large enough to produce symptoms.2 During the life of the cyst, leakage into the systemic circulation can sensitize the patient, and if a cyst ruptures suddenly, a severe allergic reaction or even anaphylaxis and death may occur.2,3,5
Most cases of echinococcosis are caused by infection with Echinococcus granulosus, which has a limiting membrane that surrounds the growing cysts.7 Much less common, but more serious, is alveolar echinococcosis caused by ingestion of Echinococcus multilocularis.5 This species does not have a limiting membrane and can grow aggressively; if left untreated, this infection is almost always fatal.3,5,7
Diagnosis
Echinococcosis should be considered in patients with an abdominal mass and no clearly defined diagnosis.12 The typical appearance on imaging is a fluid-filled mass with a well-defined outer wall and internal septations.
Serologic testing is more specific, but less sensitive, than most imaging modalities.1,3 In patients with lung cysts, false-negative results occur in up to 50 percent of infections.2 Enzyme-linked immunoelectrotransfer blot (EITB),11,13 where available, is the test of choice. In some studies, it has greater than 95 percent sensitivity and specificity.13 Enzyme-linked immunosorbent assay (ELISA) has up to 84 percent sensitivity.1 (This test can be performed at the Laboratory Corporation of America in Cypress, Calif.).
It has been reported that the detection of specific IgG1 and IgG4 serum antibodies to echinococcosis can enhance the specificity of ELISA.1,2 Serologic tests alone cannot be used to definitively establish or exclude echinococcosis. Fine-needle aspiration is a reliable method of diagnosing cystic lesions in patients with negative or equivocal serology. The characteristic findings of tapeworm anatomic parts and eosinophilic-appearing granules in the aspirated fluid confirm the diagnosis.13–15
Treatment
Therapy for echinococcosis is based on the size, location, and symptomatic complications of the cysts. Surgery is the treatment of choice2,3,13; however, removal of the main cyst mass may not be 100 percent effective because small “daughter” cysts can be left behind. Chemotherapy (e.g., albendazole [Albenza] and mebendazole [Vermox])2,6 is effective against tapeworm disease, and its use is indicated for the treatment of patients with inoperable disease or as presurgical and post-surgical treatment to reduce the risk of recurrence.7 Ultrasonographic or computed tomo-graphic (CT)-guided fine-needle aspiration of hydatid cyst contents followed by infusion of a killing agent, such as 95 percent ethanol, and reaspiration, known as PAIR therapy (Puncture, Aspiration, Injection, Reaspiration),3,11,12 has been used successfully at some centers but carries a risk of dissemination of infection or anaphylactic reaction caused by cyst puncture and leakage.2,3,13
The following case is an example of the presentation, diagnosis, and treatment of echinococcosis.
Illustrative Case
A 48-year-old man who moved to the United States three years ago from Bosnia presented to a physician's office for an initial physical examination. The patient reported a medical history of kidney stones diagnosed and treated in Bosnia in 1995, and elevated cholesterol levels. The patient's urine was positive for nonhemolyzed blood and a trace of protein. Urine culture and sensitivity were negative. Three months later, the patient presented with pelvic pain and the passage of a clot and a small stone fragment in his urine. At that point, the patient was scheduled for renal ultrasonography and a 24-hour urine collection for further work-up. Renal ultrasonography showed a normal right kidney, but the left kidney demonstrated a multicystic, septated complex area, mainly in the middle and lower portion, measuring approximately 9 cm × 4 cm × 5 cm.
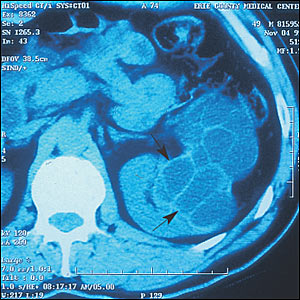
The possibility of a congenital anomaly was considered. ACT scan of theabdomen showed normal liver, gallbladder, pancreas, and spleen. A dumbbell-shaped, fluid-filled, multiseptated structure appeared in the middle portion of the left kidney (Figure 1). Diagnostic considerations were expanded to include cystic nephroma, multicystic renal cell carcinoma, or hydatid disease. Serologic testing for antibodies to Echinococcus was positive. A CT scan of the chest was negative for additional cysts.
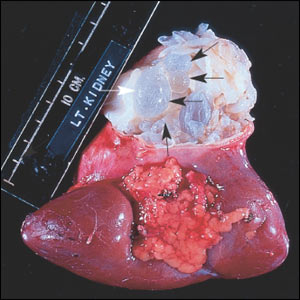
The patient opted for surgical drainage under ultrasound guidance. The procedure failed, so the patient opted for a radical nephrectomy (Figure 2), which was performed without any complications.
Prevention
Accidental ingestion of garden vegetables, water, or soil contaminated with tapeworm eggs from dog feces initiates this infection,12,13 so it is important to educate populations in endemic areas about careful washing of hands and food. The feeding of livestock entrails to dogs is a common starting point for the tapeworm life cycle and should be avoided.13 Educational programs should be initiated early because, presumably, most infections are acquired in childhood.2,13,15