
Am Fam Physician. 2018;98(9):603-606
Author disclosure: No relevant financial affiliations.
Key Clinical Issue
What are the clinical effectiveness and harms of cell-based therapies, oral glucosamine and/or chondroitin, physical treatment interventions, weight loss, and home-based and self-management therapies for knee osteoarthritis (OA)?
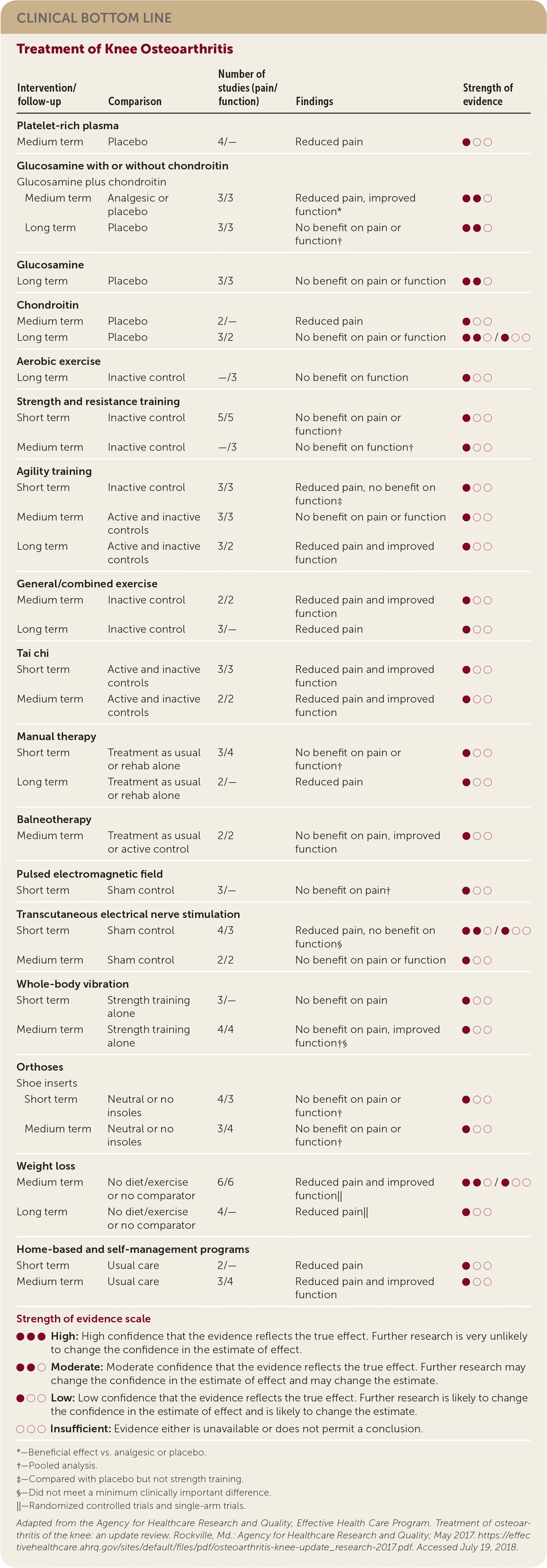
| Intervention/follow-up | Comparison | Number of studies (pain/function) | Findings | Strength of evidence |
|---|---|---|---|---|
| Platelet-rich plasma | ||||
| Medium term | Placebo | 4/— | Reduced pain | ● ○ ○ |
| Glucosamine with or without chondroitin | ||||
| Glucosamine plus chondroitin | ||||
| Medium term | Analgesic or placebo | 3/3 | Reduced pain, improved function* | ● ● ○ |
| Long term | Placebo | 3/3 | No benefit on pain or function† | ● ● ○ |
| Glucosamine | ||||
| Long term | Placebo | 3/3 | No benefit on pain or function | ● ● ○ |
| Chondroitin | ||||
| Medium term | Placebo | 2/— | Reduced pain | ● ○ ○ |
| Long term | Placebo | 3/2 | No benefit on pain or function | ● ● ○/● ○ ○ |
| Aerobic exercise | ||||
| Long term | Inactive control | —/3 | No benefit on function | ● ○ ○ |
| Strength and resistance training | ||||
| Short term | Inactive control | 5/5 | No benefit on pain or function† | ● ○ ○ |
| Medium term | Inactive control | —/3 | No benefit on function† | ● ○ ○ |
| Agility training | ||||
| Short term | Inactive control | 3/3 | Reduced pain, no benefit on function‡ | |
| Medium term | Active and inactive controls | 3/3 | No benefit on pain or function | |
| Long term | Active and inactive controls | 3/2 | Reduced pain and improved function | |
| General/combined exercise | ||||
| Medium term | Inactive control | 2/2 | Reduced pain and improved function | |
| Long term | Inactive control | 3/— | Reduced pain | |
| Tai chi | ||||
| Short term | Active and inactive controls | 3/3 | Reduced pain and improved function | |
| Medium term | Active and inactive controls | 2/2 | Reduced pain and improved function | ● ○ ○ |
| Manual therapy | ||||
| Short term | Treatment as usual or rehab alone | 3/4 | No benefit on pain or function† | ● ○ ○ |
| Long term | Treatment as usual or rehab alone | 2/— | Reduced pain | ● ○ ○ |
| Balneotherapy | ||||
| Medium term | Treatment as usual or active control | 2/2 | No benefit on pain, improved function | ● ○ ○ |
| Pulsed electromagnetic field | ||||
| Short term | Sham control | 3/— | No benefit on pain† | ● ○ ○ |
| Transcutaneous electrical nerve stimulation | ||||
| Short term | Sham control | 4/3 | Reduced pain, no benefit on function§ | ● ● ○/● ○ ○ |
