HELLP, a syndrome characterized by hemolysis, elevated liver enzyme levels and a low platelet count, is an obstetric complication that is frequently misdiagnosed at initial presentation. Many investigators consider the syndrome to be a variant of preeclampsia, but it may be a separate entity. The pathogenesis of HELLP syndrome remains unclear. Early diagnosis is critical because the morbidity and mortality rates associated with the syndrome have been reported to be as high as 25 percent. Platelet count appears to be the most reliable indicator of the presence of HELLP syndrome. The D-dimer test may be a useful tool for the early identification of patients with preeclampsia who may develop severe HELLP syndrome. The mainstay of therapy is supportive management, including seizure prophylaxis and blood pressure control in patients with hypertension. Women remote from term should be considered for conservative management, whereas those at term should be delivered. Some patients require transfusion of blood products, and most benefit from corticosteroid therapy. Rarely, patients with refractory HELLP syndrome require plasmapheresis.
The acronym HELLP was coined in 1982 to describe a syndrome consisting of hemolysis, elevated liver enzyme levels and low platelet count.1 The syndrome has been considered a variant of preeclampsia, but it can occur on its own or in association with preeclampsia. Pregnancy-induced hypertension, preeclampsia and HELLP syndrome are related and overlap in their presentations. Because of the serious associated morbidity and mortality, family physicians who provide maternity care need to be aware of HELLP syndrome so that they can identify it early.
Etiology and Pathogenesis
The pathogenesis of HELLP syndrome is not well understood. The findings of this multisystem disease are attributed to abnormal vascular tone, vasospasm and coagulation defects.2 To date, no common precipitating factor has been found. The syndrome seems to be the final manifestation of some insult that leads to microvascular endothelial damage and intravascular platelet activation. With platelet activation, thromboxane A and serotonin are released, causing vasospasm, platelet agglutination and aggregation, and further endothelial damage.2 Thus begins a cascade that is only terminated with delivery.
The hemolysis in HELLP syndrome is a microangiopathic hemolytic anemia. Red blood cells become fragmented as they pass through small blood vessels with endothelial damage and fibrin deposits. The peripheral smear may reveal spherocytes, schistocytes, triangular cells and burr cells. The elevated liver enzyme levels in the syndrome are thought to be secondary to obstruction of hepatic blood flow by fibrin deposits in the sinusoids. This obstruction leads to periportal necrosis and, in severe cases, intrahepatic hemorrhage, subcapsular hematoma formation or hepatic rupture. The thrombocytopenia has been attributed to increased consumption and/or destruction of platelets.
Although some investigators speculate that disseminated intravascular coagulopathy (DIC) is the primary process in HELLP syndrome, most patients show no abnormalities on coagulation studies. Patients who develop DIC generally do so in the setting of well-developed HELLP syndrome. All patients with HELLP syndrome may have an underlying coagulopathy that is usually undetectable.
Epidemiology and Risk Factors
HELLP syndrome occurs in approximately 0.2 to 0.6 percent of all pregnancies.3 In comparison, preeclampsia occurs in 5 to 7 percent of pregnancies.3 Superimposed HELLP syndrome develops in 4 to 12 percent of women with preeclampsia or eclampsia.3 When preeclampsia is not present, diagnosis of the syndrome is often delayed.4
The risk factors for HELLP syndrome differ from those associated with preeclampsia (Table 1). The syndrome generally presents in the third trimester of pregnancy, although it occurs at less than 27 weeks of gestation in an estimated 11 percent of patients.5 The syndrome presents antepartum in 69 percent of patients and postpartum in 31 percent of patients.2 With postpartum presentation, the onset is typically within the first 48 hours after delivery; however, signs and symptoms may not become apparent until as long as seven days after delivery.
TABLE 1 Comparison of Risk Factors for HELLP Syndrome and Preeclampsia
| HELLP syndrome | Preeclampsia |
|---|---|
| Multiparous | Nulliparous |
| Maternal age greater than 25 years | Maternal age less than 20 years or greater than 45 years |
| White race | Family history of preeclampsia |
| History of poor pregnancy outcome | Minimal prenatal care |
| Diabetes mellitus | |
| Chronic hypertension | |
| Multiple gestation |
HELLP = hemolysis, elevated liver enzyme levels and low platelet count.
