Vasectomy can be performed by means of various techniques, although each vasectomy technique requires isolation and division of the vas and operative management of the vasal ends. Removal of at least 15 mm of vas is recommended, although division of the vas without removal of a segment is effective when this technique is combined with other techniques for handling the vasal ends, such as thermal luminal fulguration and proximal fascial interposition. Ligation of the ends without the aid of surgical clips may result in necrosis and sloughing of the ends, which may cause early failure. Leaving the testicular end of the vas open has been shown to be effective and to result in a lower incidence of epididymal congestion and sperm granuloma. The no-scalpel technique offers shorter operating time, less pain and swelling, and faster recovery.
Vasectomy is among the most reliable and cost-effective methods of contraception. Family physicians perform approximately 15 percent of the estimated 500,000 vasectomies performed each year in the United States.1 Training in vasectomy techniques is frequently provided during family practice residency, yet only 33 percent of residency-trained family physicians perform the procedure.2 That many family physicians choose not to offer the procedure in their practices may in part relate to the belief that reimbursement is inadequate for making this procedure cost-effective for the practice3 or to the perception that the number of vasectomies performed during training was insufficient to provide an adequate level of comfort in doing this procedure.4
Patients often ask their family physicians about contraception. Because sterilization is the most widely used contraceptive method among married couples in the United States,5 a choice between vasectomy and tubal sterilization is often discussed. Failure rates of vasectomy and tubal sterilization are comparable, as are their rates of successful reversal.5 Although both procedures are considered very safe, patients should be advised that deaths, while rare, occur more often in association with tubal sterilization than with vasectomy6 and are usually related to complications of general anesthesia. Ultimately, the choice between tubal sterilization and vasectomy is often determined by one spouse's refusal to be sterilized, usually the husband's.7
Preoperative Evaluation
Ideally, the patient and his partner should be seen together for evaluation and counseling before vasectomy. The history should include information about the patient's marital status, number of children and motives for the procedure. His medical and surgical history should be reviewed as well. Any current or past history of genital infection, trauma, surgery or pain should be explored, as should any history of major medical illnesses such as diabetes, hypertension or bleeding disorders. A past history of surgical or anesthetic complications should be noted. Much of the history can be obtained by nursing personnel, with the aid of a standardized form (Figure 1).
FIGURE 1. Vasectomy Procedure Checklist

Sample history form for vasectomy procedure.
During counseling before the procedure, the pertinent anatomy and details of the procedure should be described. Benefits of the procedure and potential complications should be explained in detail, and the patient and his partner should be encouraged to ask questions.
The possibility of complications should be discussed with the patient (Table 1). Perhaps the most important complication to discuss is the failure rate of vasectomy as a contraceptive method. Pregnancy occurs after a vasectomy in most cases because the couple had sexual intercourse before azoospermia was documented by two separate semen samples.8 Despite the importance of verifying the lack of sperm in the semen after vasectomy, many patients do not comply with the instructions for semen analysis. In one study, as many as 45 percent of patients did not seek follow-up for semen analysis.8 True early failure, defined as the presence of motile spermatozoa in the ejaculate four months after surgery,9 is usually attributed to technical error or to early recanalization of the vas. Early failure occurs in about one out of 300 vasectomies.10 Late recanalization is thought to be rare, although in one study, the rate of recanalization was 0.6 percent one year after vasectomy.10
TABLE 1 Complications of Vasectomy
| Complication | Rate of occurrence (%) |
|---|---|
| Surgical complications | |
| Bleeding or hematoma | 1.6 to 4.6 |
| Incisional infection | 2.2 to 6.0 |
| Sperm granuloma | 15 to 40 |
| Congestive epididymitis | 0.4 to 6.1 |
| Other complications | |
| Hospitalization | 0.2 to 0.8 |
| Psychogenic impotence | Uncommon |
| Vasourinary fistula | Rare |
Reprinted from Raspa RF. Complications of vasectomy. Am Fam Physician 1993;48:1264–8.
