The Centers for Disease Control and Prevention has updated its 1997 report consolidating recommendations for the prevention of opportunistic diseases in patients with human immunodeficiency virus (HIV) infection. The second part of this three-part adaptation focuses on recommendations for prevention of certain diseases that commonly occur in patients with HIV infection.
This second part of the adapted “Guidelines for the Prevention of Opportunistic Infections in Persons Infected with Human Immunodeficiency Virus” gives recommendations for the prevention of the first episode of certain diseases that may occur in patients with human immunodeficiency virus (HIV) infection. Table 11 (page 442) gives a description of the rating system used by the U.S. Public Health Service (USPHS) and the Infectious Diseases Society of America (IDSA) to rate recommendations. In this system, a letter rating (letters A through E) signifies the strength of the recommendation, and a Roman numeral (numerals I through III) indicates the quality of the evidence supporting that recommendation.
Dosages for prophylaxis to prevent a first episode of opportunistic disease in HIV-infected adults and adolescents are given in Table 2 (page 446); effects of food on drugs used to treat opportunistic infections are given in Table 3 (page 448); effects of medications on drugs used to treat opportunistic infections are given in Table 4 (page 449); effects of opportunistic infection medications on drugs commonly administered to HIV-infected persons are given in Table 5 (page 453); adverse effects of drugs used to manage HIV infection are given in Table 6 (page 456); dosages of drugs for prevention of opportunistic infections in persons with renal insufficiency are given in Table 7(page 457); costs of agents recommended for the prevention of opportunistic infections in adults with HIV infection are given in Table 8 (page 458); and criteria for discontinuing and restarting opportunistic infection prophylaxis in adult patients with HIV infection are given in Table 9 (page 461). Table 10 (page 465) gives the immunologic categories for children with HIV, and Table 11 (page 466) summarizes prophylaxis for prevention of first episodes of opportunistic disease in infants and children with HIV. Figure 1 (page 468) gives the recommended immunization schedule for HIV-infected infants and children.
TABLE 2. Prophylaxis to Prevent the First Episode of Opportunistic Disease in Adults and Adolescents with HIV
| Pathogen | Preventive regimens | |||
|---|---|---|---|---|
| Indication | First choice | Alternatives | ||
| I. Strongly recommended as standard of care | ||||
| Pneumocystis carinii* | CD4+ count < 200 per μL or oropharyngeal candidiasis TMP-SMZ, 1 SS orally every day (AI) | TMP-SMZ, 1 DS orally every day (AI) | Dapsone, 50 mg orally twice daily or 100 mg orally every day (BI); dapsone, 50 mg orally every day plus pyrimethamine, 50 mg orally every week plus leucovorin, 25 mg orally every week (BI); dapsone, 200 mg orally plus pyrimethamine, 75 mg orally plus leucovorin, 25 mg orally every week (BI); aerosolized pentamidine, 300 mg every mo via Respirgard II nebulizer (BI); atovaquone, 1,500 mg orally every day (BI); TMP-SMZ, 1 DS orally three times weekly (BI) | |
| Mycobacterium tuberculosis | ||||
| Isoniazid-sensitive† | TST reaction ≥5 mm or prior positive TST result without treatment or contact with case of active tuberculosis | Isoniazid, 300 mg orally plus pyridoxine, 50 mg orally every day × 9 mo (AII) or isoniazid, 900 mg orally plus pyridoxine, 100 mg orally twice weekly × 9 mo (BI); rifampin, 600 mg plus pyrazinamide, 20 mg per kg orally every day × 2 mo (AI) | Rifabutin, 300 mg orally every day plus pyrazinamide, 20 mg per kg orally every day × 2 mo (BIII); rifampin 600 mg orally every day × 4 mo (BIII) | |
| Isoniazid-resistant | Same; high probability of exposure to isoniazid- resistant tuberculosis | Rifampin 600 mg plus pyrazinamide, 20 mg per kg orally every day × 2 mo (AI) | Rifabutin, 300 mg plus pyrazinamide 20 mg per kg orally every day × 2 mo (BIII); rifampin 600 mg orally every day × 4 mo (BIII); rifabutin, 300 mg orally every day × 4 mo (CIII) | |
| Multidrug- (isoniazid and rifampin) resistant | Same; high probability of exposure to multidrug-resistant tuberculosis | Choice of drugs requires consultation with public health authorities | None | |
| Toxoplasma gondii§ | IgG antibody to Toxoplasma and CD4+ count < 100 per μL | TMP-SMZ, 1 DS orally every day (AII) | TMP-SMZ, 1 SS orally every day (BIII); dapsone, 50 mg orally every day plus pyrimethamine, 50 mg orally every week plus leucovorin, 25 mg orally every week (BI); atovaquone, 1,500 mg orally every day with or without pyrimethamine, 25 mg orally every day plus leucovorin, 10 mg orally every day (CIII) | |
| Mycobacterium avium complex¶ | CD4+ count < 50 per μL | Azithromycin, 1,200 mg orally every week (AI), or clarithromycin, 500 mg orally twice daily (AI) | Rifabutin, 300 mg orally every day (BI); azithromycin, 1,200 mg orally every week plus rifabutin, 300 mg orally every day (CI) | |
| Varicella zoster virus (VZV) | Significant exposure to chickenpox or shingles for patients who have no history of either condition or, if available, negative antibody to VZV | Varicella zoster immune globulin (VZIG), 5 vials (1.25 mL each) IM, administered ≤96 hours after exposure, ideally within 48 hours (AIII) | ||
| II. Generally recommended | ||||
| Streptococcus pneumoniae** | All patients | Pneumococcal vaccine, 0.5 mL IM (CD4+ ≥ 200 per mm3 [BII]; CD4+ < 200 per mm3 [CIII]—might reimmunize if initial immunization was given when CD4+ < 200 per mm3 and if CD4+ increases to > 200 mm3 on HAART [CIII]) | None | |
| Hepatitis B virus †† | All susceptible (anti-HBc-negative) patients | Hepatitis B vaccine: 3 doses (BII) | None | |
| Influenza virus†† | All patients (annually, before influenza season) | Whole or split virus, 0.5 mL IM per year (BIII) | Rimantadine, 100 mg orally twice daily (CIII), or amantadine, 100 mg orally twice daily (CIII) | |
| Hepatitis A virus†† | All susceptible (anti-HAV- negative) patients with chronic hepatitis C | Hepatitis A vaccine: two doses (BIII) | None | |
| III. Not routinely indicated | ||||
| Bacteria | Neutropenia | Granulocyte-colony-stimulating factor (G-CSF), 5 to 10 μg per kg SC every day × 2 to 4 weeks or granulocyte-macrophage colony- stimulating factor (GM-CSF), 250 μg per m2 IV over 2 hours every day × 2 to 4 weeks (CII) | None | |
| Cryptococcus neoformans§§ | CD4+ count < 50 per mm3 | Fluconazole, 100 to 200 mg orally every day (CI) | Itraconazole, 200 mg orally every day (CIII) | |
| Histoplasma capsulatum§§ | CD4+ count < 100 per mm3, endemic geographic area | Itraconazole capsule, 200 mg orally every day (CI) | None | |
| Cytomegalovirus (CMV)¶¶ | CD4+ count < 50 per mm3, and CMV antibody positivity | Oral ganciclovir, 1 g orally three times daily (CI) | None | |
note: Information included in these guidelines might not represent U.S. Food and Drug Administration (FDA) approval or approved labeling for the particular products or indications in question. Specifically, the terms “safe” and “effective” might not be synonymous with the FDA-defined legal standards for product approval. The Respirgard II nebulizer is manufactured by Marquest, Englewood, Colo. Letters and Roman numerals in parentheses after regimens indicate the strength of the recommendation and the quality of evidence supporting it (see Table 1).