Clinical Presentation
The vague nature of the presenting complaints can make the diagnosis of HELLP syndrome frustrating to physicians. Approximately 90 percent of patients present with generalized malaise, 65 percent with epigastric pain, 30 percent with nausea and vomiting, and 31 percent with headache.3 Because early diagnosis of this syndrome is critical, any pregnant woman who presents with malaise or a viral-type illness in the third trimester should be evaluated with a complete blood cell count and liver function tests.6
The physical examination may be normal in patients with HELLP syndrome. However, right upper quadrant tenderness is present in as many as 90 percent of affected women.2 Edema is not a useful marker because swelling is a factor in up to 30 percent of normal pregnancies.3 Hypertension and proteinuria may be absent or mild. The differential diagnosis of HELLP syndrome includes acute fatty liver of pregnancy, thrombotic thrombocytopenic purpura and hemolytic uremic syndrome.
Because of the variable nature of the clinical presentation, the diagnosis of HELLP syndrome is generally delayed for an average of eight days.7 Many woman with this syndrome are initially misdiagnosed with other disorders, such as cholecystitis, esophagitis, gastritis, hepatitis or idiopathic thrombocytopenia.3 In one retrospective chart review of patients with HELLP syndrome, only two of 14 patients entered the hospital with the correct diagnosis.7
Diagnostic Tests
The three chief abnormalities found in HELLP syndrome are hemolysis, elevated liver enzyme levels and a low platelet count. The hematocrit may be decreased or normal and is typically the last of the three abnormalities to appear. The finding of a decreased serum haptoglobin level may confirm ongoing hemolysis when the hematocrit is normal.8 The serum transaminase levels may be elevated to as high as 4,000 U per L, but milder elevations are typical. Platelet counts can drop to as low as 6,000 per mm3 (6 × 109 per L), but any platelet count less than 150 per mm3 (150 × 109 per L) warrants attention. Unless DIC is present, the prothrombin time, partial thromboplastin time and fibrinogen level are normal in patients with HELLP syndrome. In a patient with a plasma fibrinogen level of less than 300 mg per dL (3 g per L), DIC should be suspected, especially if other laboratory abnormalities are also present.
Proteinuria and an increased uric acid concentration are useful in diagnosing preeclampsia but not HELLP syndrome.9 The platelet count is the best indicator of the latter. Therefore, HELLP syndrome should be suspected in any patient who shows a significant drop in the platelet count during the antenatal period.10 A positive D-dimer test in the setting of preeclampsia has recently been reported to be predictive of patients who will develop HELLP syndrome.11 The D-dimer is a more sensitive indicator of subclinical coagulopathy and may be positive before coagulation studies are abnormal.
Classification
Two classification systems are used for HELLP syndrome. The first is based on the number of abnormalities that are present. In this system, patients are classified as having partial HELLP syndrome (one or two abnormalities) or full HELLP syndrome (all three abnormalities). Women with full HELLP syndrome are at higher risk for complications, including DIC, than women with the partial syndrome. Consequently, patients with the full syndrome should be considered for delivery within 48 hours, whereas those with partial HELLP syndrome may be candidates for more conservative management.12
Alternatively, HELLP syndrome can be classified on the basis of platelet count nadir: class I, less than 50,000 per mm3 (50 × 109 per L); class II, 50,000 to less than 100,000 per mm3 (50 to 100 × 109 per L); and class III, 100,000 to 150,000 per mm3 (100 to 150 × 109 per L).13 Patients with class I HELLP syndrome are at higher risk for maternal morbidity and mortality than patients with class 2 or 3 HELLP syndrome.5
Management
