Pneumocystis carinii pneumonia (PCP) is an opportunistic infection that occurs in immunosuppressed populations, primarily patients with advanced human immunodeficiency virus infection. The classic presentation of nonproductive cough, shortness of breath, fever, bilateral interstitial infiltrates and hypoxemia does not always appear. Diagnostic methods of choice include sputum induction and bronchoalveolar lavage. The drug of choice for treatment and prophylaxis is trimethoprim-sulfamethoxazole, but alternatives are often needed because of adverse effects or, less commonly, treatment failure. Adjunctive corticosteroid therapy improves survival in moderate to severe cases. Complications such as pneumothorax and respiratory failure portend poorer survival. Prophylaxis dramatically lowers the risk of disease in susceptible populations. Although PCP has declined in incidence in the developed world as a result of prophylaxis and effective antiretroviral therapy, its diagnosis and treatment remain challenging.
Before the epidemic of acquired immunodeficiency syndrome (AIDS) in the early 1980s, Pneumocystis carinii pneumonia (PCP) was a rare infection that occurred in immunosuppressed patients with protein malnutrition or acute lymphocytic leukemia, or in patients receiving corticosteroid therapy. This opportunistic infection is now most commonly associated with advanced human immunodeficiency virus (HIV) infection.
The appearance of PCP in previously healthy gay men was one of the initial signs of the emergence of AIDS.1 Before the widespread use of prophylaxis, PCP was the AIDS-defining illness in 60 percent of cases and eventually affected 80 percent of patients with AIDS.2,3 Earlier diagnosis of HIV, antiretroviral therapy and effective prophylaxis have all contributed to a 75 percent decline in cases,4 although PCP remains the most common AIDS-defining illness. Intravenous drug users, noncompliant patients and persons whose HIV serostatus is unknown are at particularly high risk. Intolerance of antipneumocystis agents is common, making management challenging.
Controversy exists over whether PCP represents reactivation of infection acquired early in life or whether repeated exposure and reinfection cause the disease. Experiments in immunosuppressed animals and reports of case clusters support the latter theory.5 The organism is acquired by inhalation and adheres to type I alveolar cells. Proliferation produces a foamy, eosinophilic exudate that fills the alveolar spaces, leading to decreased oxygenation, a thickened interstitium and, eventually, fibrosis.6
Risk Factors and Clinical Presentation
Defective T-cell immunity is the primary risk factor for PCP. Associated clinical signs are well defined in HIV infection and reflect the degree of CD4+ cell depletion7,8 (Table 1).
TABLE 1 Risk Factors for PCP in HIV-Positive Patients
| CD4+ T-lymphocyte cell count < 200 per mm3 (200 × 106 per L)* |
| Unexplained fever of > 37.7°C (100°F) for > two weeks* |
| History of oropharyngeal candidiasis* |
| Previous episode of PCP* |
| Other AIDS-defining illness |
PCP = Pneumocystis carinii pneumonia; HIV = human immunodeficiency syndrome; AIDS = acquired immunodeficiency syndrome.
*—Risk defined in 1997 U.S. Public Health Service guidelines.
The signs and symptoms of PCP can be subtle. The diagnosis requires a heightened index of suspicion because risk factors for HIV may be denied, remote or unknown. The classic triad of fever, exertional dyspnea and nonproductive cough is noted in just over 50 percent of cases. However, almost all patients with PCP have at least two of the following: fever, cough, dyspnea, lactate dehydrogenase (LDH) level of more than 460 U per L or an arterial partial pressure of oxygen (PaO2) of less than 75 mm Hg. Symptoms typically progress slowly, over weeks to months; careful questioning may reveal longstanding, progressive exertional dyspnea (in contrast to HIV-seronegative patients, who typically have a more rapid onset9).
Physical findings are also nonspecific. Auscultation may reveal fine dry rales or may be unremarkable. In 2 to 6 percent of cases, PCP may present with a spontaneous pneumothorax.10 Extrapulmonary pneumocystosis occurs rarely, involving organs such as the heart, skin, spleen, thyroid or eyes.