HIV = human immunodeficiency virus; Anti-HBc = antibody to hepatitis B core antigen; DS = double-strength tablet; HAART = highly active anti-retroviral therapy; HAV = hepatitis A virus; mo = month; IM = intramuscular; IV = intravenous; SS = single-strength tablet; TMP-SMZ = trimethoprim-sulfamethoxazole; SC = subcutaneous; TST = tuberculin skin test.
*—Prophylaxis should also be considered for persons with a CD4+ percentage of < 14%, for persons with a history of an AIDS-defining illness and, possibly, for those with CD4+ counts > 200 but < 250 per mm3. TMP-SMZ also reduces the frequency of toxoplasmosis and some bacterial infections. Patients receiving dapsone should be tested for glucose-6 phosphate dehydrogenase deficiency. A dosage of 50 mg every day is probably less effective than one of 100 mg every day. The efficacy of parenteral pentamidine (e.g., 4 mg per kg per month) is uncertain. Fansidar (sulfadoxine-pyrimethamine) is rarely used because of severe hypersensitivity reactions. Patients who are being administered therapy for toxoplasmosis with sulfadiazine-pyrimethamine are protected against Pneumocystis carinii pneumonia and do not need additional prophylaxis against PCP.
†—Directly observed therapy is recommended for isoniazid, 900 mg twice weekly; isoniazid regimens should include pyridoxine to prevent peripheral neuropathy. Rifampin should not be administered concurrently with protease inhibitors or nonnucleoside reverse transcriptase inhibitors. Rifabutin should not be given with hard-gel saquinavir or delavirdine; caution is also advised when the drug is coadministered with soft-gel saquinavir. Rifabutin may be administered at a reduced dosage of 150 mg every day with indinavir, nelfinavir, or amprenavir; at a reduced dosage of 150 mg every other day (or 150 mg three times weekly) with ritonavir; or at an increased dosage of 450 mg every day with efavirenz; information is lacking regarding coadministration of rifabutin with nevirapine. Exposure to multidrug-resistant tuberculosis might require prophylaxis with two drugs; consult public health authorities. Possible regimens include pyrazinamide plus ethambutol or a fluoroquinolone.
§—Protection against toxoplasmosis is provided by TMP-SMZ, dapsone plus pyrimethamine and, possibly, by atovaquone. Atovaquone may be used with or without pyrimethamine. Pyrimethamine alone probably provides little, if any, protection.
¶—See footnote above(† ) regarding use of rifabutin with protease inhibitors or nonnucleoside reverse transcriptase inhibitors.
**—Vaccination should be offered to persons who have a CD4+ T-lymphocyte count < 200 per mm3, although the efficacy might be diminished. Revaccination 5 years after the first dose or sooner if the initial immunization was given when the CD4+ count was < 200 per mm3and the CD4+ count has increased to > 200 per mm3on HAART is considered optional. Some authorities are concerned that immunizations might stimulate the replication of HIV. However, one study showed no adverse effect of pneumococcal vaccination on patient survival (McNaghten AD, Hanson DL, Jones JL, Dworkin MS, Ward JW, and the Adult/Adolescent Spectrum of Disease Group. Effects of antiretroviral therapy and opportunistic illness primary chemoprophylaxis on survival after AIDS diagnosis. AIDS 1999;13:1687–95).
††—These immunizations or chemoprophylactic regimens do not target pathogens traditionally classified as opportunistic but should be considered for use in HIV-infected patients as indicated. Data are inadequate concerning clinical benefit of these vaccines in this population, although it is logical to assume that those patients who develop antibody responses will derive some protection. Some authorities are concerned that immunizations might stimulate HIV replication, although for influenza vaccination, a large observational study of HIV-infected persons in clinical care showed no adverse effect of this vaccine, including multiple doses, on patient survival (J. Ward, Centers for Disease Control and Prevention, personal communication, 1999). Hepatitis B vaccine has been recommended for all children and adolescents and for all adults with risk factors for hepatitis B virus (HBV). Rimantadine and amantadine are appropriate during outbreaks of influenza A. Because of the theoretic concern that increases in HIV plasma RNA following vaccination during pregnancy might increase the risk of perinatal transmission of HIV, providers may wish to defer vaccination until after antiretroviral therapy is initiated. For additional information regarding vaccination against hepatitis A and B and vaccination and antiviral therapy against influenza see: CDC. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1996;45(RR-15); CDC. Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1991;40(RR-13); and CDC. Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1999;48(RR-4).
§§—In a few unusual occupational or other circumstances, prophylaxis should be considered; consult a specialist.
¶¶—Acyclovir is not protective against CMV. Valacyclovir is not recommended because of an unexplained trend toward increased mortality observed in persons with AIDS who were being administered this drug for prevention of CMV disease.
TABLE 3. Effects of Food on Drugs Used to Prevent Opportunistic Infections
| Drug | Food effect | Recommendation |
|---|---|---|
| Atovaquone | Bioavailability increased up to threefold with high-fat meal. | Administer with food. |
| Ganciclovir (capsules) | High-fat meal results in 22% increase in the area under the curve (AUC). | Administer with food. |
| Itraconazole (capsules) | Significant increase in bioavailability when taken with a full meal. Grapefruit juice results in 30% decrease in itraconazole AUC. | Administer with food; avoid grapefruit juice or increased itraconazole dosage might be necessary. |
| Itraconazole (solution) | 31% increase in AUC when taken under fasting conditions. | Take without food if possible. |
TABLE 4. Effects of Medications on Drugs Used to Prevent Opportunistic Infections
| Affected drug | Interacting drugs | Mechanism/effect | Recommendation |
|---|---|---|---|
| Atovaquone | Rifampin | Induction of metabolism—decreased drug levels | Concentrations might not be therapeutic; avoid combination or increase atovaquone dose. |
| Clarithromycin | Ritonavir | Inhibition of metabolism—increased drug levels by 77% | No adjustment needed in normal renal function; adjust if creatinine clearance is < 30. |
| Clarithromycin | Nevirapine | Induction of metabolism—decrease in clarithromycin area under the curve (AUC) by 35%, increase in AUC of 14-OH clarithromycin by 27% | Effect on Mycobacterium avium prophylaxis might be decreased; monitor closely. |
| Ketoconazole | Antacids, didanosine, histamine H2-receptor blockers, proton pump inhibitors | Increase in gastric pH that impairs absorption of ketoconazole | Avoid use of ketoconazole with pH-raising agents or use alternative antifungal drug. |
| Quinolone antibiotics (ciprofloxacin, levofloxacin, ofloxacin) | Didanosine, antacids, iron products, calcium products, sucralfate | Chelation that results in marked decrease in quinolone drug levels | Administer interacting drug at least 2 hours after quinolone. |
| Rifabutin | Fluconazole | Inhibition of metabolism—marked increase in rifabutin drug levels | Monitor for rifabutin toxicity such as uveitis, nausea and neutropenia. |
| Rifabutin | Efavirenz | Induction of metabolism—significant decrease in rifabutin AUC | Increase rifabutin dosage to 450 mg daily. |
| Rifabutin | Ritonavir, saquinavir, indinavir, nelfinavir, amprenavir, delavirdine | Inhibition of metabolism—marked increase in rifabutin drug levels | Contraindicated with hard-gel saquinavir (caution also advised with soft-gel saquinavir) and delavirdine; use 1/2 dosage (150 mg daily) with indinavir, nelfinavir, amprenavir; use 1/4 dosage (150 mg every other day) with ritonavir. |
TABLE 5. Effects of Opportunistic Infection Medications on Drugs Commonly Administered to Persons Infected with HIV
| Affected drug | Interacting drugs | Mechanism/effect | Recommendation |
|---|---|---|---|
| Indinavir, saquinavir, ritonavir, nelfinavir, amprenavir | Rifampin | Induction of metabolism—marked decrease in protease inhibitor drug levels | Avoid concomitant use. |
| Delavirdine, nevirapine, efavirenz | Rifampin | Induction of metabolism—marked decrease in drug levels | Avoid concomitant use. |
| Saquinavir (hard-gel), delavirdine | Rifabutin | Induction of metabolism—marked decrease in drug levels | Avoid concomitant use. |
| Terfenadine, astemizole, cisapride | Ketoconazole, itraconazole, fluconazole, clarithromycin | Inhibition of metabolism | Cardiotoxic life-threatening effects possible; avoid concomitant use. |
| Didanosine (ddI) | Ganciclovir | Increases ddI area under the curve by approximately 100% | Clinical significance unknown; monitor for ddI-related adverse effects. |
HIV = human immunodeficiency virus.
