Venous ulcers are the most common type of chronic lower extremity ulcers, affecting 1% to 3% of the U.S. population. Venous hypertension as a result of venous reflux (incompetence) or obstruction is thought to be the primary underlying mechanism for venous ulcer formation. Risk factors for the development of venous ulcers include age 55 years or older, family history of chronic venous insufficiency, higher body mass index, history of pulmonary embolism or superficial/deep venous thrombosis, lower extremity skeletal or joint disease, higher number of pregnancies, parental history of ankle ulcers, physical inactivity, history of ulcers, severe lipodermatosclerosis, and venous reflux in deep veins. Poor prognostic signs for healing include ulcer duration longer than three months, initial ulcer length of 10 cm or more, presence of lower limb arterial disease, advanced age, and elevated body mass index. On physical examination, venous ulcers are generally irregular and shallow with well-defined borders and are often located over bony prominences. Signs of venous disease, such as varicose veins, edema, or venous dermatitis, may be present. Other associated findings include telangiectasias, corona phlebectatica, atrophie blanche, lipodermatosclerosis, and inverted champagne-bottle deformity of the lower leg. Chronic venous ulcers significantly impact quality of life. Severe complications include infection and malignant change. Current evidence supports treatment of venous ulcers with compression therapy, exercise, dressings, pentoxifylline, and tissue products. Referral to a wound subspecialist should be considered for ulcers that are large, of prolonged duration, or refractory to conservative measures. Early venous ablation and surgical intervention to correct superficial venous reflux can improve healing and decrease recurrence rates.
Venous ulcers are open skin lesions that occur in an area affected by venous hypertension.1 The prevalence of venous ulcers in the United States ranges from 1% to 3%.2,3 In the United States, 10% to 35% of adults have chronic venous insufficiency, and 4% of adults 65 years or older have venous ulcers.4 Risk factors for venous ulcers include age 55 years or older, family history of chronic venous insufficiency, higher body mass index, history of pulmonary embolism or superficial/deep venous thrombosis, lower extremity skeletal or joint disease, higher number of pregnancies, parental history of ankle ulcers, physical