Tremor is a symptom of many disorders, including Parkinson's disease, essential tremor, orthostatic tremor, cerebellar disease, peripheral neuropathy and alcohol withdrawal. Tremors may be classified as postural, rest or action tremors. Symptomatic treatment is tailored to the tremor type. Combination therapy with carbidopa and levodopa remains the first-line approach for parkinsonian tremor. Essential tremor may be amenable to propranolol or primidone. Propranolol may be useful in treating alcohol withdrawal tremor, and isoniazid may control the cerebellar tremor associated with multiple sclerosis. Clonazepam may relieve orthostatic tremor. Other agents are also available for the treatment of tremor. When medical therapy fails to control the tremor, surgical options such as thalamotomy, pallidotomy and thalamic stimulation should be considered in severe cases. Thalamic stimulation, the most recent of these surgical approaches, offers the advantage over ablative procedures of alleviating tremor without the creation of a permanent lesion.
Tremor is the involuntary, rhythmic oscillation of reciprocally innervated, antagonistic muscle groups, causing movement of a body part about a fixed plane in space.1,2 Effective treatment of tremor requires distinguishing this type of movement disorder from other movement disorders.
Rhythmicity distinguishes tremor from disorders in which tremor may be a component, such as choreoathetosis and dystonia, and its biphasic nature distinguishes tremor from clonus.1 The frequency and amplitude of a tremor vary to the degree that the tremor may be hardly noticeable or severely disabling. Frequency can be divided into three categories of oscillations per second: slow (3 to 5 Hz), intermediate (5 to 8 Hz) or rapid (9 to 12 Hz).3 Amplitude may be classified as fine, medium or coarse, depending on the displacement produced by the tremor about the fixed plane.3 A coarse tremor has a large displacement, whereas a fine tremor is barely noticeable. Tremor may be unifocal, multifocal or generalized, and may affect the head, face, jaw, voice, tongue, trunk or extremities.
Classification: Postural, Rest and Action Tremors
Tremor is primarily classified on the basis of when it occurs, either with a certain posture, at rest or during action (Table 1). A resting tremor occurs when the patient is attempting to maintain the position of a body part at rest (e.g., when the patient's hands exhibit a tremor as they are resting in the patient's lap). Postural tremor is observed when the patient tries to maintain a posture against gravity, such as holding the arms out in front of the body. An action tremor (kinetic or intention tremor) occurs during movement of the affected body part from one point to another. A task-specific tremor occurs only when the patient begins to perform a highly skilled activity, such as writing or speaking.2
TABLE 1 Classification of Tremors, and Their Characteristics and Treatment
| Type of tremor | Frequency | Occurrence | Etiology | Treatment* |
|---|---|---|---|---|
| Postural tremor | 5 to 9 Hz | When limb is positioned against gravity | Physiologic tremor, essential tremor, alcohol or drug withdrawal, metabolic disturbances, drug-induced tremor, psychogenic tremor | Beta blockers, primidone (Mysoline), acetazolamide (Diamox), clonazepam (Klonopin), botulinum toxin, brain gabapentin (Neurontin), deep stimulation, thalamotomy |
| Rest tremor | 3 to 6 Hz | When limb is fully supported against gravity and the muscles are not voluntarily activated | Parkinson's disease, multiple-systems atrophy, progressive supranuclear palsy, drug-induced tremor, rubral tremor, psychogenic tremor | Levodopa–carbidopa (Sinemet), anticholinergics and other antiparkinsonian agents, deep brain stimulation, pallidotomy, thalamotomy |
| Action tremor† | 3 to 10 Hz | During any type of movement | Cerebellar lesions, rubral tremor, psychogenic tremor | Wrist weights, isoniazid |
*—Drugs and other treatments are generally listed in the order in which they should be tried. An adequate trial of each medication must be tried before the agent is judged to be ineffective. Many of these drugs are not specifically labeled for the treatment of tremor or have not undergone extensive studies to support their use in the treatment of tremor.
†—Action tremor includes intention tremor (exacerbation toward the end of goal-directed movement), kinetic tremor (during any type of movement) and task-specific tremor (only during performance of highly skilled activities, such as writing or playing a musical instument).