TABLE 6. Adverse Effects of Opportunistic Infection Medications Used in the Management of HIV Infection
| Bone marrow suppression | Cidofovir, dapsone, ganciclovir, pyrimethamine, rifabutin, sulfadiazine, trimethoprim- sulfamethoxazole, trimetrexate |
| Diarrhea | Atovaquone, clindamycin |
| Hepatotoxicity | Clarithromycin, fluconazole, isoniazid, itraconazole, ketoconazole, pyrazinamide, rifabutin, rifampin |
| Nephrotoxicity | Amphotericin B, cidofovir, foscarnet, pentamidine |
| Ocular effects | Cidofovir, ethambutol, rifabutin |
| Pancreatitis | Pentamidine, trimethoprim-sulfamethoxazole |
| Peripheral neuropathy | Isoniazid |
| Skin rash | Atovaquone, dapsone, sulfadiazine, trimethoprim-sulfamethoxazole |
HIV = human immunodeficiency virus.
TABLE 7. Dosages of Drugs for Primary Prevention or Maintenance Therapy for Persons with Opportunistic Infections and Renal Insufficiency
| Drug | Normal dosage | CrCl (mL/min/1.73 m2 ) | Adjusted dosage | ||||
|---|---|---|---|---|---|---|---|
| Acyclovir | 200 mg orally three times daily | < 10 | 200 mg every 12 hours | ||||
| 400 mg orally every 12 hours | < 10 | 200 mg every 12 hours | |||||
| 800 mg orally every 4 hours | 10 to 25 | 800 mg every 8 hours | |||||
| 800 mg orally every 4 hours | < 10 | 800 mg every 12 hours | |||||
| Cidofovir | 5 mg per kg IV every other week (with probenecid) | Increase in serum creatinine of 0.3 to 0.4 above baseline | 3 mg per kg | ||||
| Increase in serum creatinine of 0.5 above baseline or 3+ proteinuria | Discontinue | ||||||
| Ciprofloxacin | 500 mg orally every 12 hours | 30 to 50 | 250 to 500 mg every 12 hours | ||||
| < 30 | 250 to 500 mg every 18 hours | ||||||
| Clarithromycin | 500 mg twice daily | < 30 | 1/2 dose (or double interval) | ||||
| Famciclovir | 500 mg orally every 12 hours | 20 to 39 | 250 mg every 12 hours | ||||
| < 20 | 250 mg every 24 hours | ||||||
| Fluconazole | 50 to 400 mg orally every day | 10 to 50 | 1/2 dose | ||||
| < 10 | 1/4 dose | ||||||
| Dialysis | Full dose after dialysis | ||||||
| Foscarnet | 90 to 120 mg per kg IV every day | CrCl*: | Low dose: | High dose: | |||
| > 1.4 | 90 mg every 24 hours | 120 mg every 24 hours | |||||
| 1.0 to 1.4 | 70 mg every 24 hours | 90 mg every 24 hours | |||||
| 0.8 to 1.0 | 50 mg every 24 hours | 65 mg every 24 hours | |||||
| 0.6 to 0.8 | 80 mg every 48 hours | 105 mg every 48 hours | |||||
| 0.5 to 0.6 | 60 mg every 48 hours | 80 mg every 48 hours | |||||
| 0.4 to 0.5 | 50 mg every 48 hours | 65 mg every 48 hours | |||||
| < 0.4 | Not recommended | Not recommended | |||||
| Ganciclovir | Oral: 1 g orally three times daily | 50 to 69 | 1,500 mg every day or 500 mg three times daily | ||||
| 25 to 49 | 1,000 mg every day or 500 mg twice daily | ||||||
| 10 to 24 | 500 mg every day | ||||||
| < 10 | 500 mg three times weekly after dialysis | ||||||
| IV: 5 mg per kg every day or 6 mg per kg every day × 5 days per week | 50 to 69 | 2.5 mg per kg every 24 hours | |||||
| 25 to 49 | 1.25 mg per kg every 24 hours | ||||||
| 10 to 24 | 0.625 mg per kg every 48 hours | ||||||
| < 10 | 0.625 mg per kg three times weekly | ||||||
| TMP-SMZ | 1 DS every day | < 30 | 1/2 dose | ||||
| 1 DS three times weekly | |||||||
| 1 SS every day | |||||||
CrCl = creatinine clearance; TMP-SMZ = trimethoprim-sulfamethoxazole; DS = double-strength tablet; IV = intravenous; SS = single-strength tablet.
*—Creatinine clearance for foscarnet is expressed as mL per min per kg.
