Urinary tract infections (UTIs) are among the most common bacterial infections encountered by primary care physicians. Although UTIs do not occur with as great a frequency in children as in adults, they can be a source of significant morbidity in children. For reasons that are not yet completely understood, a minority of UTIs in children progress to renal scarring, hypertension and renal insufficiency. Clinical presentation of UTI in children may be nonspecific, and the appropriateness of certain diagnostic tests remains controversial. The diagnostic work-up should be tailored to uncover functional and structural abnormalities such as dysfunctional voiding, vesicoureteral reflux and obstructive uropathy. A more aggressive work-up, including renal cortical scintigraphy, ultrasound and voiding cystourethrography, is recommended for patients at greater risk for pyelonephritis and renal scarring, including infants less than one year of age and all children who have systemic signs of infection concomitant with a UTI. Antibiotic prophylaxis is used in patients with reflux or recurrent UTI who are at greater risk for subsequent infections and complications.
Urinary tract infection (UTI) is defined as the presence of bacteria in urine along with symptoms of infection. UTIs occur in as many as 5 percent of girls and 1 to 2 percent of boys.1 The incidence of UTI in infants ranges from approximately 0.1 to 1.0 percent in all newborn infants to as high as 10 percent in low-birth-weight infants.2 Infection of the urinary tract before age one occurs more frequently in boys than in girls.2 After age one, both bacteriuria and UTI are more common in girls.
In preschool-age children, the prevalence of asymptomatic infections diagnosed by supra-pubic aspiration in girls is 0.8 percent, compared with 0.2 percent in boys.3 In the school-age group, the incidence of bacteriuria among girls is 30 times that among boys (1.2 versus 0.04 percent).4
Etiology and Pathogenesis
Escherichia coli is the most common infecting pathogen in children, accounting for up to 80 percent of UTIs. Other pathogens include Staphylococcus and Streptococcus species, a variety of enterobacteria (e.g., Klebsiella, Proteus) and, occasionally, Candida albicans. The virulence of the invading bacteria and the susceptibility of the host are of primary importance in the development of UTI.3 In neonates, the usual route of infection is presumed to be hematogenous.1 Later in life, infection is usually caused by ascension of bacteria into the urinary tract.5 Any condition that leads to urinary stasis (renal calculi, obstructive uropathy, vesicoureteral reflux and voiding disorders) may predispose to the development of UTI in children.5 Renal parenchymal infection and scarring are well-established complications of infection of the upper urinary tract in children and can lead to renal insufficiency, hypertension and renal failure. Parenchymal scarring develops in 10 to 15 percent of children with UTI. Children less than one year of age with a UTI are at much greater risk for renal scarring than older children; children over five years of age uncommonly have new renal scarring with UTI.6 A 27-year follow-up study from Sweden1 showed that focal renal scarring caused by pyelonephritis in a child carried a 23 percent risk for hypertension and a 10 percent risk for end-stage renal disease.
Controversy continues regarding the association of vesicoureteral reflux with the pathogenesis of renal scarring, reflux nephropathy, pyelonephritis and voiding disorders. Although vesicoureteral reflux is associated with renal scarring,7 its role in the pathogenesis of pyelonephritis and renal scarring is not fully understood.8 Findings from one study9 showed that scars formed in 40 percent of refluxing kidneys and 43 percent of nonrefluxing kidneys. While some researchers emphasize the risk of renal scarring from recurrent UTI without reflux,10 others are just as adamant regarding the risk of scarring from reflux in the absence of infection.11 The fact that renal scarring develops in only a minority of patients with pyelonephritis and/or vesicoureteral reflux suggests that the development of renal scarring likely involves the interplay of several factors and cannot simply be attributed to the presence of infection or reflux alone (Figure 1).
FIGURE 1.
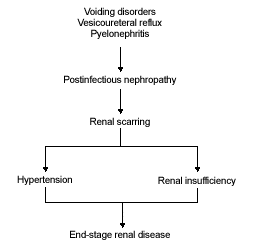
Relationship between urinary tract infection and loss of renal function.