inactivity, history of ulcers, severe lipodermatosclerosis (panniculitis that leads to skin induration or hardening, increased pigmentation, swelling, and redness), and venous reflux in deep veins.5 Poor prognostic signs for healing include ulcer duration longer than three months, ulcer length of 10 cm (3.9 in) or more, presence of lower limb arterial disease, advanced age, and elevated body mass index.6
SORT: KEY RECOMMENDATIONS FOR PRACTICE
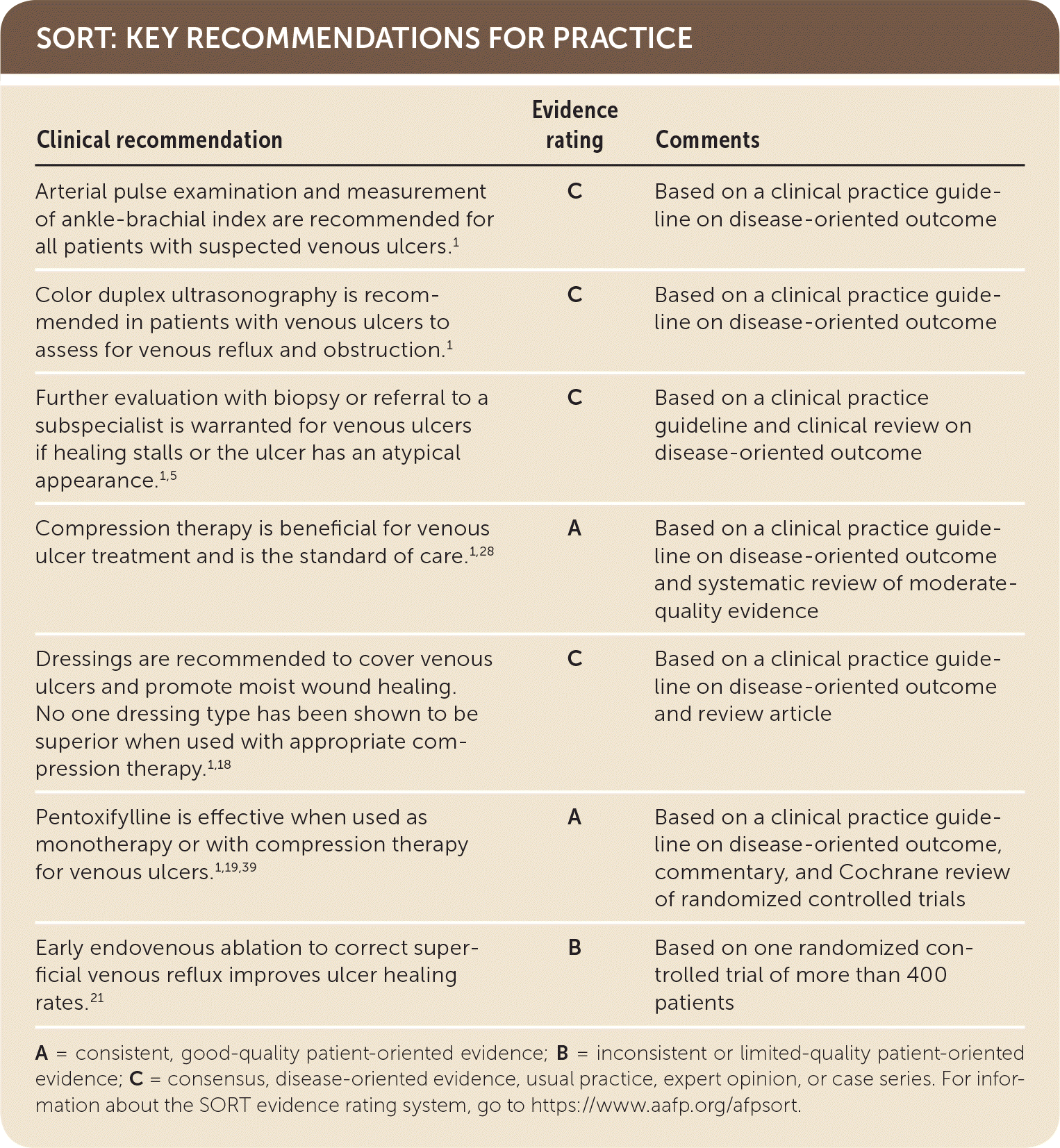
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Complications of venous ulcers include infections and skin cancers such as squamous cell carcinoma.7,8 Venous ulcers are a major cause of morbidity and can lead to high medical costs.3 Economic and personal impacts include frequent visits to health care facilities, loss of productivity, increased disability, discomfort, need for dressing changes, and recurrent hospitalizations. In one study, patients with venous ulcers used more medical resources than those without, and their annual per-patient health expenditures were increased by $6,391 for those with Medicare and $7,030 for those with private health insurance. Employed individuals with venous ulcers missed four more days of work per year than those without venous ulcers (29% increase in work-loss costs).9
Pathophysiology
Venous hypertension is defined as increased venous pressure resulting from venous reflux or obstruction. This process is thought to be the primary underlying mechanism for ulcer formation.10 Valve dysfunction, outflow obstruction, arteriovenous malformation, and calf muscle pump failure contribute to the pathogenesis of venous hypertension.8 Factors associated with venous incompetence are age, sex, family history of varicose veins, obesity, phlebitis, previous leg injury, and prolonged standing or sitting posture.11 Venous ulcers result from a complex process secondary to increased pressure (venous hypertension) and inflammation within the venous circulation, vein wall, and valve leaflet with extravasation of inflammatory cells and molecules into the interstitium.12
Clinical Presentation and Diagnosis