TABLE 8. Wholesale Acquisition Costs of Agents Recommended for the Prevention of Opportunistic Infections in Adults Infected with HIV
| Opportunistic pathogen | Drug/vaccine | Dose | Annual cost* per patient |
|---|---|---|---|
| Pneumocystis carinii | Trimethoprim-sulfamethoxazole | 160/800 mg every day | $60 |
| Dapsone | 100 mg every day | 72 | |
| Aerosolized pentamidine | 300 mg every month | 1,185 | |
| Atovaquone | 1,500 mg every day | 10,647 | |
| Mycobacterium avium complex | Clarithromycin | 500 mg twice daily | 2,347 |
| Azithromycin | 1,200 mg once weekly | 1,635 | |
| Rifabutin | 300 mg every day | 3,352 | |
| Cytomegalovirus | Ganciclovir (oral) | 1,000 mg three times daily | 17,269 |
| Ganciclovir implant† | 5,000 | ||
| Ganciclovir (IV) | 5 mg per kg every day | 9,113 | |
| Foscarnet (IV) | 90 to 120 mg per kg every day | 27,960 to 36,770 | |
| Cidofovir (IV) | 375 mg every other week | 19,812 | |
| Fomivirsen (intravitreal) | 1 vial every 4 weeks | 12,000 | |
| Mycobacterium tuberculosis | Isoniazid§ | 300 mg every day | 23 |
| Rifampin | 600 mg every day | 1,540 | |
| Pyrazinamide | 1,500 mg every day | 1,005 | |
| Ethambutol | 900 mg every day | 1,527 | |
| Fungi | Fluconazole | 200 mg every day | 4,267 |
| Itraconazole (capsules) | 200 mg every day | 4,893 | |
| Itraconazole (solution) | 200 mg every day | 5,129 | |
| Ketoconazole | 200 mg every day | 1,230 | |
| Herpes simplex virus | Acyclovir | 400 mg twice daily | 1,300 |
| Famciclovir | 500 mg twice daily | 4,826 | |
| Valacyclovir | 500 mg twice daily | 1,435 | |
| Toxoplasma gondii | Pyrimethamine | 50 mg once weekly | 45 |
| Leucovorin | 25 mg once weekly | 1,248 | |
| Sulfadiazine | 500 mg four times daily | 1,421 | |
| Streptococcus pneumoniae | 23-valent pneumococcal vaccine | 0.5 mL IM once | 13 |
| Influenza virus | Influenza vaccine | 0.5 mL IM once | 5 |
| Hepatitis B virus | Recombinant hepatitis B | 10 to 20 μg IM × 3 | 195 |
| Hepatitis A virus | Hepatitis A vaccine | 1.0 mL IM × 2 | 120 |
| Bacterial infections | G-CSF | 300 μg three times weekly | 25,780 |
| Varicella zoster virus | VZIG | 5 vials (6.25 mL) | 560 |
HIV = human immunodeficiency virus; G-CSF = granulocyte–colony-stimulating factor; IV = intravenous; IM = intramuscular; VZIG = varicella zoster immune globulin.
*—Estimated cost to the pharmacist based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 1999. Cost to the patient will be higher, depending on prescription filling fee.
†—Implant generally lasts 6 to 9 months.
§—Cost for 9 months of therapy.
TABLE 9. Criteria for Discontinuing and Restarting Opportunistic Infection Prophylaxis for Adults with HIV Infection*
| Opportunistic illness | Primary criteria for discontinuing prophylaxis | Secondary criteria for discontinuing prophylaxis | Criteria for restarting prophylaxis |
|---|---|---|---|
| Pneumocystic carinii pneumonia | CD4+ 200 cells per mm3 for > 3 to 6 months (CII) | No criteria recommended for stopping | Same as criteria for initiating (CIII) |
| Disseminated Mycobacterium avium complex | CD4+ > 100 per mm3 for > 3 to 6 months; sustained suppression of HIV plasma RNA (CII) | No criteria recommended for stopping | Same as criteria for initiating (CIII) |
| Toxoplasmosis | No criteria recommended for stopping | No criteria recommended for stopping | Not applicable |
| Cryptococcosis | Not applicable | No criteria recommended for stopping | Not applicable |
| Histoplasmosis | Not applicable | No criteria recommended for stopping | Not applicable |
| Coccidioidomycosis | Not applicable | No criteria recommended for stopping | Not applicable |
| Cytomegalovirus retinitis | Not applicable | CD4+ > 100 to 150 per mm3 for > 3 to 6 months | |
| Durable suppression of HIV plasma RNA | Restart maintenance when CD4+ < 50 to 100 per mm3 (CIII) | ||
| Non–sight-threatening lesion | |||
| Adequate vision in contralateral eye | |||
| Regular ophthalmic examinations (CIII) |
HIV = human immunodeficiency virus.
*—The safety of discontinuing prophylaxis in children whose CD4+ counts have increased in response to highly active antiretroviral therapy has not been studied.
TABLE 10. Immunologic Categories for HIV-Infected Children Based on Age-Specific CD4+ T-Lymphocyte Counts and Percentage of Total Lymphocytes
| Immunologic category | Age | ||
|---|---|---|---|
| ≤ 12 months | 1 to 5 years | 6 to 12 years | |
| Cells per mm3 (%) | Cells per mm3 (%) | Cells per mm3 (%) | |
| 1. No evidence of suppression | ≥ 1,500 (≥25) | ≥ 1,000 (≥25) | ≥500 (≥25) |
| 2. Evidence of moderate suppression | 750 to 1,499 (15 to 24) | 500 to 999 (15 to 24) | 200 to 499 (15 to 24) |
| 3. Severe suppression | < 750 (< 15) | < 500 (< 15) | < 200 (< 15) |
HIV = human immunodeficiency virus.
Adapted from Centers for Disease Control and Prevention. 1994 revised classification system for human immunodeficiency virus infection in children less than 13 years of age. MMWR 1994;43(RR-12).
TABLE 11. Prophylaxis to Prevent the First Episode of Opportunistic Disease in Infants and Children Infected with HIV
| Pathogen | Preventive regimens | ||||
|---|---|---|---|---|---|
| Indication | First choice | Alternatives | |||
| I. Strongly recommended as standard of care | |||||
| Pneumocystis carinii* | HIV-infected or HIV-indeterminate, infants aged 1 to 12 months; HIV-infected children aged 1 to 5 years with CD4+ count < 500 per mm3 or CD4+ percentage < 15%; HIV-infected children aged 6 to 12 years with CD4+ count < 200 per mm3 or CD4+ percentage < 15% | Trimethoprim-sulfamethoxazole (TMP-SMZ), 150/750 mg/m2/day in 2 divided doses orally three times weekly on consecutive days (AII) Acceptable alternative dosage schedules: (AII)
| Dapsone (children aged ≥ 1 month), 2 mg per kg (max 100 mg) orally every day or 4 mg per kg (max 200 mg) orally every day (CII); aerosolized pentamidine (children aged≥5 yr), 300 mg every month via Respirgard II nebulizer (CIII); atovaquone (children aged 1 to 3 months and > 24 months, 30 mg per kg orally every day; children aged 4 to 24 months, 45 mg per kg orally every day) (CII) | ||
| Mycobacterium tuberculosis† | |||||
| Isoniazid-sensitive | TST reaction, ≥ 5 mm or prior positive TST result without treatment or contact with case of active tuberculosis | Isoniazid, 10 to 15 mg per kg (max 300 mg) orally every day × 9 months (AII) or 20 to 30 mg per kg (max 900 mg) orally twice weekly × 9 months (BIII) | Rifampin, 10 to 20 mg per kg (max 600 mg) orally every day × 4 to 6 months (BIII) | ||