Clinical Presentation
The clinical presentation of UTI is variable. In a child with so-called “asymptomatic” bacteriuria, only subtle clues, such as enuresis or squatting, may be present. Alternatively, a systemically ill neonate may be lethargic and hypotensive (Table 1). Although children are often managed on the basis of clinical symptoms and signs alone, these may be unreliable predictors of which patients are at risk for pyelonephritis and scarring.12,13 On the other hand, radiologic tests to confirm pyelonephritis or reflux can be expensive, time-consuming, invasive and undesirable to parents and physicians.14
TABLE 1 Signs and Symptoms of Urinary Tract Infection in Children
| Urinary tract signs and symptoms |
| Dysuria |
| Frequency |
| Dribbling/hesitancy |
| Enuresis after successful toilet training |
| Malodorous urine |
| Hematuria |
| Squatting |
| Abdominal/suprapubic pain |
| Systemic signs and symptoms |
| Fever |
| Vomiting/diarrhea |
| Flank/back pain |
The physical examination of a child with a possible UTI should exclude hypertension, an abdominal or flank mass, or a palpable bladder, neurologic deficits, abnormal genitalia and an abnormal urinary stream.1 This will help the clinician to find associated disorders.
The presence of irritative urinary symptoms in the absence of bacteria suggests a non-UTI cause such as vaginitis, urethritis, pinworms, or the use of bubble baths.1
Diagnosis
Maintaining a high index of suspicion for UTI in febrile children, particularly when an unexplained fever lasts two to three days, will lessen the number of missed UTIs. The most recent guideline issued by the American Academy of Pediatrics (AAP) for the evaluation of fever (39.0°C [102.2°F] or higher) of unknown origin suggests urinalysis in all cases and a urine culture in all boys younger than six months of age and all girls younger than two years of age.15
In infants, suprapubic aspiration or bladder catheterization and, in older children, a clean-voided midstream specimen are essential for diagnosis of UTI.1 Although convenient, use of adhesive perineal bags or wringing liquid from a wet diaper to collect urine is suboptimal, as bacteria from fecal contamination or urethral colonization may be misinterpreted as UTI. Although there is debate about the best way to screen female infants for UTI,16 many support criteria set by Dagan and colleagues.17 According to these criteria, a finding of more than 5 white blood cells per high-power field in centrifuged fresh urine is a satisfactory positive screening test.
Pyuria, proteinuria and hematuria may occur with or without UTI.1 Conversely, UTI can occur without pyuria.1 The determinations of nitrite concentrations and leukocyte esterase are not sensitive enough in children to indicate the need for urine culture.1 A properly obtained positive urine culture is essential for the diagnosis of UTI. Any number of colonies from a suprapubic bladder aspiration, more than 103 colonies from an intermittent (“in-and-out”) catheterization, and more than 105 colonies from a midstream clean-catch urine collection indicate UTI.5
Most UTIs are caused by a single organism; the presence of two or more organisms usually suggests contamination. A urine culture is not mandatory in adolescent girls, particularly with a first episode. With recurrent episodes, episodes that fail therapy and in girls with pyuria without bacteriuria, a culture is recommended.
Special Issues
Recurrent UTI
Recurrent UTI is defined as two or more UTIs over a six-month period.7 It is useful to determine whether recurrence is caused by inadequate treatment of an unrecognized anatomic site of bacterial persistence (small infected calculus or unrecognized anatomic abnormality).14 As mentioned previously, recurrent UTI increases the risk of subsequent renal scarring.
Vesicoureteral Reflux
Vesicoureteral reflux is the abnormal backwash of urine into the ureter or kidney.18 The most common radiologic studies for the evaluation of reflux are the voiding cystourethrogram and the isotope cystogram. The isotope cystogram is more sensitive than the voiding cystourethrogram for detecting reflux, while only the voiding cystourethrogram provides enough anatomic detail to identify the severity of reflux and the presence of anatomic abnormalities.