Tremor may be either physiologic or pathologic. Physiologic tremor is a normal variant, occurring at a frequency of 8 to 12 Hz in the hands yet as slow as 6.5 Hz in other body parts during maintenance of a posture.2,4 It can be increased by emotions such as anxiety, stress or fear, by exercise and fatigue, hypoglycemia, hypothermia, hyperthyroidism and alcohol withdrawal. When such an increase occurs, physiologic tremor is then called enhanced or exaggerated physiologic tremor.1,4 Certain drugs can also exacerbate physiologic tremor5 (Table 2). Pathologic tremor is either idiopathic or occurs secondary to some disorders (Table 3). Essential tremor and parkinsonian tremor are two common types of pathologic tremor.
TABLE 2 Commonly Used Agents That Exacerbate Physiologic Tremor
| Caffeine |
| Fluoxetine (Prozac) |
| Haloperidol (Haldol) |
| Lithium |
| Methylphenidate (Ritalin) |
| Metoclopramide (Reglan) |
| Phenylpropanolamine |
| Pseudoephedrine |
| Theophylline |
| Valproic acid |
TABLE 3 Selected Secondary Causes of Tremor
| Alcohol or drug withdrawal |
| Brain abscess |
| Brain tumor |
| Multiple sclerosis |
| Peripheral neuropathy |
| Pheochromocytoma |
| Psychogenic disorders |
| Thyrotoxicosis |
Identification of the type of tremor depends on keen observation. The location of the tremor or the patient's position when it occurs should be identified first, and special attention must be paid to other signs of illness. Careful observation will reveal if the tremor occurs at rest, during posture maintenance or during movement. The patient should be asked what produces or modulates the amplitude and frequency of the tremor.2,3 A correct diagnosis is essential for proper treatment of the disorder, because different types of tremor require different treatments.
Tremor Types Based on Etiology
PARKINSONIAN TREMOR
The tremor in Parkinson's disease occurs at rest and is characterized by a frequency of 4 to 6 Hz and a medium amplitude. It is classically referred to as a “pill rolling” tremor of the hands but can also affect the head, trunk, jaw and lips.2,3 Although rare, a rest tremor may also be found in patients with other neurodegenerative diseases, such as multiple-systems atrophy and progressive supranuclear palsy. The tremor associated with these disorders is usually symmetric and not as prominent as the tremor that accompanies Parkinson's disease.
Parkinson's disease results from a slow degeneration of a small area in the midbrain, called the substantia nigra. Specifically, excitatory and inhibitory dopaminergic neurons degenerate in the substantia nigra pars compacta. These neurons project to the striatum and then to the globus pallidus. From there, multiple connections in the basal ganglia project to one another, to the thalamus and, finally, to the cortex, which makes up the extrapyramidal system. This system regulates the initiation and control of movement, and dysfunction of any of these connections can lead to various types of movement disorders.6 As a consequence of neuronal degeneration in the substantia nigra pars compacta, the ventral intermediate nucleus of the thalamus becomes overactive, possibly producing the tremor of Parkinson's disease. The neurons in the ventral intermediate nucleus of the thalamus fire at a rate that matches the tremor.7
ESSENTIAL TREMOR
Essential tremor is the most common movement disorder.2,3,8 This postural tremor may have its onset anywhere between the second and sixth decades of life and its prevalence increases with age.8 It is slowly progressive over a period of years.3
The specific pathophysiology of essential tremor remains unknown. Essential tremor occurs sporadically or can be inherited. While the exact genetic defect has not been identified, familial transmission seems to be autosomal dominant with variable penetrance.4
The frequency of essential tremor is 4 to 11 Hz, depending on which body segment is affected. Proximal segments are affected at lower frequencies, and distal segments are affected at higher frequencies.3 Although typically a postural tremor, essential tremor may occur at rest in severe and very advanced cases.2 It most commonly affects the hands but can also affect the head, voice, tongue and legs.2,3,9 In some patients essential tremor is alleviated by small amounts of alcohol, an effect not found in Parkinson's disease.
CEREBELLAR TREMOR
The most common type of cerebellar tremor is kinetic, or goal directed. Cerebellar tremors are due to lesions of the lateral cerebellar nuclei or superior cerebellar peduncle, or its connections. Classically, a lesion within a cerebellar hemisphere or nuclei leads to an action tremor on the ipsilateral side of the body. Midline cerebellar disease may cause tremor of both arms, the head and the trunk.2 Lesions in the location of the red nucleus produce a wing-beating type of tremor (called rubral tremor), which is also present to a lesser degree with rest and posture.