Elevated serum LDH is not specific enough to distinguish PCP from other types of pneumonia, but the degree of elevation may provide evidence of the severity of the illness. A decrease in oxygen saturation as measured by pulse oximetry during exercise suggests PCP, especially in the patient who has minimal symptoms, does not appear acutely ill and has an unimpressive chest radiograph. When blood gas analysis reveals hypoxemia or a widened alveolar-to-arterial oxygen difference ([A-a]Do2), the prognostic and therapeutic implications are unfavorable (Table 2).
TABLE 2 Use of [A-a]Do2 to Determine PCP Severity
A. Calculation of alveolar-arterial oxygen difference
| ||
| B. Grading severity of PCP by oxygenation | ||
| Severity | [A-a]Do2(mm Hg) | Pao2(mm Hg) |
| Mild | < 35 | > 70 |
| Moderate | 35 to 45 | > 70 |
| Severe | > 45 | > 50 |
[A-a]Do2 = alveolar-to-arterial oxygen difference; PCP = Pneumocystis carinii pneumonia; Flo2 = fraction of inspired oxygen; Paco2 = arterial partial pressure of carbon dioxide; Pao2 = arterial partial pressure of oxygen.
The chest film typically shows diffuse interstitial or perihilar infiltrates but can be normal in at least one third of cases11 (Figures 1a and 1b). Pneumatoceles increase the risk of pneumothorax.10 Less commonly, lobar infiltrates, effusions or cavitary lesions mimic other pulmonary processes. Many other diseases may have a similar presentation, including mycobacterial, fungal, viral or bacterial pneumonias, heart failure, Kaposi's sarcoma and pulmonary emboli.
FIGURE 1A.
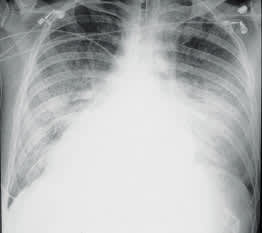
Bilateral interstitial infiltrates in an HIV-infected patient with Pneumocystis carinii pneumonia (PCP).
FIGURE 1B.
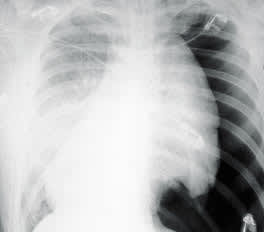
Ventilator-associated right-sided pneumothorax in the same patient.
Diagnosis
P. carinii cannot be routinely cultured and is identified by stains demonstrating the cyst wall or the trophozoite (Figure 2). Before the emergence of AIDS, PCP was diagnosed by open lung biopsy. Less invasive procedures—sputum induction and bronchoalveolar lavage—are now the methods of choice12 (Table 3).
FIGURE 2.
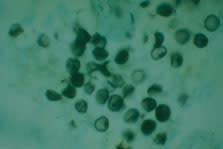
Methenamine silver stain of a bronchoalveolar lavage specimen showing a cluster of P. carinii cysts.
TABLE 3 Comparison of Induced Sputum and Bronchoalveolar Lavage
| Induced sputum | Bronchoalveolar lavage |
|---|---|
| Nebulized saline inhaled by patient to promote deep cough | Saline instilled through bronchoscope wedged in airway and fluid withdrawn |
| Inexpensive; noninvasive | More expensive, more invasive, risk of Periprocedural sedation, requires skilled personnel |
| Specimen processing more complex, may delay diagnosis of another pathogen | Larger samples can be sent for staining and can be used to diagnose other infections (bacterial, fungal, viral and mycobacterial cultures) |
| Less sensitive | > 95 percent sensitive |
In a center with experienced staff, induced-sputum examination rapidly diagnosed PCP in 80 percent of confirmed cases,13 although rates vary greatly, with sensitivities ranging from 30 to 90 percent.12 Bronchoscopy with bronchoalveolar lavage is useful because it samples the alveolar contents with a sensitivity that exceeds 95 percent. Lavage of two or more segments, particularly in the upper lobes, increases the yield.14 Because transbronchial biopsy carries a 10 percent risk of pneumothorax, it is usually reserved for use in situations in which bronchoalveolar lavage fails to reveal PCP in a patient with a compatible presentation, or when another process, such as tuberculosis, fungal infection or neoplasm, is suspected.13 These diagnostic methods may be less satisfactory in HIV-seronegative patients, because these patients have fewer organisms and a more pronounced inflammatory response.15
It is crucial to make a diagnosis as quickly as possible so that targeted therapy can be initiated. Some experts advocate employing empiric therapy when the clinical presentation is compatible with PCP. Because of the many variables in presentation and radiographic findings, empiric therapy involves the risk of unnecessary (and potentially toxic) treatment and the possibility of overlooking another important diagnosis, such as tuberculosis. In a large retrospective review,16 it was found that patients treated empirically had a significantly higher mortality rate than those who underwent diagnostic bronchoscopy. In a series of 894 AIDS patients with fever and respiratory symptoms evaluated by bronchoalveolar lavage, PCP was found in only one half—and of these, 20 percent had coexistent infections.17 It is certainly acceptable to start PCP treatment while awaiting definitive diagnostic testing, without compromising its accuracy.