| Isoniazid-resistant | Same as above; high probability of exposure to isoniazid-resistant tuberculosis | Rifampin, 10 to 20 mg per kg (max 600 mg) orally every day × 4 to 6 months (BIII) | Uncertain | ||
| Multidrug-(isoniazid and rifampin) resistant | Same as above; high probability of exposure to multidrug- resistant tuberculosis | Choice of drugs requires consultation with public health authorities | None | ||
| Mycobacterium avium complex† | For children aged ≥ 6 years, CD4+ count < 50 per mm3; aged 2 to 6 years, CD4+ count < 75 per mm3; aged 1 to 2 years, CD4+ count < 500 per mm3; aged < 1 year, CD4+ count < 750 per mm3 | Clarithromycin, 7.5 mg per kg (max 500 mg) orally twice daily (AII), or azithromycin, 20 mg per kg (max 1,200 mg) orally every week (AII) | Azithromycin, 5 mg per kg (max 250 mg) orally every day (AII); children aged ≥ 6 years, rifabutin, 300 mg orally every day (BI) | ||
| Varicella zoster virus§ | Significant exposure to varicella or shingles with no history of chickenpox or shingles | Varicella zoster immune globulin (VZIG), 1 vial (1.25 mL) per 10 kg (max 5 vials) IM, administered ≤96 hours after exposure, ideally within 48 hours (AII) | None | ||
| Vaccine-preventable pathogens ¶ | HIV exposure/infection | Routine immunizations (see Figure 1) | None | ||
| II. Generally recommended | |||||
| Toxoplasma gondii** | IgG antibody to Toxoplasma and severe immunosuppression | TMP-SMZ, 150/750 mg per m2 in 2 divided doses orally every day (BIII) | Dapsone (children aged ≥1 month), 2 mg per kg or 15 mg per m2 (max 25 mg) orally every day plus pyrimethamine, 1 mg per kg orally every day plus leucovorin, 5 mg orally every 3 days (BIII) Atovaquone (children aged 1 to 3 months and > 24 months, 30 mg per kg orally every day; 14 to 24 months, 45 mg per kg orally every day) (CIII) | ||
| Varicella zoster virus¶ | HIV-infected children who are asymptomatic and not immunosuppressed | Varicella zoster vaccine (see vaccine-preventable pathogens section of this table) (BII) | None | ||
| Influenza virus¶ | All patients (annually, before influenza season) | Influenza vaccine (see vaccine- preventable pathogens section of this table) (BIII) | Rimantadine or amantadine (during outbreaks of influenza A); children aged 1 to 9 years, 5 mg per kg in 2 divided doses orally every day; ≥10 years, use adult doses (CIII) | ||
| III. Not recommended for use in most children; indicated for use only in unusual circumstances | |||||
| Invasive bacterial infections†† | Hypogammaglobulinemia (i.e., IgG < 400 mg per dL) | IVIG (400 mg per kg every 2 to 4 weeks) (AI) | None | ||
| Cryptococcus neoformans | Severe immunosuppression | Fluconazole, 3 to 6 mg per kg orally every day (CII) | Itraconazole, 2 to 5 mg per kg orally every 12 to 24 hours (CII) | ||
| Histoplasma capsulatum | Severe immunosuppression, endemic geographic area | Itraconazole, 2 to 5 mg per kg orally every 12 to 24 hours (CIII) | None | ||
| Cytomegalovirus (CMV)§§ | CMV antibody positivity and severe immunosuppression | Oral ganciclovir, 30 mg per kg orally three times daily (CII) | None | ||
note: Information included in these guidelines might not represent U.S. Food and Drug Administration (FDA) approval or approved labeling for the particular products or indications in question. Specifically, the terms “safe” and “effective” might not be synonymous with the FDA-defined legal standards for product approval. The Respirgard II nebulizer is manufactured by Marquest, Englewood, Colo. Letters and Roman numerals in parentheses after regimens indicate the strength of the recommendation and the quality of the evidence supporting it (see Table 1).
HIV = human immunodeficiency virus; TST = tuberculosis skin test; IVIG = intravenous immune globulin.; IM = intramuscular.
*—Daily TMP-SMZ reduces the frequency of some bacterial infections. TMP-SMZ, dapsone-pyrimethamine and, possibly, atovaquone (with or without pyrimethamine) appear to protect against toxoplasmosis, although data have not been prospectively collected. When compared with weekly dapsone, daily dapsone is associated with a lower incidence of Pneumocystis carinii pneumonia (PCP) but higher hematologic toxicity and mortality (McIntosh K, Cooper E, Xu J, Mirochnik M, Lindsey J, Jacobus D, et al. Toxicity and efficacy of daily vs. weekly dapsone for prevention of Pneumocystis carinii pneumonia in children infected with HIV. Pediatr Infect Dis J 1999;18:432–9). The efficacy of parenteral pentamidine (e.g., 4 mg per kg every 2 to 4 weeks) is controversial. Patients receiving therapy for toxoplasmosis with sulfadiazine-pyrimethamine are protected against P. carinii pneumonia and do not need TMP-SMZ.
†—Significant drug interactions might occur between rifamycins (rifampin and rifabutin) and protease inhibitors and nonnucleoside reverse transcriptase inhibitors. Consult a specialist.
§—Children routinely being administered intravenous immune globulin (IVIG) should receive VZIG if the last dose of IVIG was administered >21 days before exposure.
¶—HIV-infected and HIV-exposed children should be immunized according to the childhood immunization schedule in this report (see Figure 1), which has been adapted from the January-December 1999 schedule recommended for immunocompetent children by the Advisory Committee on Immunization Practices, the American Academy of Pediatrics and the American Academy of Family Physicians. This schedule differs from that for immunocompetent children in that inactivated poliovirus vaccine (IPV) replaces oral poliovirus vaccine (OPV), and vaccination against influenza (BIII) and Streptococcus pneumoniae (BII) should be offered. MMR should not be administered to severely immunocompromised children (DIII). Vaccination against varicella is indicated only for asymptomatic nonimmunosuppressed children (BII), and rotavirus vaccine is contraindicated in all HIV-infected children (EIII). Once an HIV-exposed child is determined not to be HIV infected, the schedule for immunocompetent children applies.
**—Protection against toxoplasmosis is provided by the preferred antipneumocystis regimens and, possibly, by atovaquone. Atovaquone may be used with or without pyrimethamine. Pyrimethamine alone probably provides little, if any, protection (for definition of severe immunosuppression, see Table 10).
††—Respiratory syncytial virus (RSV) IVIG (750 mg per kg), not monoclonal RSV antibody, may be substituted for IVIG during the RSV season to provide broad anti-infective protection, if this product is available.
§§—Oral ganciclovir results in reduced CMV shedding in CMV-infected children. Acyclovir is not protective against CMV.