Clinical history, presentation, and physical examination findings help differentiate venous ulcers from other lower extremity ulcers (Table 15 ). History of superficial or deep venous thrombosis, pulmonary embolism, and ulcer recurrence should be ascertained with comorbid conditions. Venous ulcers typically have an irregular shape and well-defined borders.3 Reported symptoms often include limb heaviness, pruritus, pain, and edema that worsens throughout the day and improves with elevation.5 During physical examination, signs of venous disease, such as varicose veins, edema, or venous dermatitis, may be present. Other findings suggestive of venous ulcers include location over bony prominences such as the gaiter area (over the medial malleolus; Figure 1), telangiectasias, corona phlebectatica (abnormally dilated veins around the ankle and foot), atrophie blanche (atrophic, white scarring; Figure 2), lipodermatosclerosis (Figure 3), and inverted champagne-bottle deformity of the lower leg.1,5
TABLE 1. Common Lower Extremity Ulcers
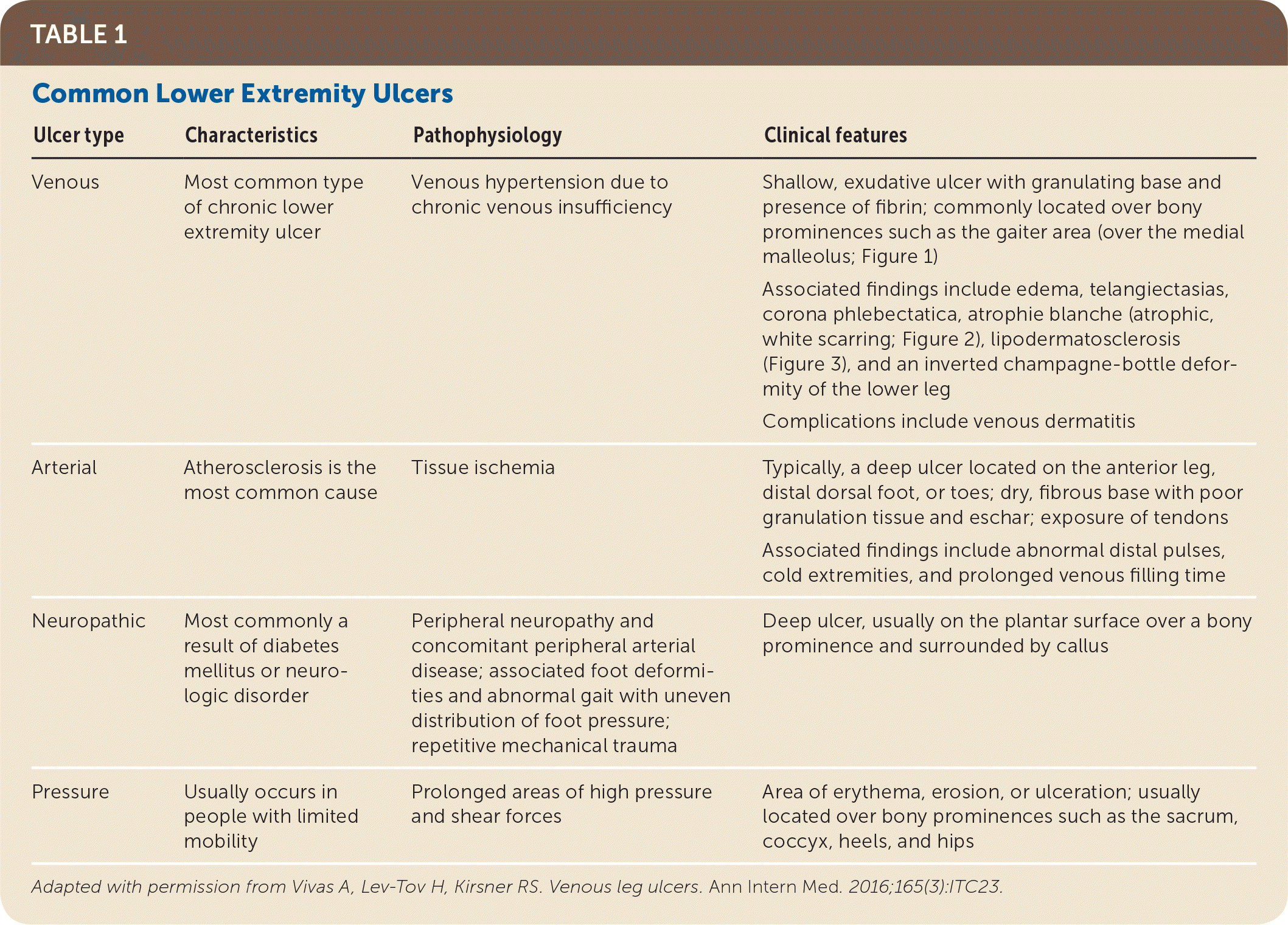
| Ulcer type | Characteristics | Pathophysiology | Clinical features |
|---|---|---|---|
| Venous | Most common type of chronic lower extremity ulcer | Venous hypertension due to chronic venous insufficiency | Shallow, exudative ulcer with granulating base and presence of fibrin; commonly located over bony prominences such as the gaiter area (over the medial malleolus; Figure 1) |
| Associated findings include edema, telangiectasias, corona phlebectatica, atrophie blanche (atrophic, white scarring; Figure 2), lipodermatosclerosis (Figure 3), and an inverted champagne-bottle deformity of the lower leg | |||
| Complications include venous dermatitis | |||
| Arterial | Atherosclerosis is the most common cause | Tissue ischemia | Typically, a deep ulcer located on the anterior leg, distal dorsal foot, or toes; dry, fibrous base with poor granulation tissue and eschar; exposure of tendons |
| Associated findings include abnormal distal pulses, cold extremities, and prolonged venous filling time | |||
| Neuropathic | Most commonly a result of diabetes mellitus or neurologic disorder | Peripheral neuropathy and concomitant peripheral arterial disease; associated foot deformities and abnormal gait with uneven distribution of foot pressure; repetitive mechanical trauma | Deep ulcer, usually on the plantar surface over a bony prominence and surrounded by callus |
| Pressure | Usually occurs in people with limited mobility | Prolonged areas of high pressure and shear forces | Area of erythema, erosion, or ulceration; usually located over bony prominences such as the sacrum, coccyx, heels, and hips |
Adapted with permission from Vivas A, Lev-Tov H, Kirsner RS. Venous leg ulcers. Ann Intern Med. 2016;165(3):ITC23.