Management of PCP
Available therapies have many adverse effects, and treatment is more often limited by toxicity than by lack of response. Patients with AIDS require three weeks of treatment for PCP, compared with two weeks in other populations.9 Hospitalized patients should receive intravenous therapy until they improve enough to reliably absorb oral medication; those with mild disease can be treated orally from the outset. Table 4 presents suggested treatment choices based on the authors' interpretation of clinical trial results, the severity of the episode and the potential adverse effects. All patients should be monitored closely for signs of toxicity and clinical deterioration.
TABLE 4 Suggested Hierarchy of Treatment Choices for PCP
| Mild to moderate PCP (oral regimens) | ||
| First choice | Trimethoprim-sulfamethoxazole (Bactrim, Septra) | |
| Second choice | Trimethoprim (Proloprim) and dapsone | |
| or | ||
| Clindamycin (Cleocin) and primaquine | ||
| Third choice | Atovaquone (Mepron) | |
| Moderate to severe PCP (IV regimens) | ||
| First choice | Trimethoprim-sulfamethoxazole | |
| Second choice | Trimetrexate/leucovorin and oral dapsone | |
| or | ||
| Clindamycin (Cleocin phosphate) and oral primaquine | ||
| Third choice | Pentamidine | |
note:Alternative regimens are considered to be equivalent or near-equivalent choices.
PCP = Pneumocystis carinii pneumonia; IV = intravenously.
The mainstay of treatment is trimethoprim-sulfamethoxazole (Bactrim, Septra), given intravenously or orally (Table 5). Trimethoprim-sulfamethoxazole sequentially inhibits two enzymes in folate metabolism essential for DNA synthesis: dihydrofolate reductase (DHFR) and dihydropteroate synthetase (DHPS).18 Treatment failure occurs in 9 to 20 percent of cases, depending on severity.19,20 However, adverse effects are reported in more than one half of cases, and treatment-limiting toxicities may occur in up to one third.19 Common problems include nausea and vomiting, maculopapular rash, bone marrow suppression, hepatitis and drug fever20 (Table 4). Management of adverse effects by means of antiemetics, antihistamines or antipyretics may enable completion of a course of therapy.
