Although testicular cancer accounts for only 1 percent of all tumors in males, it is the most common malignancy in males between 15 and 34 years of age. Cryptorchidism is the most significant risk factor for testicular cancer, increasing the risk up to 11-fold. A painless testicular mass is the classic presentation for testicular cancer, although a number of patients present with diffuse pain or swelling. Ultrasonography may be helpful in confirming the presence of a scrotal mass within the testicle. Intratesticular masses are considered malignant until proved otherwise. Radical orchiectomy is the treatment for the primary tumor. Staging of disease is based on tumor histology, serum tumor markers and presence of lymphnode or other metastatic disease. Depending on the stage of disease, further treatment may include observation, radio-therapy, chemotherapy or surgery. Survival rates in patients with testicular cancer have improved dramatically in the past 20 years and now exceed 90 percent overall.
Testicular cancer accounts for only about 1 percent of all cancers in males. It is, however, the most common tumor in males between 15 and 34 years of age.1 About 7,600 cases were expected in the United States in 1998,2 or about 4.2 cases per 100,000 males.3 Incidence rates vary across the globe, with higher rates in Scandinavia and Germany and lower rates in Asia and Africa.
The incidence of testicular cancer in the United States has almost doubled since the 1930s and continues to climb, while more effective treatments have led to a decline in mortality3,4 (Figure 13). The survival rate in 1963 was 63 percent2; the five-year survival rate now exceeds 95 percent.3
FIGURE 1. Testicular cancer
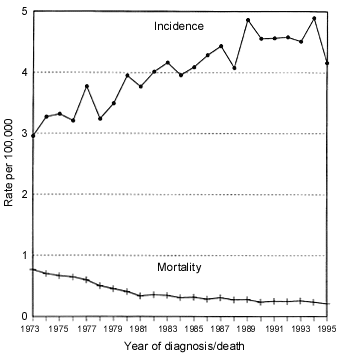
Testicular cancer incidence and rates of mortality in the United States, 1973 to 1995.
Information from Ries LG, Kosary CL, Hankey BF, Miller BA, Edwards BK, eds. SEER cancer statistics review, 1973-1995. Bethesda, Md.: U.S. Dept of Health and Human Services, Public Health Institute, National Institutes of Health, 1998. Retrieved November 1998 from the World Wide Web: http://www-seer.ims.nci.nih.gov/Publications/CSR7395.
The risk factors associated with testicular cancer include white race, cryptorchidism, testicular atrophy or dysgenesis, and family history.5 Up to 10 percent of testicular tumors are diagnosed in males with a history of an undescended testicle. A male with a history of cryptorchidism has a risk of developing testicular cancer that is 2.5- to 11-fold greater than that of an unaffected male.6 The higher the testicle is located (inguinal canal versus intra-abdominal), the higher the risk; the risk is greatest in cases of bilateral cryptorchidism.
Interestingly, up to 20 percent of testis tumors in males with cryptorchidism occur in the contralateral, normally descended testicle.7 Orchiopexy performed before puberty is purported to decrease the risk of cancer according to some authors,8 while others believe that the timing of the procedure does not alter the malignant potential of a cryptorchid testis.9,10 Orchiopexy is recommended for several reasons. The longer the testicle remains undescended, the lower the rate of fertility. Secondly, since these men are at increased risk for testicular cancer, moving the testicle to the scrotum permits screening by palpation.
Testicular cancer occurs approximately four to five times more frequently in whites than in blacks8,11 and seems to be more common in patients of higher socioeconomic status. Ten percent of males diagnosed with testicular cancer give a history of recent trauma to the testicle. It is not thought that the trauma causes the cancer, but rather that it brings an existing tumor to the attention of the patient and physician. Epidemiologic studies have failed to show an association between the incidence of testicular cancer and vasectomy,12 occupational and environmental exposures, or viral illnesses.
Histology
Germ cell tumors account for approximately 95 percent of tumors of the testis. Leydig cell tumors and lymphomas are more common only in elderly patients.
Germ cell tumors are divided into seminomas and nonseminomas. Nonseminomatous tumors include yolk sac, embryonal cell, teratoma and choriocarcinoma. Most nonseminomas are a combination of several cell lines. The distinction between seminoma and nonseminoma is the main factor in directing treatment. Pure seminomas, which account for approximately 40 percent of germ cell tumors, are extremely radiosensitive, while nonseminomas have to be treated with platinum-based chemotherapy or surgery. Seminomas that have any other cell types or that secrete alpha-fetoprotein require the more aggressive treatment prescribed for nonseminomatous tumors.