FIGURE 1
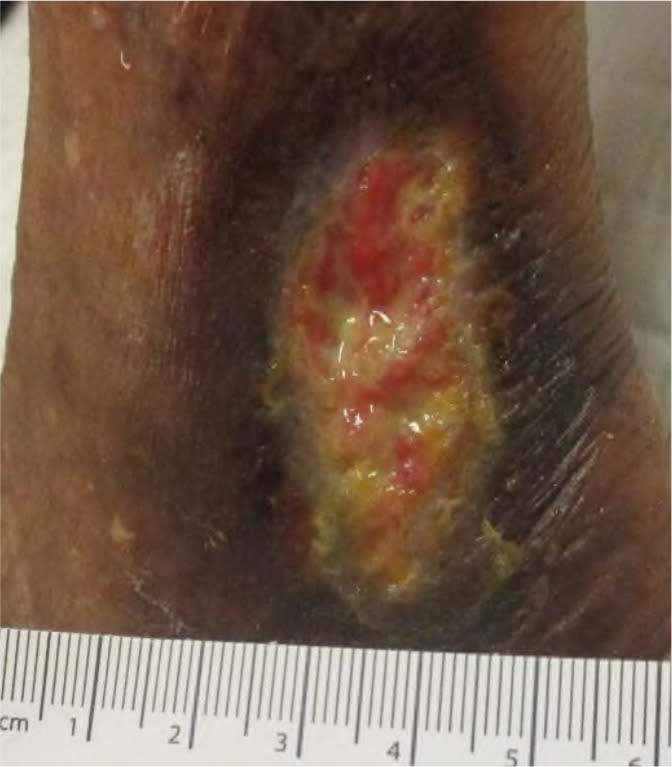
Venous ulcer over the medial malleolus.
FIGURE 2
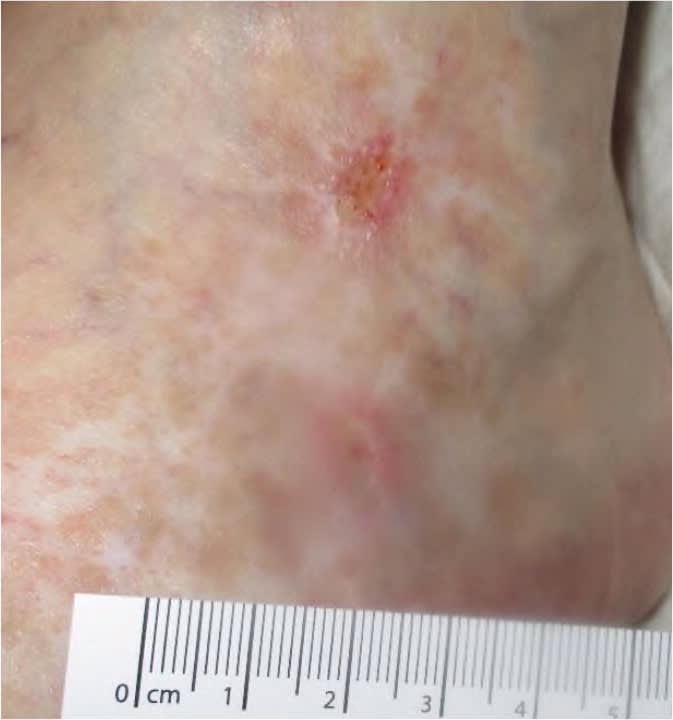
Atrophie blanche (atrophic, white scarring) in a patient with a venous ulcer.
FIGURE 3
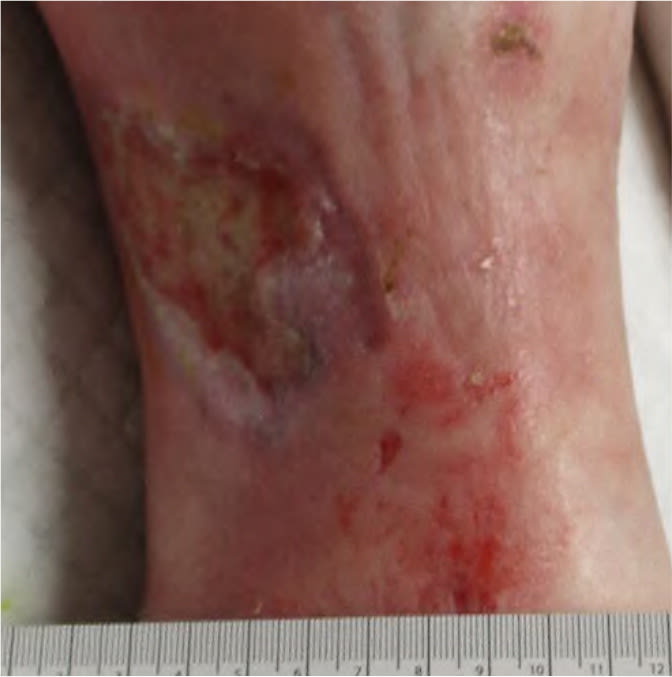
Lipodermatosclerosis with an inverted champagne-bottle deformity in a patient with a venous ulcer.