TABLE 5 Drugs for Treatment of PCP
| Regimen | Dosage | Common adverse effects | Cost* |
|---|---|---|---|
| Trimethoprim-sulfamethoxazole (Bactrim, Septra) | 5 mg per kg of trimethoprim component every 8 hours, IV or orally (for most patients, oral dosage is 2 double-strength tablets three times daily) | Maculopapular rash, fever, bone marrow suppression, hepatitis, nausea, vomiting, hyperkalemia | $137, generic† |
| 8, brand‡ | |||
| Pentamidine | 4 mg per kg IV once daily, infused over 60 minutes | Hypo- or hyperglycemia and subsequent diabetes mellitus, arrhythmias, QT interval prolongation, leukopenia, pancreatitis, bone marrow suppression, hepatitis, fever | 99, brand |
| Trimetrexate (Neutrexin) and leucovorin | Trimetrexate§: for patients < 50 kg—1.5 mg per kg per day IV; for patients 50 to 80 kg—1.2 mg per kg per day IV; for patients > 80 kg—1.0 mg per kg per day IV | Neutropenia, thrombocytopenia | 264, brand |
| Leucovorin§: for patients < 50 kg—0.8 mg per kg IV or orally every 6 hours; for patients ≥50 kg—0.5 mg per kg IV or orally every 6 hours (round off to next highest dosage when using leucovorin orally); continue leucovorin for 72 hours after last trimetrexate dose. | 20 to 70, generic† 98 to 163, generic‡ | ||
| May add dapsone, 100 mg per day orally. | 0.20 to 0.30, generic | ||
| Trimethoprim (Proloprim) and dapsone | Trimethoprim, 5 mg per kg orally every 8 hours and dapsone, 100 mg orally once daily | Nausea, vomiting, fever, rash, bone marrow suppression, hepatitis, hemolysis, methemoglobinemia | 7, brand (2 to 3, generic) |
| 0.20 to 0.30, generic | |||
| Clindamycin (Cleocin) and primaquine | Clindamycin, 600 to 900 mg IV or 300 to 400 mg orally every 6 to 8 hours Primaquine base, 15 to 30 mg orally once daily∥ | Rash, anemia, neutropenia, methemoglobinemia, hemolysis | 42, brand (2 to 3, generic)† 10, brand‡ (3 to 7, generic) 0.75, brand |
| Atovaquone (Mepron) | 750 mg of suspension orally three times daily with fatty foods (do not use in patients with diarrhea or malabsorption) | Nausea, vomiting, rash | 42, brand |
PCP = Pneumocystis carinii pneumonia; IV = intravenously.
*—Estimated cost to the pharmacist based on average wholesale prices (rounded to the nearest dollar for prices above $1) for one day of treatment based on a patient weighing 70 kg (154 lb) in Red book. Montvale, N.J.: Medical Economics Data, 1999. Cost to the patient may be greater, depending on prescription filling fee.
†—IV.
‡—Oral.
§—Updated basis for dosing.
∥—One tablet (26.3 mg) is the equivalent of 15 mg base.
Intravenous pentamidine may be somewhat less effective than trimethoprim-sulfamethoxazole for the treatment of moderate to severe PCP.20 It is usually reserved for use in patients who do not respond to, or cannot tolerate, trimethoprim-sulfamethoxazole. Serious adverse effects include nephrotoxicity, hyperglycemia and hypoglycemia, pancreatitis, arrhythmias (including torsade de pointes) and the subsequent development of frank diabetes.18 It is important to monitor blood glucose and creatinine levels and to watch for QT prolongation, especially during the last two weeks of therapy.
Trimetrexate (Neutrexin) is a significantly more potent inhibitor of DHFR than trimethoprim (Proloprim),18 so potent that hematopoietic cells must be protected through the coadministration of leucovorin. Although trimetrexate is significantly less toxic than trimethoprim-sulfamethoxazole, it is also less effective.21 Because it is administered once daily, trimetrexate can be used in outpatients even though it is given intravenously. To mimic the sequential enzyme blockade provided by trimethoprim-sulfamethoxazole, dapsone (100 mg orally) can be added to the regimen.
Besides trimethoprim-sulfamethoxazole, several alternative treatment regimens can be used in patients with mild or moderate PCP. Therapy with dapsone and trimethoprim duplicates the sequential enzyme blockade of trimethoprim-sulfamethoxazole.19 Although dapsone can trigger hemolytic anemia in patients with absolute (homozygous) glucose-6-phosphate dehydrogenase (G6PD) deficiency, black patients typically have relative (heterozygous) G6PD deficiency and tolerate dapsone therapy without significant hemolysis.