Figure 1. Recommended Immunization Schedule for Human Immunodeficiency Virus-Infected Children*
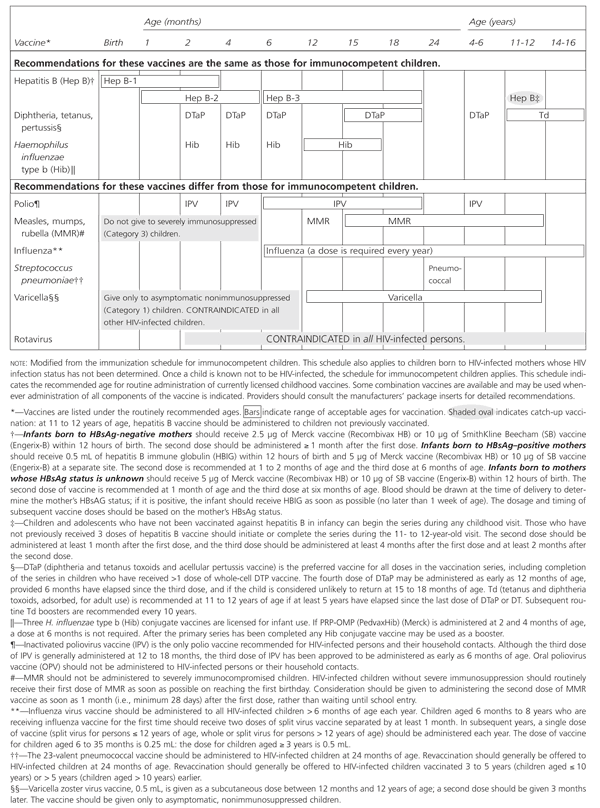
This report is oriented toward the prevention of specific opportunistic infections in HIV-infected persons in the United States and other industrialized countries. Recommendations for use of antiretroviral therapy, which is designed to prevent immunologic deterioration and delay the need for many of the chemoprophylactic strategies described in this report, are published elsewhere,2 as are integrated approaches to the care of HIV-infected persons.3
Disease-Specific Recommendations for the Prevention of the First Episode
PNEUMOCYSTIS CARINII PNEUMONIA
Initiation of Primary Prophylaxis. Adults and adolescents who have HIV infection, including pregnant women and those receiving highly active antiretroviral therapies (HAART), should receive chemoprophylaxis against Pneumocystis carinii pneumonia (PCP) if they have a CD4+ T-lymphocyte count of less than 200 per mm3 (200 ×106 per L) (AI) or a history of oropharyngeal candidiasis (AII).4 Persons who have a CD4+ T-lymphocyte percentage of less than 14 percent or a history of an acquired immunodeficiency syndrome (AIDS)–defining illness but do not otherwise qualify should be considered for prophylaxis (BII).5,6 When monitoring the CD4+ T-lymphocyte count at least every three months is not possible, initiation of chemoprophylaxis at a CD4+ T-lymphocyte count of greater than 200 but less than 250 per mm3 also should be considered (BII).5
Trimethoprim-sulfamethoxazole (TMP-SMZ) is the recommended prophylactic agent (AI).6–8 One double-strength tablet per day is the preferred regimen (AI).7 However, one single-strength tablet per day9 is also effective and might be better tolerated (AI). One double-strength tablet three times per week is also effective (BI).10 TMP-SMZ at a dosage of one double-strength tablet per day confers cross-protection against toxoplasmosis11 and some common respiratory bacterial infections.7,12 Lower dosages of TMP-SMZ also might confer such protection. In patients who have an adverse reaction that is not life-threatening, treatment with TMP-SMZ should be continued if clinically feasible; in those who have discontinued such therapy because of an adverse reaction, reinstitution of TMP-SMZ should be strongly considered after the adverse event has resolved (AII). Patients who have experienced adverse events, especially fever and rash, might better tolerate reintroduction of the drug with a gradual increase in dosage (desensitization) as per published regimens (BI)13,14 or reintroduction of TMP-SMZ at a reduced dosage or frequency (CIII); up to 70 percent of patients can tolerate such reinstitution of therapy.12
If TMP-SMZ cannot be tolerated, prophylactic regimens that can be recommended as alternatives include dapsone (BI),7 dapsone plus pyrimethamine plus leucovorin (BI),15,16 aerosolized pentamidine administered by the Respirgard II nebulizer (Marquest, Englewood, Colo.) (BI),8 and atovaquone (BI).17,18 Atovaquone appears to be as effective as aerosolized pentamidine18 or dapsone (BI)17 but is substantially more expensive than the other regimens. In patients seropositive for Toxoplasma gondii who cannot tolerate TMP-SMZ, recommended alternatives to TMP-SMZ for prophylaxis against PCP and toxoplasmosis include dapsone plus pyrimethamine (BI)15,16 or atovaquone with or without pyrimethamine (CIII). The following regimens generally cannot be recommended as alternatives because data regarding their efficacy for PCP prophylaxis are insufficient for a firm recommendation: aerosolized pentamidine administered by other nebulization devices, intermittently administered parenteral pentamidine, oral pyrimethamine plus sulfadoxine, oral clindamycin plus primaquine, and intravenous trimetrexate. However, clinicians may consider using these agents in unusual situations in which the recommended agents cannot be administered (CIII).
Discontinuation of Primary Prophylaxis. Initial reports from three prospective observational studies,19–21 one retrospective review22 and one randomized trial23 suggest that PCP prophylaxis can be safely discontinued in patients responding to HAART with a sustained increase in CD4+ T-lymphocyte counts from less than 200 per mm3 to greater than 200 per mm3. Such reports have mostly included patients receiving primary prophylaxis (no prior episode of PCP) and protease inhibitor-containing regimens. In these studies, median follow-up ranged from six to 12 months, and the median CD4+ T-lymphocyte count at the time prophylaxis was discontinued was greater than 300 per mm3 (300 × 106 per L). At the time PCP prophylaxis was discontinued, many patients had sustained suppression of HIV plasma RNA levels below detection limits of the available assays. Although optimal criteria for discontinuing PCP prophylaxis are still being assessed, providers may wish to discontinue prophylaxis when patients have sustained a CD4+ T-lymphocyte count of greater than 200 per mm3 for at least three to six months (CII). Additional criteria might include sustained reduction in viral load for at least three to six months (CIII).
Restarting Primary Prophylaxis. No data are available to guide recommendations for reinstituting primary prophylaxis. Pending the availability of such data, a reasonable approach would be to use the criteria for initiating prophylaxis.
Children. Children born to HIV-infected mothers should be administered prophylaxis with TMP-SMZ beginning at four to six weeks of age24 (AII). Prophylaxis should be discontinued in children who are subsequently found not to be infected with HIV. HIV-infected children and children whose infection status remains unknown should continue to receive prophylaxis for the first year of life. The need for subsequent prophylaxis should be determined on the basis of age-specific CD4+ T-lymphocyte count thresholds (Table 10) (AII). The safety of discontinuing prophylaxis in HIV-infected children receiving HAART has not been studied.
Pregnant Women. Chemoprophylaxis for PCP should be administered to pregnant women as in other adults and adolescents (AIII). TMP-SMZ is the recommended prophylactic agent; dapsone is an alternative. Because of theoretic concerns regarding possible teratogenicity associated with drug exposures during the first trimester, providers may choose to withhold prophylaxis during the first trimester. In such cases, therapy with aerosolized pentamidine may be considered because of its lack of systemic absorption and the resultant lack of exposure of the developing embryo to the drug (CIII).
TOXOPLASMIC ENCEPHALITIS
Initiation of Primary Prophylaxis. Toxoplasma-seropositive patients who have a CD4+ T-lymphocyte count of less than 100 per mm3 (100 ×106 per L) should be administered prophylaxis against toxoplasmic encephalitis (AII).11 The daily double-strength tablet of TMP-SMZ recommended as the preferred regimen for PCP prophylaxis appears to be effective against toxoplasmic encephalitis as well and is therefore recommended (AII).11 If patients cannot tolerate TMP-SMZ, the recommended alternative is dapsone-pyrimethamine, which is also effective against PCP (BI).15,16 Atovaquone, with or without pyrimethamine, also may be considered (CIII). Prophylactic monotherapy with dapsone, pyrimethamine, azithromycin or clarithromycin cannot be recommended on the basis of current data (DII). Aerosolized pentamidine does not protect against toxoplasmic encephalitis and is not recommended (EI).7,11
Toxoplasma-seronegative persons who are not taking a PCP prophylactic regimen known to be active against toxoplasmic encephalitis should be retested for IgG antibody to Toxoplasma when their CD4+ T-lymphocyte count declines below 100 per mm3 to determine whether they have seroconverted and are therefore at risk for toxoplasmic encephalitis (CIII). Patients who have seroconverted should be administered prophylaxis for toxoplasmic encephalitis as described above (AII).