DIFFERENTIAL DIAGNOSIS
Although venous ulcers are the most common type of chronic lower extremity ulcers, the differential diagnosis should include arterial occlusive disease (or a combination of arterial and venous disease), ulceration caused by diabetic neuropathy, malignancy, pyoderma gangrenosum, and other inflammatory ulcers.13 Among chronic ulcers refractory to vascular intervention, 20% to 23% may be caused by vasculitis, sickle cell disease, pyoderma gangrenosum, calciphylaxis, or autoimmune disease.14
Initial noninvasive imaging with comprehensive venous duplex ultrasonography, arterial pulse examination, and measurement of ankle-brachial index is recommended for all patients with suspected venous ulcers.1 Color duplex ultrasonography is recommended to assess for deep and superficial venous reflux and obstruction.1,15 Because standard therapy for venous ulcers can be harmful in patients with ischemia, additional ultrasound evaluation to assess arterial blood flow is indicated when the ankle-brachial index is abnormal and in the presence of certain comorbid conditions such as diabetes mellitus, chronic kidney disease, or other conditions that lead to vascular calcification.1 Further evaluation with biopsy or referral to a subspecialist is warranted if ulcer healing stalls or the ulcer has an atypical appearance.1,5
Treatment
Treatment options for venous ulcers include conservative management, mechanical modalities, medications, advanced wound therapy, and surgical options. Although the main goal of treatment is ulcer healing, secondary goals include reduction of edema and prevention of recurrence. Table 2 includes treatment options for venous ulcers.16–22
TABLE 2. Recommended Treatment Options for Venous Ulcers

| Treatment option | Comments | |
|---|---|---|
| Conservative management | ||
| Compression therapy | Standard care; recommended for at least one hour per day at least six days per week to prevent recurrence16,17 | |
| Dressings | Recommended to cover ulcers and promote moist wound healing18 | |
| Medications | ||
| Antibiotics | Oral antibiotic treatment is warranted if infection is suspected1 | |
| Pentoxifylline | Improves healing with or without compression therapy19 | |
| Debridement | May benefit ulcer healing20 | |
| Surgical management | ||
| Endovenous ablation | Early endovenous ablation to correct superficial venous reflux may increase healing rates and prevent recurrence21 | |
| Skin grafting | Primary therapy for large ulcers (larger than 25 cm2 [3.9 in2]) or secondary therapy for ulcers that do not heal with standard care22 | |
DEBRIDEMENT
Removal of necrotic tissue by debridement has been used to expedite wound healing. Debridement may be sharp, enzymatic, mechanical, larval, or autolytic. Two prospective randomized controlled trials reviewed the effect of sharp debridement on the healing of venous ulcers. Patients treated with debridement at each physician's office visit had significant reduction in wound size compared with those not treated with debridement.20
Enzymatic debridement using collagenase has been shown to effectively remove nonviable tissue. There is no evidence that collagenase is superior to sharp debridement.23 Larval therapy using maggots is an effective method of debridement with added potential for disinfection, stimulation of healing, and biofilm inhibition and eradication.24,25 Potential barriers to larval debridement include patient acceptability and pain.26,27 Autolytic debridement uses moisture-retentive dressings and may be used in addition to other forms of debridement. Mechanical debridement (wet-to-dry dressings, pulsed lavage, whirlpool) has fallen out of favor.