Clindamycin (Cleocin) plus primaquine is another useful oral combination. Anemia is a common complication of this therapy, but it is rarely due to hemolysis. Rash, which typically appears in the second week of use, may be severe and is more common than rash related to trimethoprim-sulfamethoxazole.19
When trimethoprim-sulfamethoxazole, dapsone-trimethoprim and clindamycin-primaquine were compared in patients with mild-to-moderate PCP, no significant differences were found in terms of either therapeutic failure or treatment-limiting toxicity; 54 percent of the subjects did not complete a full course of assigned treatment, primarily because of toxicity. Different problems occurred: the trimethoprim-sulfamethoxazole group had a higher incidence of hepatitis; the dapsone-trimethoprim group had more nausea and vomiting; and the clindamycin-primaquine group had more anemia than the other groups.19 These results suggest that treatment selection may be tailored to the individual patient's preexisting laboratory abnormalities or other medical problems.
Atovaquone (Mepron), an antiprotozoal agent, has good activity against P. carinii but is limited by its modest and unpredictable bioavailability. Although it is less effective than either trimethoprim-sulfamethoxazole or pentamidine, it is better tolerated.22,23
Despite effective antimicrobial therapy, mild to moderate episodes of PCP still carry a mortality risk of up to 9 percent.22 Determination of [A-a]DO2 is critical (Table 2) because the degree of impairment is the most important prognostic indicator. Almost all patients experience some deterioration in oxygenation within the first few days of therapy, probably because of the inflammatory response triggered by dying organisms.24 Although this decrease is not clinically important in mild disease, patients with poor pulmonary reserve may develop respiratory failure.
Administration of corticosteroids within the first 72 hours of anti-Pneumocystis treatment helps to prevent respiratory failure and death in AIDS patients.24 All patients with an [A-a]DO2 greater than 35 mm Hg or a PaO2 of less than 70 mm Hg should receive cortico-steroids when antimicrobial therapy is initiated.25 The standard approach is to use prednisone for 21 days (Table 6). Patients who are severely ill or unable to take oral medication may be given an equivalent dosage of intravenous methylprednisolone. The risk of reactivating tuberculosis or acquiring another infection appears to be minimal.24
TABLE 6 Adjunctive Corticosteroid Therapy in Moderate to Severe PCP
| Schedule | Dosage |
|---|---|
| Days 1 to 5 | 40 mg of prednisone twice daily |
| Days 6 to 10 | 40 mg of prednisone once daily |
| Days 11 to 21 | 20 mg of prednisone once daily* |
PCP = Pneumocystis carinii pneumonia.
*—No further tapering is necessary.
The prognosis for patients who develop respiratory failure is poor, although those who require mechanical ventilation early are more likely to recover than those who develop late respiratory failure.26 In unventilated patients, pneumothorax results in prolonged hospitalization but does not increase mortality, whereas pneumothorax in ventilator-dependent patients almost always has a fatal outcome.27 Pneumothorax may be bilateral and recurrent, necessitating long-term chest tube placement or pleurodesis.
Determining whether a patient is responding to treatment may be difficult. It is important to remember that oxygenation typically reaches a nadir approximately 72 hours after therapy is begun.24 Because it generally takes several days for clinical improvement, therapy usually should not be changed for at least five to seven days. If a patient is not responding, a bronchoscopy should be performed to rule out the presence of another opportunistic process. P. carinii will still be present but, when no change or an increase in the organism count (if available) is found, the treatment may be failing and should be modified.28
Prevention of PCP