During examination, a cerebellar tremor increases in severity as the extremity approaches its target. Other signs of cerebellar pathology, such as abnormalities of gait, speech and ocular movements, and the ability to perform rapidly alternating movements, may be present and may help to confirm the diagnosis of cerebellar tremor.3
Another type of tremor may also be associated with damage to the cerebellum. Termed “cerebellar postural tremor,” it is prominent with both action and posture.4 In its most severe form, cerebellar postural tremor has a frequency of 2.5 to 4 Hz and may wax and wane in amplitude, increasing progressively with prolonged posture. It persists and worsens with goal-directed movement.4 The milder form of the tremor has a more rapid frequency, approaching 10 Hz, and appears more distally, making it harder to identify than the severe type.4
Multiple sclerosis is the most common cause of the cerebellar postural tremor.4 Other causes of this tremor include tumors and strokes, as well as neural degeneration in the cerebellum.
ALCOHOL WITHDRAWAL TREMOR
Alcohol withdrawal tremor is similar to essential tremor on examination but with subtle differences. Alcohol withdrawal tremor has a frequency between 6 and 10.5 Hz. In one study,10 74 percent of the patients with alcohol withdrawal tremors had tremors at a frequency above 8 Hz. In this same series, tremors in all of the patients who had essential tremor were at a frequency below 8 Hz. Thus, the tremor of alcohol withdrawal tends to be more rapid than essential tremor.
A family history of tremor was found in only 1 percent of the patients with alcohol withdrawal tremor, as compared with almost one half of the patients with essential tremor.10 In addition, severity and degree of functional disability were less with alcohol withdrawal tremor.
Only the hands are affected in patients with alcohol withdrawal tremor, but multiple sites of involvement are possible in patients with essential tremor. Overactivity of the sympathetic nervous system is thought to be responsible for alcohol withdrawal tremor, and prolonged alcohol abuse can result in a chronic tremor disorder.10
PSYCHOGENIC TREMOR
Psychogenic tremor is a complex tremor that can occur at rest, during postural movement and during kinetic movement. The etiology and pathophysiology of psychogenic tremor are likely to differ from patient to patient, and the main focus of treatment should be psychotherapy, not medication.
Clinical features of psychogenic tremor include an abrupt onset, a static course, spontaneous remission and unclassifiable tremors.11 Unresponsiveness to antitremor drugs, an increase in frequency and amplitude with attention and a decrease in frequency and amplitude with distraction, responsiveness to placebo, absence of other neurologic signs and remission with psychotherapy are also signs of psychogenic tremor.11 Clinical inconsistencies, such as being able to write words yet not being able to draw a spiral, and changing characteristics, such as direction and affected body part, are also representative of psychogenic tremor.11
OTHER TREMORS
Other types of tremor occur much less commonly than the previously described tremors. Orthostatic tremor is defined as a postural tremor of the legs, occurring at a frequency of 13 to 18 Hz, initiated on standing and alleviated by walking or sitting.12 It is more readily noticeable during palpation than by sight and is not influenced by peripheral feedback.13 Unsteadiness, feelings of imbalance or weakness, and trembling and shaking in the lower limbs are associated features of orthostatic tremor.14 The etiology of orthostatic tremor is unknown, but it is currently regarded as an entity separate from essential tremor.12–14
Tremor associated with peripheral neuropathy is clinically similar to essential tremor. Its etiology is diverse. Not only can it be idiopathic, it can also be caused by demyelination from immunoglobulin M paraproteinemic neuropathies.2 Tremor in association with peripheral neuropathy can also result from Charcot-Marie-Tooth disease, diabetes mellitus, uremia and porphyria.2
Drug Treatment of Tremor
PARKINSONIAN TREMOR
Treatment of Parkinson's disease includes both medical and surgical intervention. Dopamine replacement therapy by means of levodopa clearly revolutionized the treatment of Parkinson's disease. Levodopa is almost exclusively given in combination with the peripheral decarboxylase inhibitor carbidopa (Sinemet). Carbidopa blocks the peripheral metabolism of levodopa to dopamine, decreasing the peripheral adverse effects of levodopa, such as nausea and vomiting, while increasing levodopa's availability in the brain.15,16 In addition to modulating the tremor associated with Parkinson's disease, levodopa improves bradykinesia, rigidity and other commonly associated symptoms. Carbidopa–levodopa is available in formulations of 10/100 mg, 25/100 mg and 25/250 mg. It is advantageous to begin treatment of mild disease with the 25/100-mg dosage, one tablet three times a day, and then increase the dosage as symptoms become less manageable.