If the patient's history is unremarkable for major medical illness, a directed genital examination can be performed. The scrotal skin should be inspected for infectious lesions and rashes. The testes should be examined to exclude the presence of tenderness, nodularity or hydrocele. Both vasa should be palpated, with particular attention paid to the mobility and the number of vasa. Congenital absence of a vas (i.e., a single vas) requires further urologic evaluation before vasectomy can be performed, because congenital absence of a vas may be associated with an absent kidney on the ipsilateral side.11 Palpation for the presence of an accessory vas should be performed, because an accessory vas may result in early failure of the procedure. Finally, both sides should be checked for the presence of a varicocele or a hernia.
Written instructions, emphasizing the importance of postoperative ice applications, scrotal support, rest and semen analysis, should be provided to the patient during the counseling visit. At the conclusion of counseling, the patient should be given a surgical consent form to sign in the presence of a witness. A prescription for a preoperative anxiolytic medication, such as diazepam (Valium), may be given at this time, with dosing instructions.
Preparation
The patient should be instructed to shower and cleanse the genital area thoroughly on the day of surgery. He should not eat for two hours before the procedure. Aspirin should not be taken for one to two weeks before the procedure, and other nonsteroidal anti-inflammatory agents, platelet inhibitors and anticoagulants should be withheld for three to four days before the procedure. The patient may take any other regular medications. If an anxiolytic has been prescribed, he should take it 30 minutes before the procedure.
The proper surgical environment includes a warm room to facilitate scrotal relaxation and soft music to help alleviate anxiety. The procedure is performed with the patient in the supine or dorsolithotomy position. The dorsolithotomy position is favored by some because it allows the weight of the testes to stretch the vasa and thus facilitate the procedure.12
The instruments in a typical vasectomy tray are listed in Table 2. Intravenous access is generally not required, although if the patient is particularly anxious, he may benefit from administration of intravenous midazolam (Versed). The need for blood pressure monitoring and pulse oximetry may limit the use of this procedure in some offices.
TABLE 2 Equipment for a Vasectomy Tray
| Mosquito hemostats (3) |
| Adson forceps |
| Towel clamps (3) |
| No. 7 scalpel handle |
| No. 15 scalpel blade |
| Needle holder |
| Iris scissors |
| Surgical clip applicator |
| Stainless steel surgical clips |
| Disposable thermal cautery |
| 4-0 chromic suture |
| 10-mL syringe with 27-gauge needle |
| 1% lidocaine (Xylocaine) without epinephrine |
| Sterile gauze |
| Fenestrated drape |
| Sterile gloves |
The surgical area should be shaved or trimmed, and warmed povidone-iodine solution applied. The penis may then be retracted from the surgical field by taping it to the abdomen. Other techniques for retracting the penis from the surgical field have been described,13 although simple draping with a fenestrated sterile drape will allow complete visualization of the scrotum.
Anesthesia
A cursory examination of the genital area should be performed to confirm anatomic landmarks before administration of local anesthesia. The scrotum may be lubricated with saline or lidocaine (Xylocaine) at this time to facilitate examination.
For providing anesthesia, a 1- to 2-cm wheal should be made at the desired incision site, usually at the junction of the middle and upper one third of the scrotum bilaterally. The needle is advanced through the wheal, without injecting the anesthetic, parallel and adjacent to the vas and toward the external inguinal ring (Figure 2).14 After gentle aspiration, 2 to 5 mL of 1 percent lidocaine without epinephrine is injected into the external spermatic fascia.14 Another technique has been advocated, utilizing a eutectic mixture of topical local anesthetic (EMLA cream) to anesthetize the scrotal skin, followed by spermatic cord injection.15
FIGURE 2.
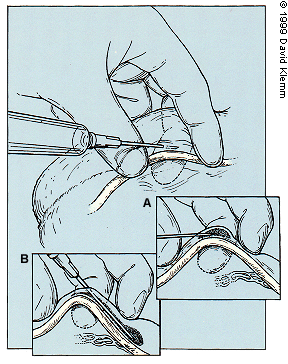
Three-finger grasp with injection of lidocaine. (A) Injection directly over the vas. (B) Paravasal injection after 2.5 cm advancement of needle in cephalic direction.
Adapted with permission from Li PS, Li S, Schlegel PN, Goldstein M. External spermatic sheath injection for vasal nerve block. Urol 1992;39:173–-6.
Selecting a Technique
Although various vasectomy techniques have been described, all share three essential steps: isolation of the vas deferens, delivery and interruption of the vas, and management of the vasal ends.