| Medium term | Sham control | 2/2 | No benefit on pain or function | ● ○ ○ |
| Whole-body vibration | ||||
| Short term | Strength training alone | 3/— | No benefit on pain | ● ○ ○ |
| Medium term | Strength training alone | 4/4 | No benefit on pain, improved function†§ | ● ○ ○ |
| Orthoses | ||||
| Shoe inserts | ||||
| Short term | Neutral or no insoles | 4/3 | No benefit on pain or function† | ● ○ ○ |
| Medium term | Neutral or no insoles | 3/4 | No benefit on pain or function† | ● ○ ○ |
| Weight loss | ||||
| Medium term | No diet/exercise or no comparator | 6/6 | Reduced pain and improved function|| | ● ○ ○/ |
| Long term | No diet/exercise or no comparator | 4/— | Reduced pain|| | ● ○ ○ |
| Home-based and self-management programs | ||||
| Short term | Usual care | 2/— | Reduced pain | ● ○ ○ |
| Medium term | Usual care | 3/4 | Reduced pain and improved function | ● ● ○/● ○ ○ |
Evidence-Based Answer
For short-term (four to 12 weeks) pain relief and/or improvement of function, beneficial interventions include transcutaneous electrical nerve stimulation (TENS), tai chi, and home-based and self-management programs. (Strength of Recommendation [SOR]: B, based on inconsistent or limited-quality patient-oriented evidence.) Platelet-rich plasma injections and home-based and self-management programs reduce pain in the medium term (12 to 26 weeks). (SOR: B, based on inconsistent or limited-quality patient-oriented evidence.) Glucosamine and chondroitin supplements have medium-term but not long-term benefits. (SOR: B, based on inconsistent or limited-quality patient-oriented evidence.) Shoe inserts are ineffective. (SOR: B, based on inconsistent or limited-quality patient-oriented evidence.) For long-term (more than 26 weeks) outcomes, beneficial interventions include weight loss, agility training, combined exercise programs, and manual therapy (i.e., massage, self-massage, and acupressure). (SOR: B, based on inconsistent or limited-quality patient-oriented evidence.) There is insufficient evidence to determine whether treatment outcomes or adverse events vary in different populations.1
Practice Pointers
Symptomatic OA of the knee affects an estimated 10% of men and 13% of women 60 years or older in the United States.2 Knee OA is characterized by destruction of articular and subchondral bone cartilage and osteophyte formation, and causes pain with weight-bearing, limitation of movement, and reduction in function, including the ability to complete activities of daily living.1,3 Goals of treatment include pain relief and improvement in function and health-related quality of life.