Discontinuation of Primary Prophylaxis. Limited data suggest that discontinuing prophylaxis in patients whose CD4+ T-lymphocyte counts increase to greater than 100 per mm3 in response to HAART is associated with a low risk for toxoplasmic encephalitis infection. However, the numbers of patients who have been evaluated are insufficient to recommend routine discontinuation of prophylaxis in such patients. Persons whose CD4+ T-lymphocyte count remains less than 200 per mm3 or who have a history of PCP or oropharyngeal candidiasis still require prophylaxis against PCP, as noted previously.
Children. TMP-SMZ, when administered for PCP prophylaxis, also provides prophylaxis against toxoplasmosis. Atovaquone might also provide protection (CIII). Children more than 12 months of age who qualify for PCP prophylaxis and are receiving an agent other than TMP-SMZ or atovaquone should have serologic testing for Toxoplasma antibody (BIII), because alternative drugs for PCP prophylaxis might not be effective against Toxoplasma. Severely immunosuppressed children who are not receiving TMP-SMZ or atovaquone and who are found to be seropositive for Toxoplasma should be administered prophylaxis for PCP and toxoplasmosis (i.e., dapsone plus pyrimethamine) (BIII).
Pregnant Women. TMP-SMZ can be administered for prophylaxis against toxoplasmic encephalitis as described for PCP (AIII). Because of the low incidence of toxoplasmic encephalitis during pregnancy and the possible risk associated with pyrimethamine treatment, chemoprophylaxis with pyrimethamine-containing regimens can reasonably be deferred until after pregnancy (CIII). For prophylaxis against recurrent toxoplasmic encephalitis, the health care provider and clinician should be well informed about the benefit of lifelong therapy and the concerns about teratogenicity of pyrimethamine. Most clinicians favor lifelong therapy for the mother, given the high likelihood that disease will recur promptly if therapy is stopped (AIII).
In rare cases, HIV-infected pregnant women who have serologic evidence of remote toxoplasmic infection have transmitted Toxoplasma to the fetus in utero. Pregnant HIV-infected women who have evidence of primary toxoplasmic infection or active toxoplasmosis (including toxoplasmic encephalitis) should be evaluated and managed during pregnancy in consultation with appropriate specialists (BIII). Infants born to women who have serologic evidence of infections with HIV and Toxoplasma should be evaluated for congenital toxoplasmosis (BIII).
CRYPTOSPORIDIOSIS
No agents have been proved to be effective as chemoprophylaxis against cryptosporidiosis. Rifabutin or clarithromycin, when taken for Mycobacterium avium complex prophylaxis, were associated with a reduced risk for cryptosporidiosis in one study,25 but data are insufficient to warrant a recommendation for using these drugs.
Children. At present, no data indicate that formula-preparation practices for infants should be altered in an effort to prevent cryptosporidiosis (CIII). However, in the event of a “boil-water” advisory, similar precautions for the preparation of infant formula should be taken as for drinking water for adults (AII).
MICROSPORIDIOSIS
No chemoprophylactic regimens are known to be effective in preventing microsporidiosis.
TUBERCULOSIS
When HIV infection is first recognized, the patient should receive a tuberculin skin test (TST) by administration of intermediate-strength (5-TU) purified protein derivative (PPD) by the Mantoux method (AI). Routine evaluation for anergy is not recommended. However, there are selected situations in which anergy evaluation might assist in guiding individual decisions about preventive therapy.26
All HIV-infected persons who have a positive TST result (5 mm or more of induration) should undergo chest radiography and clinical evaluation to rule out active tuberculosis. HIV-infected persons who have symptoms suggestive of tuberculosis should promptly undergo chest radiography and clinical evaluation regardless of their TST status (AII).
All HIV-infected persons, regardless of age, who have a positive TST result but no evidence of active tuberculosis and no history of treatment or prophylaxis for tuberculosis should be administered preventive chemotherapy. Options include isoniazid daily (AII) or twice weekly (BI) for nine months or two months of therapy with rifampin and pyrazinamide (AI) or rifabutin and pyrazinamide (BIII).26 Because HIV-infected persons are at risk for peripheral neuropathy, those receiving isoniazid should also receive pyridoxine (BIII). A decision to use a regimen containing rifampin or rifabutin should be made after careful consideration of potential drug interactions, especially those related to protease inhibitors and nonnucleoside reverse transcriptase inhibitors (see Drug Interactions below). Directly observed therapy should be used with intermittent dosing regimens (AI) and when otherwise operationally feasible (BIII).26
HIV-infected persons who are close contacts of persons who have infectious tuberculosis should be administered preventive therapy—regardless of their TST results, age or prior courses of chemoprophylaxis—after the diagnosis of active tuberculosis has been excluded (AII).26 In addition to household contacts, such persons might also include contacts in the same drug treatment or health care facility, coworkers and other contacts if transmission of tuberculosis is demonstrated.
For persons exposed to isoniazid- or rifampin-resistant tuberculosis, the decision to use chemoprophylactic antimycobacterial agents other than isoniazid alone, rifampin plus pyrazinamide, or rifabutin plus pyrazinamide should be based on the relative risk for exposure to resistant organisms and should be made in consultation with public health authorities (AII).
TST-negative, HIV-infected persons from risk groups or geographic areas with a high prevalence of Mycobacterium tuberculosis infection might be at increased risk for primary or reactivation tuberculosis. However, the efficacy of preventive therapy in this group has not been demonstrated. Decisions concerning the use of chemoprophylaxis in these situations must be considered individually.
Although the reliability of the TST might diminish as the CD4+ T-lymphocyte count declines, annual repeat testing should be considered for HIV-infected persons who are TST-negative on initial evaluation and who belong to populations in which there is a substantial risk for exposure to M. tuberculosis (BIII). Clinicians also may consider repeating TSTs for persons whose immune function has improved because of HAART (i.e., those whose CD4+ T-lymphocyte count has increased to greater than 200 per mm3) (CIII). In addition to confirming tuberculous infection, TST conversion in an HIV-infected person should alert health care providers to the possibility of recent M. tuberculosis transmission and should prompt notification of public health officials for investigation to identify a possible source case.
The administration of bacille Calmette-Guérin (BCG) vaccine to HIV-infected persons is contraindicated because of its potential to cause disseminated disease (EII).
Drug Interactions. Rifampin should not be administered with protease inhibitors or nonnucleoside reverse transcriptase inhibitors (EI).26 Rifabutin is an acceptable alternative but should not be used with the protease inhibitor hard-gel saquinavir; caution is also advised if the drug is coadministered with soft-gel saquinavir, but data are lacking. Rifabutin can be administered at one half the usual daily dosage (i.e., reduce from 300 mg to 150 mg per day) with indinavir, nelfinavir or amprenavir, or with one fourth the usual dosage (i.e., 150 mg every other day or three times a week) with ritonavir. Similarly, rifabutin should not be used with the nonnucleoside reverse transcriptase inhibitor delavirdine. Pharmacokinetic data suggest that rifabutin at an increased dosage can be administered with efavirenz; a dosage of 450 mg per day has been suggested.26 Information is lacking regarding coadministration of rifabutin with nevirapine.