ISOLATION OF THE VAS
Isolation of the vas can occasionally be challenging. The technique used to position the vas is the key to a successful procedure. A “three-finger technique” for positioning the vas has been described13 in which the non-dominant hand is used to manipulate the vas into a subcutaneous position (Figure 3).
FIGURE 3.
Vas isolation using the three-finger technique.
We have also found success with a modification of this technique. The thumb of the nondominant hand is placed posterior to the cord and scrotum, and the vas is trapped between the posterior thumb and the middle finger in a pincer grasp (Figure 4). The index finger is then used to retract the skin, yielding improved exposure of the vas.
FIGURE 4.

Simulation of vas isolation using the “pincer-grasp” technique.
With either technique, the cord is then maneuvered subcutaneously to the desired operative site, which is usually at the junction of the middle and upper one third of the scrotum bilaterally. A midline approach may be selected, although the bilateral technique is often preferred because of the ease of grasping and positioning the vas.12
INTERRUPTION OF THE VAS
After the vas is maneuvered to the desired location, a 1- to 2-cm horizontal or vertical incision is made (Figure 5). Next, the soft tissue is bluntly dissected with a fine curved hemostat. An Allis or towel clamp is then advanced through the incision to grasp the vas and surrounding tissue (Figure 6). Pushing the vas forward into the clamp while it is grasped allows less tissue to be ensnared along with the vas. The vas and surrounding tissue may then be elevated through the incision. After further blunt dissection, the fibrous layer surrounding the vas is incised longitudinally (Figure 7). Now fully isolated, the vas is ready for division (Figure 8).
FIGURE 5.
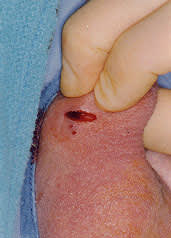
A 1- to 2-cm incision is made in the scrotal skin overlying the vas.
FIGURE 6.
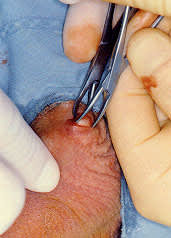
A towel clamp is used to isolate the vas.
FIGURE 7.
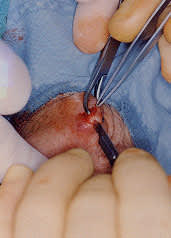
The fibrous tissue surrounding the vas is incised longitudinally.
FIGURE 8.
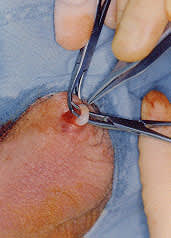
The vas is fully isolated and ready for division.
Controversy exists regarding the length, if any, of the vas segment to be removed. In one study, removal of segments that were less than 14 to 16 mm in length was associated with increased rates of recanalization.16 Removal of at least 15 mm is therefore recommended. It is worth noting that about 1 percent of patients will request a vasectomy reversal (vasovasostomy), and that removal of an excessively long segment of vas decreases the potential for successful reversal of the procedure.17 The removed segment may be sent for pathologic examination to confirm that the vas has been interrupted, although many clinicians experienced in this procedure believe that pathologic examination of the specimen adds unnecessary cost with little benefit. An alternative is to save the removed vasal segments in fixative in a labeled container should pathologic examination be subsequently desired because of an early failure.
MANAGEMENT OF THE VASAL ENDS
Techniques for management of the vasal ends vary. Options include ligation, clipping, cautery, proximal fascial interposition and open-ended techniques. Combinations of these methods are usually performed. Ligation of the vasal ends is a common practice, yet it is reported to result in failure in approximately 1.5 percent of cases.18 Failure after ligation is most likely the result of ischemic necrosis and sloughing of the vasal ends. Placement of surgical clips before ligation has been shown to reduce failure rates18 (Figure 9). If surgical clips are used, they should be placed before excision of the vasal segment.
FIGURE 9.
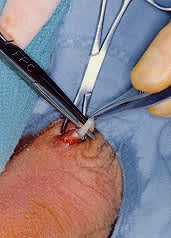
Metal surgical clips are placed before division of the vas.
Luminal fulguration (cauterization) of the vasal ends is also frequently used. This method creates a third-degree burn, which subsequently produces a scar that seals the end. When done correctly, cauterization is accomplished without necrosis and sloughing of the vasal ends. The cautery should be inserted 3 to 4 mm into the cut ends and withdrawn slowly (Figure 10).