Because the isotope cystogram exposes the patient to less radiation than the voiding cystourethrogram, it may be the study of choice for follow-up evaluations and may be used as the initial study in girls.18 In boys, however, initial work-up should include a voiding cystourethrogram to detect urethral abnormalities such as urethral diverticulum or posterior urethral valves. Grades I and II reflux can be treated with antimicrobial prophylaxis along with a strict voiding regimen19; however, urologic consultation should be considered in grades III to V reflux as the condition may merit surgical correction.5
Breakthrough UTI
Breakthrough UTI may be caused by a change in the resistance pattern of organisms colonizing the urethra, noncompliance, vesicoureteral reflux or voiding dysfunction. Recognizing and addressing these associated factors are essential in treating breakthrough UTI. A study in girls showed that treatment of voiding dysfunction combined with double antimicrobial prophylaxis was significantly successful in preventing breakthrough UTI.20
Voiding Dysfunction
Voiding dysfunction is a general term encompassing several patterns of detrusor instability and incomplete bladder emptying seen on urodynamic testing. It is often associated with daytime enuresis and constipation.20 Patients with otherwise unexplained recurrent UTI, especially in the setting of daytime enuresis or constipation, may merit urodynamic testing. Children with voiding dysfunction are at increased risk for the development of vesicoureteral reflux and UTI. Treatment of voiding dysfunction includes timed voiding, treatment of constipation, prophylactic antibiotics and, in some cases, use of anticholinergic medication (e.g., oxybutynin [Ditropan] or propantheline [Pro-Banthine]) or biofeedback.
Asymptomatic Bacteriuria
Controversy continues regarding the need for antibiotic treatment of asymptomatic bacteriuria.21–25 If recurrent bacteriuria is truly asymptomatic, no antimicrobial treatment may be the best option, as some studies have shown that asymptomatic children are at very low risk of renal scarring, and prophylactic treatment did not decrease the risk of UTI recurrence.14
Diagnostic Imaging
There is more controversy than consensus regarding the appropriateness of different diagnostic imaging modalities in the evaluation of UTI in children.26–28 The most commonly used imaging techniques are discussed in the following sections.
Ultrasonography
Although intravenous urography has been a time-honored examination in the initial radiologic evaluation of UTI in children,9 ultrasonography has largely replaced intravenous urography as the initial screening examination.8 Ultrasonography alone is not generally adequate for investigation of UTI in children, as it is unreliable in detecting vesicoureteral reflux, renal scarring or inflammatory changes.29 If reflux or morphologic abnormalities are identified, renal scintigraphy and voiding cystourethrography are recommended to further search for renal scarring or urinary tract abnormalities.
Intravenous Urography
Intravenous urography provides a precise anatomic image of the kidneys and can readily identify some urinary tract abnormalities (e.g., cysts, hydronephrosis).30 The major disadvantages of intravenous urography include decreased sensitivity compared with renal scintigraphy in the detection of both pyelonephritis and renal scarring.30 Higher dosage of radiation and risk of reaction to contrast medium are also reasons for concern. Given these disadvantages, intravenous urography appears to have little role in the work-up of UTI in children.
Renal Cortical Scintigraphy
Renal cortical scintigraphy has replaced intravenous urography as the standard technique for the detection of renal inflammation and scarring.8 Renal cortical scintigraphy with either technetium-99m–labeled glucoheptonate or dimercaptosuccinic acid (DMSA) are both highly sensitive and specific.8 DMSA scanning offers the advantages of earlier detection of acute inflammatory changes and permanent scars compared with ultrasound or intravenous urography. It is also useful in neonates and patients with poor renal function. Computed tomography (CT) is sensitive and specific for the detection of acute pyelonephritis, but no study is available that compares CT and scintigraphy.8 Furthermore, CT is more expensive than scintigraphy and exposes the patient to higher levels of radiation, and its use is not supported by evidence.