Diagnosis
Early diagnosis of testicular cancer is crucial since the doubling time of testis tumors is estimated to be 10 to 30 days.11 While survival rates at all stages are very high, an earlier stage at diagnosis carries a better long-term prognosis. Men are often reluctant to report a swelling or lump in the testicle, resulting in a delay in presentation to a physician. It is common for these tumors to be misdiagnosed as epididymitis and treated ineffectively with antibiotics or neglected for months. In studies, the duration of symptoms before a correct diagnosis ranges from 17 to 87 weeks.13
The classic presentation of testicular cancer is a painless lump in the testis, although a substantial proportion of patients report diffuse pain, swelling or hardness in the scrotum.8 The changes are usually found during self-examination, after testicular trauma or by a sexual partner. Signs of metastatic disease include swelling of the lower extremities, back pain, cough, hemoptysis or dyspnea. Approximately 10 percent of males present with gynecomastia from tumors that secrete beta human chorionic gonadotropin (β-HCG).14,15
In any patient with a testicular mass, or unexplained scrotal pain or swelling, an ultrasonogram of the scrotum should be obtained. Scrotal ultrasonography is nearly 100 percent accurate in distinguishing between intratesticular and extratesticular pathology.16 All intra-testicular masses are considered cancer until proved otherwise. After an intratesticular neoplasm is identified, a chest radiograph, a computed tomographic (CT) scan of the abdomen and serum tumor markers are obtained for staging.
Testicular tumors metastasize via the lymphatic system, with the exception of chorio-carcinoma, which spreads hematogenously. The primary path for testicular cancer metastases is to the retroperitoneal lymph nodes near the renal vessels. The inguinal nodes are usually not involved unless the tumor has invaded the scrotum, a scrotal incision was made during biopsy or orchiectomy, or there was prior scrotal surgery such as repair of a hydrocele or varicocele.
Radical orchiectomy, performed for definitive diagnosis, is also the first step in most treatment regimens. The spermatic cord and vessels are clamped early, and the entire specimen is brought out of the scrotum. Trans-scrotal testicular biopsy is absolutely contraindicated in the diagnosis or management of testicular cancer.
Staging
The most common staging system for testicular tumors is outlined in Table 1. Clinical staging involves several components: findings at orchiectomy, chest radiograph, CT scans of the abdomen and pelvis, and serum tumor markers. CT scanning is the most reliable method of determining retroperitoneal lymph node involvement. This method has largely replaced the pedal lymphangiogram. The limitation of CT is that it will under- or over-stage the disease approximately 25 percent of the time.11
TABLE 1 Staging and Five-Year Survival Rates in Patients with Testicular Cancer
| Stage | Criteria | Five-year survival rate (%) |
|---|---|---|
| I | Confined to testicle | 98 |
| II | Metastases to retroperitoneal nodes; sometimes categorized as bulky or nonbulky, depending on size and number of involved nodes | 97 |
| III | Metastases above the diaphragm or to visceral organs | 72 |
Information from Ries LG, Kosary CL, Hankey BF, Miller BA, Edwards BK, eds. SEER cancer statistics review. Bethesda, Md.: U.S. Dept of Health and Human Services, Public Health Institute, National Institutes of Health, 1998.
Another approach to staging is retroperitoneal lymph node dissection (RPLND). RPLND is performed more commonly in patients with nonseminomatous tumors and is a more invasive but also more accurate means of staging. Nerve-sparing procedures are employed for dissection of the lymph nodes, but they still carry a risk for retrograde ejaculation and subsequent infertility. RPLND is also curative in many patients with microscopic disease limited to the retroperitoneal nodes.
Serum Tumor Markers
Serum tumor markers are routinely used for diagnosis, staging and follow-up. The markers (Table 217–19 ) that are commonly identified in the serum are the beta subunit of HCG, alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH).
TABLE 2 Histology of Primary Germ Cell Tumors and Frequency of Serum Tumor Markers
| Tumor type | Frequency by cell type (%) | Presence of tumor markers by cell type (%) | ||
|---|---|---|---|---|
| AFP | HCG | |||
| All germ cell tumors | 100 | 50 to 75 | 40 to 60 | |
| Seminoma | 42 | 0 | 9 | |
| Nonseminomatous germ cell tumors | 58 | 65 | 56 | |
| Embryonal cell carcinoma | 26 | 70 | 60 | |
| Teratocarcinoma | 26 | 64 | 57 | |
| Teratoma | 5 | 37 | 25 | |
| Choriocarcinoma | 1 | 0 | 100 | |
| Yolk sac | <1 | 75 | 25 | |
AFP = alpha-fetoprotein; HCG = human chorionic gonadotropin.
HCG is not detectable in healthy males. It is more commonly secreted by nonseminomatous tumors than by seminomas. The half-life of HCG is 24 to 36 hours, and levels should return to normal within five to eight days after the tumor is eliminated.
AFP is detectable only in minute amounts in healthy men (less than 25 ng per mL [25 μg per L]). It is never produced by pure seminomas or choriocarcinomas. A histologically reported pure seminoma in a patient with an elevated AFP level should be treated as a nonseminomatous germ cell tumor. The half-life of AFP is five to seven days, and the level should return to normal within 25 to 35 days of tumor removal.
LDH is a ubiquitous enzyme that is elevated in 50 percent of all patients with testis tumors. Levels of LDH-1 are particularly likely to be elevated. LDH is not specific to any one tumor type and is generally elevated in proportion to tumor volume.