When tremor is the predominant presenting symptom of Parkinson's disease or when tremor persists despite adequate control of other parkinsonian symptoms with low dosages of levodopa, an anticholinergic agent such as trihexyphenidyl (Artane) or benztropine (Cogentin) may be the treatment of choice. In most patients, however, anticholinergics do not significantly improve bradykinesia and rigidity. Trihexyphenidyl dosages necessary to improve tremor are between 4 and 10 mg per day (maximum: 32 mg), and useful benztropine dosages range from 1 to 4 mg per day. The side effects of these agents are their limiting factor, particularly in the elderly. Side effects include memory impairment, hallucinations, dry mouth, urinary difficulties and blurred vision.15
Other antiparkinsonian drugs—for example, amantadine (Symmetrel), tolcapone (Tasmar) and dopamine agonists such as pergolide (Permax), bromocriptine (Parlodel), ropinirole (Requip) and pramipexole (Mirapex)—are most helpful in patients whose tremor responds poorly to levodopa alone.
ESSENTIAL TREMOR
As with other tremors, effective treatment of essential tremor is not found in a single, universal agent. Some therapies may be satisfactory in some patients and ineffective in others. The most widely used drugs for essential tremor are the beta-adrenergic blocker propranolol (Inderal) and the anticonvulsant primidone (Mysoline). The typical dosage range for propranolol is 80 to 320 mg per day and for primidone, 25 to 750 mg per day.3 Other beta-adrenergic receptor antagonists used in the treatment of essential tremor include metoprolol (Lopressor) and nadolol (Corgard).2 Alcohol is also effective in relieving essential tremor, but abuse may be an adverse consequence.3
In our experience, propranolol and primidone are equally effective in the treatment of essential tremor. Patients who do not respond to one medication after a few weeks of therapy should be tried on the other one. Primidone may be preferred, because of the exercise intolerance associated with high-dose beta blockade. Patients who have a very-low-amplitude rapid tremor are generally more responsive to these agents than those who have a slower tremor with greater amplitude. Patients who have tremor of the head and voice may also be more resistant to treatment than patients with essential tremor of the hands.
OTHER TREMORS
There is no established treatment for cerebellar tremor.2 In patients with multiple sclerosis, severe cerebellar tremor may be improved with isoniazid, in a dosage of 600 to 1,200 mg per day, given together with pyridoxine.4
Propranolol in a dosage of 160 mg per day is very effective in reducing the tremor associated with alcohol withdrawal.10
Treatment of orthostatic tremor should first be attempted with clonazepam (Klonopin). In one small study,14 eight of nine patients responded to clonazepam in dosages ranging from 0.5 to 2.0 mg per day. The patient who did not respond to clonazepam responded to chlordiazepoxide (Librium), in a dosage of 30 mg twice a day. In another study,12 10 of 18 patients had sustained improvement with clonazepam, and valproic acid was effective in the remaining eight patients. However, propranolol in daily dosages of up to 320 mg had no effect on controlling orthostatic tremor.
Tremor due to peripheral neuropathy may be ameliorated with propranolol, primidone, benzodiazepines or baclofen (Lioresal), but the underlying cause of the neuropathy itself should be treated as well.2
Other medications have been shown to be helpful in the management of tremor but should probably only be tried in consultation with a neurologist, when the previously mentioned drugs have failed to control the tremor.
Surgical Treatment of Tremor
THALAMOTOMY
Surgical therapy for tremor should only be considered if drug therapy fails to produce adequate relief. Stereotactic thalamotomy is the surgical procedure most often used to quell essential tremor. Before the introduction of levodopa, thalamotomy was an often-selected option in the treatment of Parkinson's disease. Because the benefits of levodopa wane after four to seven years of therapy, this procedure remains an option in some patients with severe parkinsonian tremor refractory to drug therapy. However, problems associated with bilateral thalamotomy, such as dysphagia and dysarthria, limit its use. Thalamotomy is usually only considered in patients with severe, drug-resistant essential tremor and in a very small subset of patients with Parkinson's disease who have severe, disabling, predominantly unilateral tremor.
In one study of the use of stereotactic thalamotomy in the treatment of tremor,17 86 percent of the 42 patients with parkinsonian tremor and 83 percent of the six patients with essential tremor had cessation of tremor or moderate to marked improvement in tremor after the procedure. Follow-up in some patients was as long as 13 years (mean follow-up: 53.4 months). The investigators used three criteria for patient selection: (1) predominantly unilateral, severe and incapacitating tremor, (2) a poor response to or intolerance of optimal medical therapy and (3) no potentially serious risk factors for surgery. Postoperative complications included weakness, dysarthria and confusion, but these problems subsided with time.