COMPRESSION THERAPY
Compression therapy is a standard treatment modality for initial and long-term treatment of venous ulcers in patients without concomitant arterial disease.1,28 Goals of compression therapy include reduced edema and pain, improved venous reflux, and enhanced healing.16 Compression therapy is useful for ulcer healing and prevention of recurrence. Multicomponent compression systems comprised of various layers are more effective than single-component systems, and elastic systems are more effective than nonelastic systems.28
Important barriers to the use of compression therapy include wound drainage, pain, application or donning difficulty, physical impairment (weakness, obesity, decreased range of motion), and leg shape deformity (leading to compression material rolling down the leg or wrinkling). Contraindications to compression therapy include significant arterial insufficiency and uncompensated congestive heart failure.29
Elastic. Elastic compression bandages conform to the size and shape of the leg, accommodate to changes in leg circumference, provide sustained compression during rest and walking, have absorptive capacity, and require infrequent changes (about once a week).5 There is strong evidence for the use of multiple elastic layers vs. single layers to increase ulcer healing.30
Inelastic. Inelastic compression wraps, most commonly zinc oxide–impregnated Unna boots, provide high compression only during ambulation and muscle contraction. They should not be used in nonambulatory patients or in those with arterial compromise. Unna boots have limited capacity for fluid absorption in patients with highly exudative ulcers and are best used for early, small, dry ulcers and for venous dermatitis because of the skin soothing effects of zinc oxide.30
Stockings. Compression stockings can be used for ulcer healing and prevention of recurrence (recommended strength is at least 20 to 30 mm Hg, but 30 to 40 mm Hg is preferred).31,32 Donning stockings over dressings can be challenging. Choice of knee-high, thigh-high, toes-in, or toes-out compression stockings depends on patient preference. Compression stockings are removed at night and should be replaced every six months because of loss of compression with regular washing. Once the ulcer has healed, continued use of compression stockings is recommended indefinitely. For patients who have difficulty donning the stockings, use of layered (additive) compression stockings; stockings with a Velcro or zippered closure; or donning aids, such as a donning butler (Figure 4), may be helpful.33
FIGURE 4.

Compression stocking donning butler.
Intermittent Pneumatic Compression. Intermittent pneumatic compression may be considered when there is generalized, refractory edema from venous insufficiency; lymphatic obstruction; and significant ulceration of the lower extremity. Although intermittent pneumatic compression is more effective than no compression, its effectiveness compared with other forms of compression is unclear. Intermittent pneumatic compression may improve ulcer healing when added to layered compression.30,34
LEG ELEVATION