The absence of elevated levels of HCG, AFP and LDH in a patient with a testicular mass does not rule out the presence of a tumor. Likewise, normal levels after treatment for a germ cell tumor do not rule out the presence of residual tumor. Following tumor markers after treatment is important, since these markers can signal an incomplete cure or relapse long before it is evidenced by physical or radiologic examination.
Treatment
Treatment after orchiectomy depends on the stage and histology of the tumor—pure seminoma versus mixed or nonseminoma (Table 3). Seminoma is extremely radiosensitive, and low-stage disease is treated with radiation to the inguinal and retroperitoneal areas. Cure rates for low-stage disease now approach 99 percent.8 Men with nonseminomatous germ cell tumors of the testis can be managed with observation, chemotherapy or RPLND. In carefully selected cases, observation is advocated by some authors for low-stage, less aggressive tumors, since relapses are very responsive to subsequent chemotherapy or surgery.8,20 The National Cancer Institute's CancerNet Internet site (http://cancernet.nci.nih.gov) frequently updates treatment protocols for each type and stage of testicular cancer.
TABLE 3 Overview of Treatment for Testicular Cancer by Stage and Type
| Stage | Type of cancer | Treatment |
|---|---|---|
| I | Seminoma | Radiation to retroperitoneal and ipsilateral inguinal nodes |
| Nonseminoma | RPLND or surveillance | |
| IIA (nonbulky) | Seminoma | Radiation to retroperitoneal and inguinal nodes, possibly with mediastinal and supraclavicular nodes |
| Nonseminoma | RPLND, possibly with platinum-based combination chemotherapy | |
| IIB (bulky) | Seminoma | Platinum-based combination chemotherapy or radiation, as in IIA |
| Nonseminoma | Platinum-based combination chemotherapy, followed by resection | |
| III | Seminoma | Platinum-based combination chemotherapy, possibly with resection of residual mass |
| Nonseminoma | Platinum-based combination chemotherapy, possibly with resection of residual mass |
RPLND = retroperitoneal lymph node dissection.
Information from National Cancer Institute. Testicular cancer treatment summary for health professionals. CancerNet [database on Internet] 1992 [updated December 1997; cited February 1, 1998]. Available at: http://cancernet.nci.nih.gov/clinpdq/soa/Testicular_cancer_Physician.html.
Platinum-based chemotherapy regimens have revolutionized the treatment of testicular cancer since their development 25 years ago. Now chemotherapy is extremely effective even in patients with high-volume disease and relapse. Since survival rates for testicular cancer are among the highest of any cancer, most research now focuses on ways to optimize treatment to decrease side effects. One of the major complications of treatment that is especially important, given the young age of most patients, is potential infertility.
Testicular cancer is a highly curable neoplasm and a model for cancer management. The overall five-year survival rate now exceeds 95 percent. Stage for stage, patients with seminoma have slightly higher survival rates than patients with nonseminomatous tumors.
Follow-up and Prognosis
Many of the deaths that are related to testicular cancer are a result of severe metastatic relapse after inadequate follow-up. It is all too common for young males to believe they are cured after a few negative follow-up visits and to then forgo routine examinations. Testicular cancer is a disease in which even recurrence is highly curable, especially if detected early. Given the young patient population, many years of life can be lost as a result of metastatic cancer that is not detected and treated early. Most relapses occur in the first year. After three years without recurrence, the probability that a patient is cured is greater than 95 percent.21
Fairly rigorous follow-up protocols usually include physical examinations, chest radiographs and detection of serum tumor markers.21–23 CT scans are used to detect recurrence in the retroperitoneum and occasionally in the chest. The physical examination should focus on a careful examination of the lymph nodes, abdomen and remaining testicle. Follow-up protocols vary by institution and by type, stage and treatment of the primary disease. Most protocols recommend examinations every two to three months during the first year of follow-up, every three to six months the second year, and then every six months for the remainder of five years. Most protocols utilize abdominal CT scans every three to six months during the first year or two and then annually for the remainder of five years.
Patients who have been cured of testicular cancer have a 2 to 5 percent risk of developing cancer in the opposite testicle9,24 and a slightly increased incidence of malignancies elsewhere in the body.25,26
Screening
Large-scale screening for testicular cancer is a controversial issue. Detractors state that given a low incidence and a high cure rate, without any studies to show a decrease in mortality as a result of screening, present evidence does not permit a recommendation for or against routine screening. Other public health groups recommend incorporating a testicular examination at the time of an annual physical examination and teaching testicular self-examination to young males. It is generally agreed that men in high-risk groups (those with cryptorchidism, testicular atrophy or dysgenesis, and those with a family history of testicular cancer) should be checked regularly and should perform testicular self-examination monthly. Regular screening with tumor markers is not necessary.
Most young males are not aware of testicular cancer and its prevalence in their age group. These patients may benefit from a reminder that testicular cancer is the leading cancer in their age group and from education about the performance of periodic testicular self-examinations.