Catastrophic complications in the perioperative period include bleeding in the thalamus or the subdural or epidural area, which can lead to death, paralysis, aphasia or significant cognifive deficits.
PALLIDOTOMY
Producing lesions in the globus pallidus by means of pallidotomy is an alternative to thalamotomy in the treatment of parkinsonian tremor. Pallidotomy also improves other symptoms of Parkinson's disease, such as bradykinesia and levodopa-induced dyskinesias.18 As with thalamotomy, pallidotomy should only be considered in cases of severe tremor unresponsive to medical treatment.
In a series of 259 patients who underwent pallidotomy for parkinsonian tremor,18 complete relief of all symptoms on the side contralateral to the procedure occurred in 212 patients (81.9 percent). Of the remaining 47 patients, 36 experienced substantial improvement and 11 had only minor or no improvement. In many of the patients, pallidotomy also produced a significant reduction in bradykinesia, rigidity and levodopa-induced dyskinesias. The side effects associated with the procedure were similar to those of thalamotomy and included visual field defects, such as lower central visual field scotomas, and hemiparesis. Cognitive deficits, dysarthria and foot apraxia occurred in less than 1 percent.
If the pallidal lesion is large enough and placed at the posteroventral margin of the lateral pallidum, it abolishes the tremor as often as thalamotomy. However, because of theoretic concerns that bilateral pallidotomy may cause cognitive deficits, this approach must be explored before it is comonly used in the treatment of tremor.
THALAMIC STIMULATION
During physiologic localization in preparation for thalamotomy, the observation that high-frequency stimulation of the ventral intermediate nucleus of the thalamus abolished tremor led to investigation of thalamic stimulation as a treatment for tremor. The first study of this technique as a long-term therapy for tremor was reported in 1993.19
Thalamic stimulation involves implanting an electrode in the thalamic area found to be responsible for the tremor. After the wire of the electrode leaves the skull, it is tunneled under the scalp and down the neck to a purse generator located in the subclavicular pouch (Figure 1). The implanted stimulating device is much like a modified pacemaker, and its electrical impulses can suppress tremor indefinitely. The stimulator can be reprogrammed by using a small portable computer that communicates with the device by radio frequency. Moreover, the patient can turn the device on and off with a magnet. Patients usually turn the device on in the morning, leave it on during waking hours and turn it off at bedtime, since most tremors cease during sleep.
FIGURE 1.
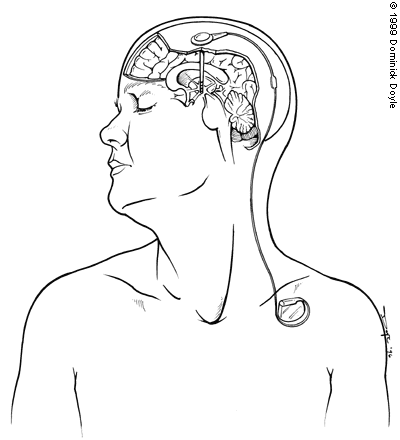
Thalamic stimulation, showing position of the electrode within the thalamus and path of the wire to the pulse generator in the subclavicular pouch.
In the first study of this technique,19 as many as 88 percent of the patients with Parkinson's disease had either good or excellent relief of tremor. The operative risk of implanting the device is proving to be similar to that of thalamotomy; death, paralysis, aphasia and significant cognitive deficits are possible complications.
Tremor recurrence after placement of the electrode can be controlled by adjusting the stimulation parameter rather than by reoperation.20 The U.S. Food and Drug Administration has approved thalamic stimulation as an accepted therapy for unilateral suppression of uncontrolled essential tremor or parkinsonian tremor in an upper extremity. As with the other surgical techniques, thalamic stimulation is an option that should be chosen only after medical therapy has failed.
PROMISING SURGICAL APPROACHES
At the forefront of new surgical therapies for tremor are pallidal stimulation and sub-thalamic nucleus stimulation.21–23 With new advances in deep brain stimulation, procedures can be performed bilaterally to relieve tremor in patients with bilateral involvement. Either a combination of thalamotomy and stimulation or bilateral stimulation without ablation is now a possibility.23 Targets in the brain that are too dangerous to approach for producing a lesion by means of thalamotomy may be treated with stimulation instead, and electrical stimulation can be modified to alleviate tremor as it progresses.22 Thus, deep brain stimulation has become a promising option for abolishing tremors that cannot be controlled by medical therapy.