Although leg elevation can increase deep venous flow and reduce venous pressure, leg elevation added to compression may not improve ulcer healing.17 However, one prospective study found that leg elevation for at least one hour per day at least six days per week can reduce venous ulcer recurrence when used with compression.17,35
EXERCISE
A systematic review evaluating progressive resistance exercise, resistance exercise plus prescribed physical activity, only walking, and only ankle exercises found that progressive resistance exercise with prescribed physical activity may result in an additional nine to 45 venous ulcers healed per 100 patients.36
DRESSINGS
Dressings are recommended to cover ulcers and promote moist wound healing.1,18 Dressings should be chosen based on wound location, size, depth, moisture balance, presence of infection, allergies, comfort, odor management, ease and frequency of dressing changes, cost, and availability. Evidence has not shown that any one dressing is superior when used with appropriate compression therapy.18 Types of dressings are summarized in Table 3.37
TABLE 3. Types of Dressings Used for Venous Ulcers
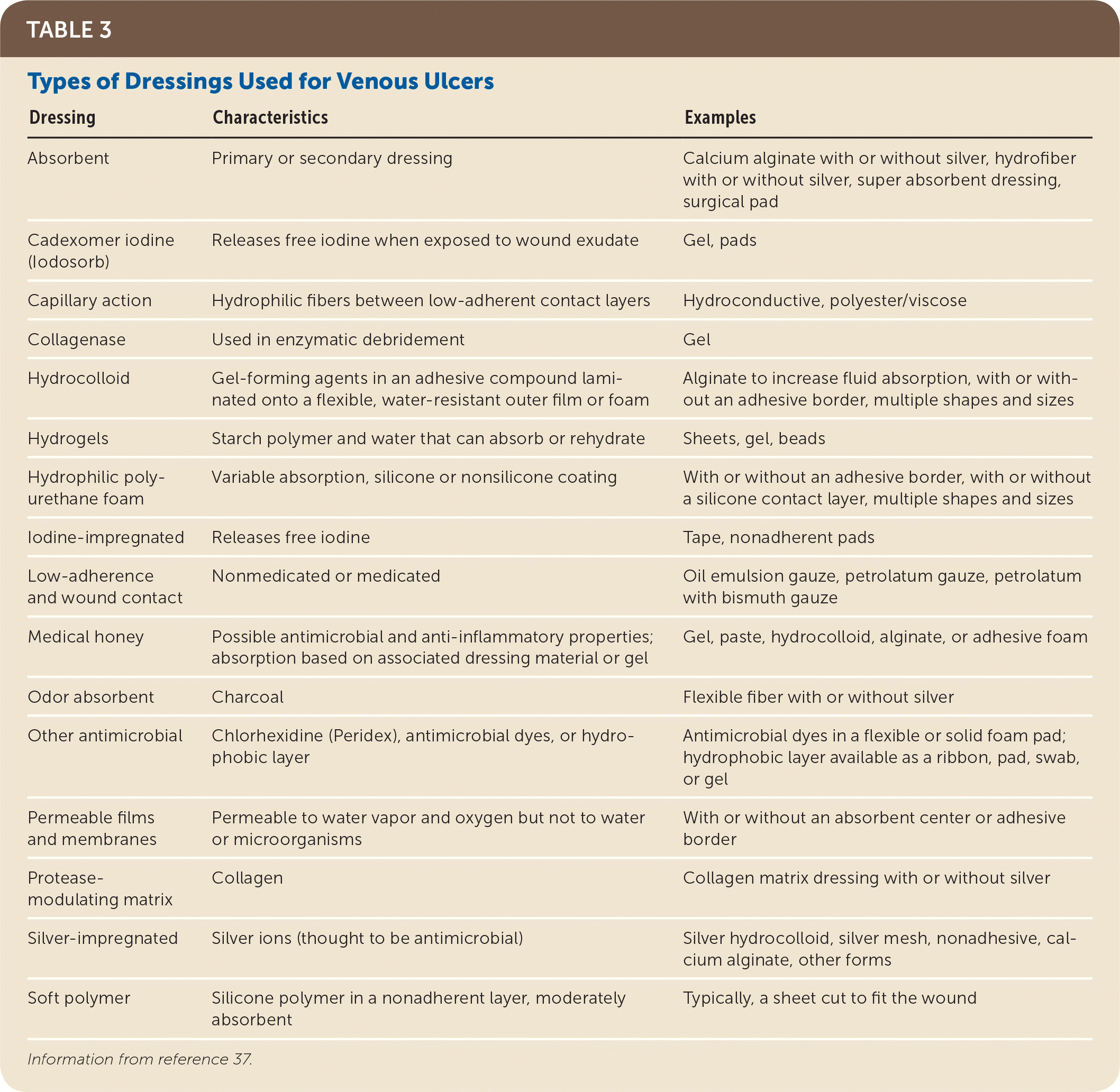
| Dressing | Characteristics | Examples |
|---|---|---|
| Absorbent | Primary or secondary dressing | Calcium alginate with or without silver, hydrofiber with or without silver, super absorbent dressing, surgical pad |
| Cadexomer iodine (Iodosorb) | Releases free iodine when exposed to wound exudate | Gel, pads |
| Capillary action | Hydrophilic fibers between low-adherent contact layers | Hydroconductive, polyester/viscose |
| Collagenase | Used in enzymatic debridement | Gel |
| Hydrocolloid | Gel-forming agents in an adhesive compound laminated onto a flexible, water-resistant outer film or foam | Alginate to increase fluid absorption, with or without an adhesive border, multiple shapes and sizes |
| Hydrogels | Starch polymer and water that can absorb or rehydrate | Sheets, gel, beads |
| Hydrophilic polyurethane foam | Variable absorption, silicone or nonsilicone coating | With or without an adhesive border, with or without a silicone contact layer, multiple shapes and sizes |
| Iodine-impregnated | Releases free iodine | Tape, nonadherent pads |
| Low-adherence and wound contact | Nonmedicated or medicated | Oil emulsion gauze, petrolatum gauze, petrolatum with bismuth gauze |
| Medical honey | Possible antimicrobial and anti-inflammatory properties; absorption based on associated dressing material or gel | Gel, paste, hydrocolloid, alginate, or adhesive foam |
| Odor absorbent | Charcoal | Flexible fiber with or without silver |
| Other antimicrobial | Chlorhexidine (Peridex), antimicrobial dyes, or hydrophobic layer | Antimicrobial dyes in a flexible or solid foam pad; hydrophobic layer available as a ribbon, pad, swab, or gel |
| Permeable films and membranes | Permeable to water vapor and oxygen but not to water or microorganisms | With or without an absorbent center or adhesive border |
| Protease-modulating matrix | Collagen | Collagen matrix dressing with or without silver |
| Silver-impregnated | Silver ions (thought to be antimicrobial) | Silver hydrocolloid, silver mesh, nonadhesive, calcium alginate, other forms |
| Soft polymer | Silicone polymer in a nonadherent layer, moderately absorbent | Typically, a sheet cut to fit the wound |
Information from reference 37.