Once the diagnosis of HELLP syndrome has been established, the best markers to follow are the maternal lactate dehydrogenase level and the maternal platelet count.14 Laboratory abnormalities typically worsen after delivery and peak at 24 to 48 hours postpartum.14 The peak lactate dehydrogenase level signals the beginning of recovery and subsequent normalization of the platelet count.14 The platelet count nadir is somewhat predictive for hemorrhagic complications. The incidence of hemorrhagic complications is higher when platelet counts are less than 40,000 per mm3 (40 × 109 per L).15 However, hepatic imaging and liver biopsy have shown that laboratory abnormalities do not correlate with the severity of HELLP syndrome.16,17 Therefore, patients with HELLP syndrome who complain of severe right upper quadrant pain, neck pain or shoulder pain should be considered for hepatic imaging regardless of the severity of the laboratory abnormalities, to assess for subcapsular hematoma or rupture.17
Prompt recognition of HELLP syndrome and timely initiation of therapy are vital to ensure the best outcome for mother and fetus. When the syndrome was first described, prompt delivery was recommended.1 Recent research suggests that morbidity and mortality do not increase when patients with HELLP are treated conservatively.18 The treatment approach should be based on the estimated gestational age and the condition of the mother and fetus (Figure 1).19
FIGURE 1. Management of HELLP Syndrome*
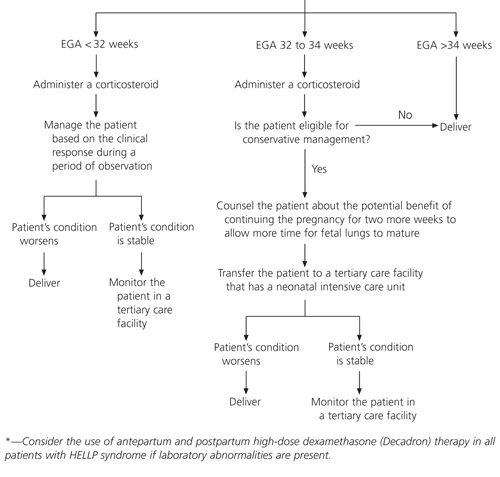
Suggested approach to the management of patients with HELLP syndrome. (HELLP = hemolysis, elevated liver enzyme levels and low platelet count; EGA = estimated gestational age)
Adapted with permission from Sibai BM, Frangieh AY. Management of severe preeclampsia. Curr Opin Obstet Gynecol 1996;8:110–3.
Patients with HELLP syndrome may be eligible for conservative management if hypertension is controlled at less than 160/110 mm Hg, oliguria responds to fluid management and elevated liver function values are not associated with right upper quadrant or epigastric pain. One study20 found that pregnancy was prolonged by an average of 15 days when conservative management (i.e., bed rest, fluids and close observation) was used in patients who were at less than 32 weeks of gestation. Maternal morbidity was not increased. For infants, the prolongation of pregnancy translated into less time in the neonatal intensive care unit, a decreased incidence of necrotizing enterocolitis and a decreased incidence of respiratory distress syndrome.20 Women treated conservatively should be managed in a tertiary care center that has a neonatal intensive care unit and a perinatologist available for consultation.
In the past, delivery in patients with HELLP syndrome was routinely accomplished by cesarean section. Patients with severe HELLP syndrome, superimposed DIC or a gestation of less than 32 weeks should be delivered by cesarean section. A trial of labor is appropriate in patients with mild to moderate HELLP syndrome who are stable, have a favorable cervix and are at 32 weeks of gestation or greater.2
Patients with HELLP syndrome should be routinely treated with corticosteroids. The antenatal administration of dexamethasone (Decadron) in a high dosage of 10 mg intravenously every 12 hours has been shown to markedly improve the laboratory abnormalities associated with HELLP syndrome.21 Patients treated with dexamethasone exhibit longer time to delivery; this facilitates maternal transfer to a tertiary care center and postnatal maturity of fetal lungs.
Steroids given antenatally do not prevent the typical worsening of laboratory abnormalities after delivery. However, laboratory abnormalities resolve more quickly in patients who continue to receive steroids postpartum.10 Corticosteroid therapy should be instituted in patients with HELLP syndrome who have a platelet count of less than 100,000 per mm3 (100 × 109 per L) and should be continued until liver function abnormalities are resolving and the platelet count is greater than 100,000 per mm3 (100 × 109 per L).
Patients with HELLP syndrome should be treated prophylactically with magnesium sulfate to prevent seizures, whether hypertension is present or not. A bolus of 4 to 6 g of magnesium sulfate as a 20 percent solution is given initially. This dose is followed by a maintenance infusion of 2 g per hour. The infusion should be titrated to urine output and magnesium level. Patients should be observed for signs and symptoms of magnesium toxicity. If toxicity occurs, 10 to 20 mL of 10 percent calcium gluconate should be given intravenously.