Voiding Cystourethrography
Because vesicoureteral reflux is a risk factor for reflux nephropathy and renal scars, early identification of this condition is warranted.4 Voiding cystourethrography should be delayed until after urinary infection is controlled, because vesicoureteral reflux may be the transient effect of infection. However, because of low sensitivity and specificity, and because voiding cystourethrography involves gonadal irradiation and catheterization, its use in diagnosing vesicoureteral reflux has been questioned.31
Isotope Cystogram
Although the isotope cystogram causes the same discomfort as bladder catheterization used in voiding cystourethrography, it has the advantage of an ionization radiation dose that is only 1 percent of that used for voiding cystourethrography,5 and its continuous monitoring is also more sensitive for identifying reflux than the intermittent flouroscopic monitoring of voiding cystourethrography.
Table 2 reviews the medical imaging techniques used in evaluating UTI in children.
TABLE 2 Advantages and Disadvantages of Diagnostic Imaging in Evaluation of Urinary Tract Infection in Children
| Imaging study | Advantages | Disadvantages |
|---|---|---|
| Ultrasound | Measures renal size and shape | Not reliable to detect vesicoureteral reflux, renal scarring or inflammatory changes |
| Identifies hydronephrosis, structural or anatomic abnormalities and renal calculi | ||
| No radiation | ||
| Intravenous urography | Precise anatomic image of the kidneys | Not as reliable to detect renal scarring or pyelonephritis |
| Estimates renal function | High radiation dose | |
| Risk of reaction to contrast medium | ||
| Poor detail in infants | ||
| Renal cortical scintigraphy | Detects pyelonephritis and renal scarring even in early stages | Does not evaluate collecting system |
| Cannot detect obstruction | ||
| Useful in neonates | ||
| Little radiation | ||
| Useful in patients with poor renal function | ||
| Computed tomography | Provides both anatomic and functional information about the kidney Possibly more sensitive in diagnosing pyelonephritis | Expensive |
| High radiation | ||
| Few clinical or experimental data to support its use at present | ||
| Voiding cystourethrography | Assesses the size and shape of bladder | Gonadal radiation |
| Detects and grades vesicoureteral reflux | Catheterization | |
| Evaluates posterior urethral anomalies in boys |
Treatment
Therapeutic trials in children with UTI are rare and poorly controlled.32 Thus, controversy regarding dosage or length of therapy with antimicrobials continues. In patients who appear toxic, it is reasonable to initiate treatment with intravenous antibiotics and follow them closely for signs and symptoms of infection (fever, severe pain); these usually resolve in three to five days.33 Initial antibiotic therapy should be based on age, clinical severity, location of infection, presence of structural abnormalities, and allergy to certain antibiotics. Treatment generally begins with a broad-spectrum antibiotic, but it may need to be changed based on the results of urine culture and sensitivity testing.
Hospitalization is suggested for symptomatic young infants (less than three months of age) and all children with clinical evidence of acute severe pyelonephritis (high fever, toxic appearance, severe flank pain).33
The duration of outpatient treatment for patients with a less toxic appearance and uncomplicated UTI (no systemic signs of infection) is also controversial.1 Evidence is lacking for the use of short-course therapy in children with UTI.26 Although conventional therapy lasts seven to 10 days, a three- to seven-day trial of oral antibiotics has been suggested for uncomplicated infection of the lower urinary tract.32
Reasonable choices for initial inpatient and outpatient oral antibiotic therapy are shown in Table 3.24 Because of the possibility of bacterial resistance to a prophylactic agent used for long-term suppression, the treating anti-microbial agent for a breakthrough UTI should, ideally, be different from the prophylactic agent17 (Table 3).
Follow-up and Chemoprophylaxis
A urine culture should be obtained three to seven days after the completion of treatment to exclude relapse. Prophylaxis is recommended for all children younger than five years of age with vesicoureteral reflux (who are not surgical candidates) or other structural abnormalities and in children who have had three documented UTIs in one year.1 With careful monitoring for side effects, a prophylactic trial of a single nightly dose of nitrofurantoin (Furadantin, Macrodantin), 1 to 2 mg per kg per day, or trimethoprim-sulfamethoxazole (Bactrim, Septra), 2 mg per kg of trimetho-prim per day, may be used for six months or more.8 Using low doses of antibiotics for prophylaxis has a theoretic advantage since this may minimize serum levels and subsequent enteric bacterial resistance while urinary concentration of the antibiotic remains high enough to maintain sterile urine.14
Prevention/Patient Education
A common-sense approach to prevention is advised by most authors.24,34 Good hygiene (including “front-to-back” wiping after urination in girls), avoidance or correction of constipation, and avoidance of bubble baths, chemical irritants and tight clothing might be recommended.