Topical antiseptics, including cadexomer iodine (Iodosorb), povidone-iodine (Betadine), peroxide-based preparations, honey-based preparations, and silver, have been used to treat venous ulcers. Some evidence supports the use of cadexomer iodine to improve healing of venous ulcers, but evidence for other agents is lacking.38
MEDICATIONS
Pentoxifylline. This hemorheologic agent affects microcirculation and oxygenation, and can be used effectively as monotherapy or with compression therapy for venous ulcers.1,39 In seven randomized controlled trials, pentoxifylline plus compression improved healing of venous ulcers compared with placebo plus compression. Four trials showed that pentoxifylline alone improved healing compared with placebo alone.19 Common adverse effects of pentoxifylline include nausea, gastrointestinal discomfort, headaches, dizziness, and prolonged bleeding time.
Aspirin. There is inconsistent evidence concerning the benefits and harms of oral aspirin in the treatment of venous ulcers.40,41
Statins. Statins have vasoactive and anti-inflammatory effects. In a small study, patients receiving simvastatin (Zocor), 40 mg once daily, had a higher rate of ulcer healing than matched patients given placebo.42
Phlebotonics. These venoactive drugs theoretically work by improving venous tone and decreasing capillary permeability. Common drugs in this class include saponins (e.g., horse chestnut seed extract), flavonoids (e.g., rutosides, diosmin, hesperidin), and micronized purified flavonoid fraction.43 Although phlebotonics may improve edema and other signs and symptoms of chronic venous insufficiency (e.g., trophic disorders, cramps, restless legs, swelling, paresthesia), there was no difference in venous ulcer healing when compared with placebo.44
Antibiotics. Because bacterial colonization and infection may contribute to poor healing, systemic antibiotics are often used to treat venous ulcers. Colonization refers to the presence of replicating bacteria without a host reaction or clinical signs of infection.45 Colonized venous ulcers generally should not be treated with antibiotics. Infection occurs when microorganisms invade tissues, leading to systemic and/or local responses such as changes in exudate, increased pain, delayed healing, leukocytosis, increased erythema, or fevers and chills.46 Venous ulcers with obvious signs of infection should be treated with antibiotics. Oral antibiotics are preferred, and therapy should be limited to two weeks unless evidence of wound infection persists.1
Hyperbaric Oxygen Therapy. A systematic review of hyperbaric oxygen therapy in patients with chronic wounds found only one trial that addressed venous ulcers. Because of limited evidence and no long-term benefit, hyperbaric oxygen therapy is not recommended for treatment of venous ulcers.47
Negative Pressure Wound Therapy. Traditional negative pressure wound therapy systems are bulky and cannot be used with compression therapy. Negative pressure wound therapy is not recommended as a primary treatment of venous ulcers.48 There may be a future role for newer, ultraportable, single-use systems that can be used underneath compression devices.49,50
ADVANCED THERAPIES
Venous ulcers that do not improve within four weeks of standard wound care should prompt consideration of adjunctive treatment options.51
Cellular and Tissue-Based Products. There are many cellular and tissue-based products approved for the treatment of refractory venous ulcers, including allografts, animal-derived extracellular matrix products, human-derived cellular products, and human amniotic membrane–derived products. Compared with compression plus a simple dressing, one study showed that advanced therapies can shorten healing time and improve healing rates.52
Skin Grafting. Skin grafting should be considered as primary therapy only for large venous ulcers (larger than 25 cm2 [3.9 in2]), in which healing is unlikely without grafting. It can be used as secondary therapy for ulcers that do not heal with standard care.22
ENDOVENOUS INTERVENTION
Like conservative therapies, the goal of operative and endovascular management of venous ulcers (i.e., endovenous ablation, ligation, subfascial endoscopic perforator surgery, and sclerotherapy) is to improve healing and prevent ulcer recurrence. Historically, trials comparing venous intervention plus compression with compression alone for venous ulcers showed that surgery reduced recurrence but did not improve healing.53 Recent trials show faster healing of venous ulcers when early endovenous ablation to correct superficial venous reflux is performed in conjunction with compression therapy, compared with compression alone or with delayed intervention if the ulcer did not heal after six months.21 The most common complications of endovenous ablation were pain and deep venous thrombosis.21
Prevention of Recurrence
The recurrence rate of venous ulcers has been reported as high as 70%.35 Venous intervention and long-term use of compression stockings are important for preventing recurrence, and leg elevation can be beneficial when used with compression stockings. Exercise should be encouraged to improve calf muscle pump function.35,54 Good social support and self-efficacy have also been shown to help prevent venous ulcer recurrence.35
This article updates a previous article on this topic by Collins and Seraj.55
Data Sources: We conducted searches in PubMed, Essential Evidence Plus, the Cochrane database, and Google Scholar using the key terms venous ulcers and venous stasis ulcers. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. Search dates: November 12, 2018; December 27, 2018; January 8, 2019; and March 21, 2019.

