
This is a corrected version of the article that appeared in print.
Am Fam Physician. 2013;88(4):263-264
Author disclosure: No relevant financial affiliations.
Vilazodone (Viibryd) is a serotonin1A partial agonist and reuptake inhibitor labeled for the treatment of major depressive disorder in adults.
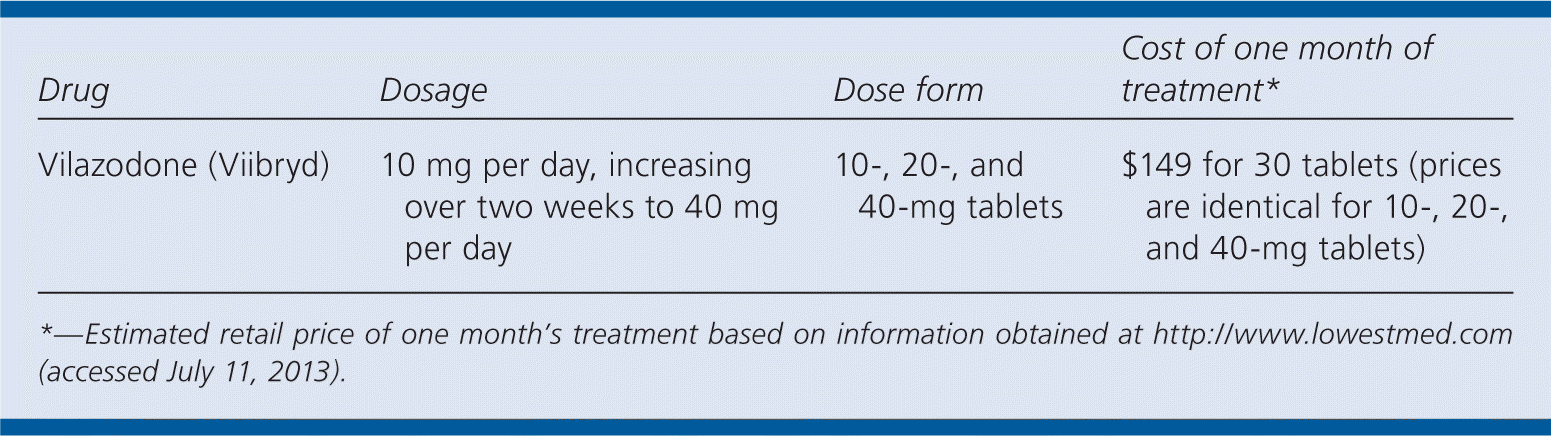
| Drug | Dosage | Dose form | Cost of one month of treatment* |
|---|---|---|---|
| Vilazodone (Viibryd) | 10 mg per day, increasing over two weeks to 40 mg per day | 10-, 20-, and 40-mg tablets | $149 for 30 tablets (prices are identical for 10-, 20-, and 40-mg tablets) |
SAFETY
Vilazodone causes few serious adverse effects, although long-term safety has not been established.1 In a one-year open-label case series, 3.8% of patients experienced serious adverse effects, including pneumonia, suicidal ideation or behavior, and serotonin syndrome.1 As with other antidepressants, vilazodone carries a boxed warning for increased risk of suicidality in patients 24 years and younger.2 Vilazodone is contraindicated in patients taking monoamine oxidase inhibitors, and its use should be avoided with other serotonergic drugs, including antidepressants, tramadol (Ultram), and triptans.2 Vilazodone does not cause electrocardiographic changes or significant abnormal findings on basic laboratory tests.1–4 Strong inhibitors of the cytochrome P450 3A4 liver enzyme, such as ketoconazole, may cause excessive accumulation of vilazodone; therefore, the dosage of vilazodone should be reduced to 20 mg per day when used concurrently with these types of medications.2,5
TOLERABILITY
About one in 14 patients taking vilazodone for eight weeks will discontinue the medication because of unwanted effects, which is about twice as common as in patients taking placebo.2–4 The most common adverse effects are nausea and diarrhea, although these are usually transient.2–4 Less commonly, vilazodone may cause dizziness, insomnia, vomiting, abnormal dreams, akathisia, palpitations, tremor, and paresthesias.2–4 Vilazodone causes sexual dysfunction in about 16% of men and 5% of women.2–4 Patients can expect to gain about 3.8 lb (1.7 kg) after 12 months.1
EFFECTIVENESS
Vilazodone has not been studied in patients who have mild to moderate major depression. It has been studied in patients with moderate to severe depression, with investigators looking at clinical remission and treatment response (characterized as a 50% or greater reduction in depression score). Among outpatients with moderate to severe depression, vilazodone will induce clinical remission in about 25% of patients compared with 18% taking placebo,2–4 with one additional patient achieving relief after eight weeks for every 14 patients treated with vilazodone compared with placebo (number needed to treat [NNT] = 14; 95% confidence interval [CI], 8 to 55).2–4 Similarly, about 42% of patients who have moderate to severe depression will respond to treatment (NNT = 8; 95% CI, 6 to 16).2–4 To date, no research has been conducted to determine the rate of relapse in patients taking vilazodone. There are no studies comparing vilazodone with other depression treatments.
PRICE
SIMPLICITY
Bottom Line
The available evidence does not support the routine use of vilazodone in the treatment of depression. It is significantly more expensive than generic alternatives and is no more effective for moderate to severe depression than selective serotonin reuptake inhibitors or tricyclic antidepressants.8 Furthermore, vilazodone has not been studied in the treatment of mild to moderate depression, the kind most commonly seen by family physicians. Although vilazodone appears to be safe for long-term use based on a small study of one year's duration, other antidepressants have much more research supporting their long-term safety.