Antihypertensive therapy should be initiated if blood pressure is consistently greater than 160/110 mm Hg despite the use of magnesium sulfate. This reduces the risk of maternal cerebral hemorrhage, placental abruption and seizure. The goal is to maintain diastolic blood pressure between 90 and 100 mm Hg. The most commonly used antihypertensive agent has been hydralazine (Apresoline), which is given intravenously in small incremental doses of 2.5 to 5 mg (with 5 mg as the initial dose) every 15 to 20 minutes until the desired blood pressure is achieved. Labetolol (Normodyne) and nifedipine (Procardia) have also been used with success.
Because of reported potentiation of effect, care should be taken when nifedipine and magnesium sulfate are given concurrently.22 Diuretics may compromise placental perfusion and therefore are not used to control blood pressure in patients with HELLP syndrome. A hypertensive crisis may be treated with a continuous infusion of nitroglycerin or sodium nitroprusside (Nipride).
Between 38 and 93 percent of patients with HELLP syndrome receive some form of blood product.15 Patients with a platelet count greater than 40,000 per mm3 (40 × 109 per L) are unlikely to bleed. These patients do not require transfusion unless the platelet count drops to less than 20,000 per mm3 (20 × 109 per L). Patients who undergo cesarean section should be transfused if their platelet count is less than 50,000 per mm3 (50 × 109 per L). Prophylactic transfusion of platelets at delivery does not reduce the incidence of postpartum hemorrhage or hasten normalization of the platelet count.15 Patients with DIC should be given fresh frozen plasma and packed red blood cells.
The laboratory abnormalities in HELLP syndrome typically worsen after delivery and then begin to resolve by three to four days postpartum.14 Plasmapheresis has been successful in patients with severe laboratory abnormalities (i.e., a platelet count of less than 30,000 per mm3 [30 × 109 per L] and continued elevation of liver function values) who have required repeat transfusions to maintain their hematocrit at 72 hours postpartum. In these patients, plasmapheresis has resulted in an increase in the platelet count and a decrease in the lactate dehydrogenase level.23–25
ANESTHESIA CONSIDERATIONS
Pain relief with intravenous narcotics and local anesthesia is acceptable but certainly not optimal for pain control. Epidural anesthesia has been controversial but is the technique of choice when it can be accomplished safely.26 Insertion of an epidural catheter is generally safe in patients with a platelet count greater than 100,000 per mm3 (100 × 109 per L), normal coagulation studies and a normal bleeding time.26 General anesthesia can be used when regional anesthesia is considered unsafe.
COMPLICATIONS
The mortality rate for women with HELLP syndrome is approximately 1.1 percent.5 From 1 to 25 percent of affected women develop serious complications such as DIC, placental abruption, adult respiratory distress syndrome, hepatorenal failure, pulmonary edema, subcapsular hematoma and hepatic rupture. A significant percentage of patients receive blood products.5
Infant morbidity and mortality rates range from 10 to 60 percent, depending on the severity of maternal disease.3 Infants affected by HELLP syndrome are more likely to experience intrauterine growth retardation and respiratory distress syndrome.27
Prognosis
Patients who have had HELLP syndrome should be counseled that they have a 19 to 27 percent risk of developing the syndrome in subsequent pregnancies.28 They also have up to a 43 percent risk of developing preeclampsia in another pregnancy.28 Patients with class I HELLP syndrome have the highest risk of recurrence.28 When the syndrome recurs, it tends to develop later in gestation and is generally less severe after two episodes. Patients who have had HELLP syndrome may subsequently use oral contraceptive pills safely.29 Patients who develop atypical early-onset preeclampsia or HELLP syndrome should be screened for the presence of antiphospholipid antibodies.30
Much controversy surrounds the use of acetylsalicylic acid (aspirin) or calcium to prevent preeclampsia. To date, neither calcium nor aspirin has been specifically studied in patients with HELLP syndrome. Although one study suggested that aspirin therapy might be helpful in selected patients with early-onset severe preeclampsia,31 a large, double-blind, randomized, controlled trial failed to demonstrate benefit for this approach.32 In this recently released large study, aspirin therapy did not reduce the incidence of preeclampsia or improve perinatal outcomes in pregnant women at high risk for this complication of pregnancy.32 Likewise, several studies suggested that calcium might be useful in preventing preeclampsia in high-risk patients.33,34 Yet a recent large, multicenter study found that calcium supplementation during pregnancy did not prevent preeclampsia or adverse perinatal outcomes.35