FIGURE 10.
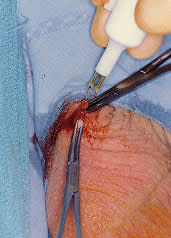
A thermal cautery is inserted into the free ends of the vas and withdrawn slowly.
The combination of electrocautery and proximal fascial interposition, without removal of a segment of the vas, was demonstrated in one study to reduce the failure rate to one tenth that of standard ligation alone.19 Importantly, however, thermal cautery as compared with electrocautery has been shown to produce a lower incidence of vasitis nodosa and sperm granuloma.20 A sperm granuloma is the result of an inflammatory reaction to the leakage of sperm from the vas. Sperm granuloma develops in 15 to 40 percent of patients undergoing vasectomy and may cause pain in up to 3 percent of patients in whom it develops.6 With vasitis nodosa, blind channels lined with epithelium arise from the lumen of the vas. Both of these complications may lead to spontaneous recanalization and late failure. Proximal fascial interposition involves the closure of the fascial sheath over the prostatic cut end of the vas in a purse-string fashion (Figure 11). This technique in combination with luminal fulguration of both ends of the vas was shown to be effective and to cause few postoperative complications in a study of over 6,000 patients.21 As with the study of electrocautery combined with proximal fascial interposition, a segment of vas was not removed in this series,21 suggesting that low failure rates may relate more to the appropriate treatment of the vasal ends than to the length of the segment removed.
FIGURE 11.
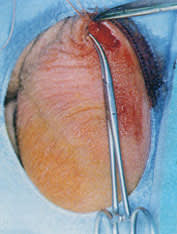
The fascia is closed over the proximal end in a purse-string fashion.
An open-ended technique first reported in 198622 has been investigated as a method for preventing painful side effects and allowing easier reversibility of vasectomy. With the open-ended technique, the testicular end of the vas is left open and the prostatic end is managed with cautery and fascial interposition. In two separate studies,22,23 a marked decrease in epididymal congestion was observed along with fewer instances of symptomatic sperm granuloma and failure.
Following removal of the vasal segment and management of the ends, the wound is then inspected for active bleeding, and hemostasis is ensured. The incision is closed with absorbable suture (Figure 12).
FIGURE 12.
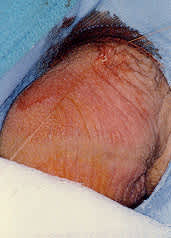
The wound is closed with absorbable suture material.
No-Scalpel Technique
The no-scalpel, or Li, technique was developed in China in 1974 in the hope of improving the acceptance of vasectomy in that country. This technique differs from conventional vasectomy techniques primarily by the unique method of incision and delivery of the vas. A ringed extracutaneous vas clamp (Figure 13) is used to fixate the vas to the overlying skin, and a single midline incision is made by means of a sharpened clamp. Compared with other vasectomy techniques, the benefits of no-scalpel vasectomy include a shorter operating time,24 less pain and swelling, and faster recovery.25 Failure rates are similar to those associated with conventional techniques.26 Although not universally performed, no-scalpel vasectomy is increasing in popularity. The no-scalpel technique, with prostatic-end cautery and fascial interposition, combined with leaving the testicular end open, has been advocated as the “ideal technique.”23
FIGURE 13.
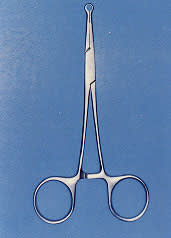
The ringed extracutaneous vas clamp used in the no-scalpel technique.
Postoperative Care
After the procedure is completed, postoperative instructions should be reviewed with the patient. Appropriate recommendations include intermittent ice applications to the scrotum for eight hours, bed rest and scrotal support for 48 hours, and avoidance of heavy exertion for one week. Acetaminophen usually provides sufficient analgesia, although occasionally narcotic analgesics are necessary. Sexual activity should be avoided for one week. Another method of contraception should be used until semen analysis has confirmed azoospermia. A telephone call 48 to 72 hours after surgery and a one-week follow-up office visit provide opportunities for the physician to assess the patient's condition and reemphasize the postoperative instructions.