This Agency for Healthcare Research and Quality (AHRQ) review included 107 studies regarding the effectiveness of cell-based therapies, oral glucosamine and/or chondroitin, physical treatment interventions, weight loss, or home-based and self-management therapies in patients with knee OA. Based on low strength of evidence from four randomized controlled trials (RCTs), platelet-rich plasma injections reduce pain and improve quality of life compared with placebo for up to 26 weeks. Based on moderate strength of evidence from three RCTs, the combination of glucosamine and chondroitin improves medium-term outcomes but has no benefits beyond 26 weeks. Compared with sham controls, TENS reduces pain in the short term but not function and does not improve function or pain after 12 weeks.
Regarding exercise, 10 studies found no statistically significant benefit of strength and resistance training on pain or function for up to 26 weeks. Similarly, three RCTs of aerobic exercise alone found no long-term benefits on function. However, three RCTs of agility training showed modest benefits on long-term pain and function, and programs that combine strength and aerobic exercise interventions reduce pain and improve function in the medium term and reduce pain in the long term, based on four RCTs. Home-based exercise and self-management programs that include strength, agility, and pain-coping skills have beneficial effects in the short and medium term. Tai chi also appears to improve short- and medium-term outcomes, based on low strength of evidence from three RCTs. There was insufficient evidence to assess the effects of yoga.
Weight loss (achieved with diet and/or exercise) reduces pain and improves function in the medium term and reduces pain in the long term, based on low to moderate strength of evidence.
Shoe inserts had no effect on pain or function in eight RCTs. There was insufficient evidence to determine the effectiveness of knee braces, custom shoes, or cane use.
Adverse events were reported in 57 studies. Of therapies found to be at least partially effective, platelet-rich plasma injections were associated with knee pain and stiffness in 44% of patients who received two injections in one RCT. There were no differences in adverse events between glucosamine and/or chondroitin and placebo or active controls. There was no difference in worsening of knee symptoms following TENS between intervention and sham control groups. Persons on weight loss diets reported more nonserious gastrointestinal symptoms (e.g., constipation). Serious adverse events were rarely reported and not limited to active treatment groups.
The AHRQ review findings were mostly consistent with 2013 practice guidelines from the American Academy of Orthopaedic Surgeons (AAOS), which recommend self-management programs and low-impact aerobic exercise for persons with symptomatic knee OA and weight loss for persons with a body mass index of 25 kg per m2 or greater.4 The AAOS recommends against the use of glucosamine, chondroitin, and lateral wedge insoles. The recommendation against the use of glucosamine and chondroitin is based on the absence of evidence that these supplements slow cartilage loss or have long-term benefits and the lack of regulation of these therapies by the U.S. Food and Drug Administration.4
Nonsurgical interventions for knee OA that were not evaluated in the AHRQ review include corticosteroid and hyaluronic acid injections, acupuncture, and oral and topical medications. A Cochrane review found that most RCTs that suggest short-term benefits of intra-articular corticosteroids are limited by methodological issues, primarily unblinding.5 Another systematic review and meta-analysis found that double-blinded, sham-controlled trials of hyaluronic acid injections do not show a clinically significant benefit.6 The AAOS guideline recommends against acupuncture for knee OA because the few studies that reported positive outcomes were statistically, but not clinically, significant.4
Editor's Note:American Family Physician SOR ratings are different from the AHRQ Strength of Evidence (SOE) ratings.