The role of circumcision in preventing UTI is controversial.25,35 The AAP states that “newborn circumcision has potential medical benefits and risks.” When circumcision is being considered, the benefits and risks should be explained to the parents, and informed consent should be obtained.36
Final Comment
In light of the controversies and current literature, we propose our outline of management of UTI in children (Figures 2, 2a and 2b).
FIGURE 2.
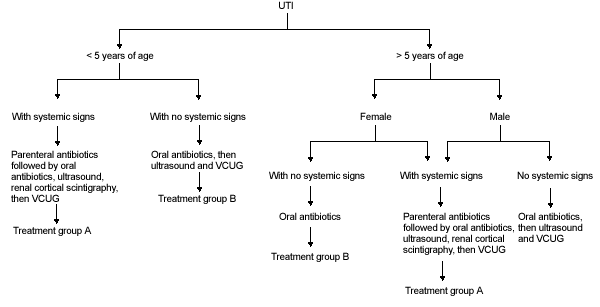
Algorithm for the management of urinary tract infection in children. See Figures 2a and 2b for treatment groups A and B. (UTI = urinary tract infection; VCUG = voiding cystourethrography)
FIGURE 2A.
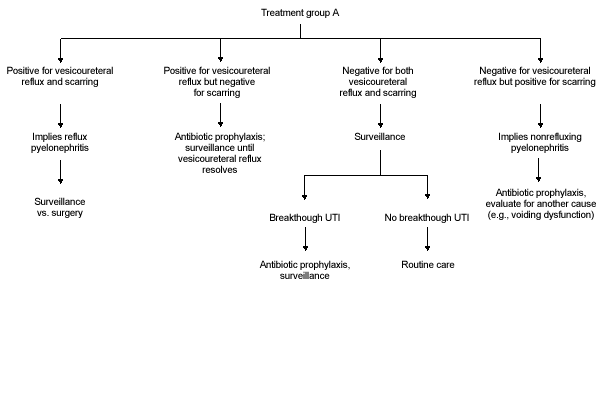
Algorithm for treatment group A.
FIGURE 2B.
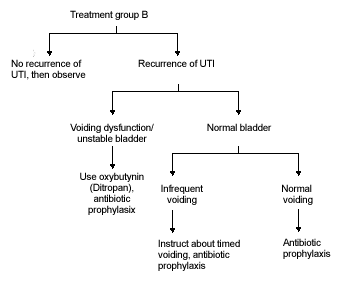
Algorithm for treatment group B.
For a primary care physician, it is imperative to maintain a high index of suspicion for UTI in children. By uncovering UTI and associated disorders, the goal of preventing renal infections, renal insufficiency, hypertension and end-stage renal disease can be realized.
In any child with systemic signs of illness, treatment with parenteral antibiotics should be initiated, and after clinical improvement, therapy should be switched to oral antibiotics for 10 to 14 days. Diagnostic imaging with ultrasound and renal cortical scintigraphy should be considered to document the presence of pyelonephritis or renal scarring; voiding cystourethrography should be performed when the urine is sterile and the patient is clinically improved. Further management is dictated by the clinical course and findings on medical imaging (Figure 2a).
In all patients less than five years of age with no systemic signs and in boys over age five with no systemic signs, treatment with oral antibiotics should be carried out for 10 to 14 days. Afterward, diagnostic imaging with ultrasound and voiding cystourethrogram should be considered. Further management is dictated by findings on diagnostic imaging and clinical course (Figure 2b).
In girls over five years of age with no systemic signs, treatment with oral antibiotics should be carried out for seven to 10 days. Diagnostic imaging in these patients is not necessary with the first UTI but may be indicated in cases of recurrent UTI. Further management is outlined in Figure 2b.