Prophylaxis is also limited by adverse effects, and failures may occur at rates up to 33 percent, depending on the specific agent and the degree of immunosuppression defined by absolute CD4+ count.29 In addition to the factors outlined in the U.S. Public Health Service (PHS) guidelines for PCP prophylaxis,8 patients with other AIDS-defining illnesses should be considered for PCP prophylaxis (Table 1). The advent of potent combination antiretroviral therapy has led to considerably increased CD4+ cell counts in many patients whose cell counts were previously below 200 per mm3 (200 × 106 per L). Recent data indicate that such patients are indeed protected against PCP. The latest revision to the PHS guidelines will recommend discontinuation of primary PCP prophylaxis in patients whose CD4+ cell counts are sustained above 200 per mm3 for at least six months and who have well-controlled HIV viral loads.30–32 Patients who do not have HIV infection but are at high risk for PCP, including those receiving aggressive therapy for leukemia or lymphoma and transplant recipients, should also receive prophylaxis.18
Trimethoprim-sulfamethoxazole is the first choice for prophylaxis (Table 7). In a meta-analysis of 35 randomized trials,33 patients regularly taking trimethoprim-sulfamethoxazole had only a 5 percent likelihood of developing PCP. Even though the dosage used for prophylaxis is much lower than that used for treatment, side effects are still common. Lower dosage regimens should be considered, because they may diminish the risk of side effects without compromising protection.8,33 If a mild reaction to trimethoprim-sulfamethoxazole develops, antihistamines often permit continued use of the agent.29,33 Desensitization has not been studied rigorously but has been useful in some cases.34 Other benefits of the trimethoprim-sulfamethoxazole regimen include protection against toxoplasmosis and bacterial infections.35
TABLE 7 Drugs for Prophylaxis of PCP
| Drug | Dosage | Cost* | ||
|---|---|---|---|---|
| TMP-SMZ (Bactrim, Septra) | 1 double-strength tablet (160 mg TMP/800 mg SMZ) orally once daily | $ 38, brand 11 to 14, generic | ||
| or | ||||
| 1 tablet (80 mg TMP/400 mg SMZ) orally once daily | 23, brand 7 to 10, generic | |||
| or | ||||
| 1 double-strength tablet orally three times every week | 15, brand 5 to 6, generic | |||
| Dapsone | 100 mg orally once daily for PCP. | 5 to 10, generic 3 to 5, generic 3, brand | ||
| In toxoplasma IgG–positive-patients: use 50-mg dapsone orally once daily, plus pyrimethamine (Daraprim), 50 mg orally every week and leucovorin, 25 mg orally every week | 96 to 108, generic | |||
| or | ||||
| dapsone, 200 mg orally every week, plus pyrimethamine, 75 mg orally every week, and leucovorin, 25 mg orally every week | 1 to 3, generic 5, generic 96 to 108, generic | |||
| Aerosolized pentamidine (NebuPent) | 300 mg aerosolized by Respirgard II jet nebulizer every month; administer in negative-airflow room to patients screened for active TB; pretreat with inhaled bronchodilator in patients who experience cough or bronchospasm | 99, brand | ||
| Atovaquone (Mepron) | 750 mg orally twice daily (1,500 mg per day) | 833, brand | ||
TMP = trimethoprim; SMZ = sulfamethoxazole; TB = tuberculosis.
*—Estimated cost to the pharmacist based on average wholesale prices (rounded to the nearest dollar) for one month of treatment in Red book. Montvale,N.J.: Medical Economics Data, 1999. Cost to the patient may be greater, depending on prescription filling fee.
Dapsone is a reasonable alternative to trimethoprim-sulfamethoxazole. Dapsone in combination with weekly pyrimethamine (Daraprim) protects against PCP and toxo-plasmosis.8,36 Atovaquone is considerably more expensive than dapsone and is not yet sanctioned for this use by the Public Health Service.
Aerosolized pentamidine (NebuPent) is another option, although it is the least effective form of prophylaxis, particularly in patients with very low CD4+ cell counts.29 Although this agent is well tolerated, it is expensive and does not protect against systemic pneumocystosis, toxoplasmosis or bacterial infections. Patients should be screened for active tuberculosis before starting aerosolized pentamadine therapy to avoid the unnecessary exposure of others. Pretreatment with inhaled bronchodilators minimizes cough and bronchospasm.
Final Comment
Although advances in the care of HIV-infected patients have dramatically lowered its incidence, PCP remains a challenging clinical problem. Signs and symptoms may be insidious in onset and difficult to distinguish from those of other pathogenic processes. Patients must be monitored carefully for clinical response and drug toxicity. Family physicians need to be familiar with the presentation, diagnosis, treatment and prevention of PCP, particularly as the AIDS epidemic continues to expand beyond familiar risk groups and geographic locations.