Children. Infants born to HIV-infected mothers should have a TST (5-TU PPD) at or before nine to 12 months of age and should be retested at least once a year (AIII). HIV-infected children living in households with TST-positive persons should be evaluated for tuberculosis (AIII); children exposed to a person who has active tuberculosis should be administered preventive therapy after active tuberculosis has been excluded, regardless of their TST results (AII).
Pregnant Women. Chemoprophylaxis for tuberculosis is recommended during pregnancy for HIV-infected patients who have a positive TST or a history of exposure to active tuberculosis, after active tuberculosis has been excluded (AIII). A chest radiograph should be obtained before treatment and appropriate abdominal/pelvic lead apron shields should be used to minimize radiation exposure to the embryo/fetus. When an HIV-infected person has not been exposed to drug-resistant tuberculosis, isoniazid daily or twice weekly is the prophylactic regimen of choice. Because of concerns regarding possible teratogenicity associated with drug exposures during the first trimester, providers may choose to initiate prophylaxis after the first trimester. Preventive therapy with isoniazid should be accompanied by pyridoxine to reduce the risk for neurotoxicity. Experience with rifampin or rifabutin during pregnancy is more limited, but anecdotal experience with rifampin has not been associated with adverse pregnancy outcomes. Pyrazinamide should generally be avoided, particularly in the first trimester, because of lack of information concerning fetal effects.
DISSEMINATED INFECTION WITH MYCOBACTERIUM AVIUM COMPLEX
Initiation of Primary Prophylaxis. Adults and adolescents who have HIV infection should receive chemoprophylaxis against disseminated Mycobacterium avium-complex (MAC) disease if they have a CD4+ T-lymphocyte count of less than 50 per mm3 (50 × 106 per L) (AI).27 Preferred prophylaxis is clarithromycin28,29 or azithromycin30 (AI). The combination of clarithromycin and rifabutin is no more effective than clarithromycin alone for chemopro-phylaxis and is associated with a higher rate of adverse effects than either drug alone; this combination should not be used (EI).28 The combination of azithromycin with rifabutin is more effective than azithromycin alone; however, the additional cost, increased occurrence of adverse effects, potential for drug interactions and absence of a difference in survival when compared with azithromycin alone do not warrant a routine recommendation for this regimen (CI).30 In addition to their preventive activity for MAC disease, clarithromycin and azithromycin each confer protection against respiratory bacterial infections (BII). If clarithromycin or azithromycin cannot be tolerated, rifabutin is an alternative prophylactic agent for MAC disease (BI)28,30,31 Tolerance, cost and drug interactions are among the issues that should be considered in decisions regarding the choice of prophylactic agents for MAC disease. Particular attention to interactions with antiretroviral protease inhibitors and nonnucleoside reverse transcriptase inhibitors is warranted (see Drug Interactions, below). Before prophylaxis is initiated, disseminated MAC disease should be ruled out by clinical assessment, which might include obtaining a blood culture for MAC if warranted. Because treatment with rifabutin could result in the development of resistance to rifampin in persons who have active tuberculosis, active tuberculosis should also be excluded before rifabutin is used for prophylaxis.
Although the detection of MAC organisms in the respiratory or gastrointestinal tract might predict the development of disseminated MAC infection, no data are available on the efficacy of prophylaxis with clarithromycin, azithromycin, rifabutin or other drugs in patients with MAC organisms at these sites and a negative blood culture. Therefore, routine screening of respiratory or gastrointestinal specimens for MAC cannot be recommended (DIII).
Discontinuation of Primary Prophylaxis. Information from observational studies22,32 suggested a low rate of disseminated infection with MAC among persons who responded to HAART with an increase in CD4+ T-lymphocyte count from less than 50 per mm3 to greater than 100 per mm3. Although the optimal criteria for discontinuing MAC prophylaxis remain to be defined, a reasonable option would be to consider discontinuing prophylaxis in patients with a CD4+ T-lymphocyte count of greater than 100 per mm3 for a sustained period (e.g., greater than three to six months) and sustained suppression of HIV plasma RNA for a similar period (CII).
Restarting Primary Prophylaxis. No data are available on which to base recommendations for reinstituting prophylaxis. Pending the availability of such data, a reasonable approach would be to use the criteria for initiating MAC prophylaxis (CIII).
Drug Interactions. Rifabutin should not be administered with certain protease inhibitors or nonnucleoside reverse transcriptase inhibitors (see Drug Interactions in Tuberculosis section). Although protease inhibitors might also increase clarithromycin levels, no recommendation to adjust the dosage of clarithromycin or protease inhibitors can be made on the basis of existing data.
Children. HIV-infected children less than 13 years of age who have advanced immunosuppression also can develop disseminated MAC infections, and prophylaxis should be offered to high-risk children according to the following CD4+ T-lymphocyte thresholds:
- Children 6 years of age or more, less than 50 per mm3
- Children two to six years of age, less than 75 per mm3
- Children one to two years of age, less than 500 per mm3
- Children less than 12 months of age, less than 750 per mm3 (750 × 106 per L) (AII).
For the same reasons that clarithromycin and azithromycin are the preferred prophylactic agents in adults, they should also be considered for children (AII); oral suspensions of both agents are commercially available in the United States. No liquid formulation of rifabutin suitable for pediatric use is commercially available in the United States. The safety of discontinuing MAC prophylaxis in children whose CD4+ T-lymphocyte counts have increased in response to HAART has not been studied.
Pregnant Women. Chemoprophylaxis for MAC disease should be administered to pregnant women as in other adults and adolescents (AIII). However, because of general concerns about administering drugs during the first trimester of pregnancy, some providers may choose to withhold prophylaxis during the first trimester. Animal studies and anecdotal evidence of safety in humans suggest that of the available agents, azithromycin is the drug of choice (BIII).33 Experience with rifabutin is limited. Clarithromycin has been demonstrated to be a teratogen in animals and should be used with caution during pregnancy.34 For secondary prophylaxis (chronic maintenance therapy), azithromycin plus ethambutol are the preferred drugs (BIII).
BACTERIAL RESPIRATORY INFECTIONS
As soon as feasible after HIV infection is diagnosed, adults and adolescents who have a CD4+ T-lymphocyte count of 200 per mm3 or more should be administered a single dose of 23-valent polysaccharide pneumococcal vaccine if they have not had this vaccine during the previous five years (BII).35,36 For persons who have a CD4+ T-lymphocyte count of less than 200 per mm3, vaccination can be offered, although the humoral response and clinical efficacy are likely to be diminished (CIII).
The recommendation to vaccinate is increasingly pertinent because of the increasing incidence of invasive infections with drug-resistant (including TMP-SMZ-, macrolide-, penicillin- and beta-lactam–resistant) strains of Staphylococcus pneumoniae. Limited data suggest that administration of certain bacterial vaccines might transiently increase HIV replication and plasma HIV-1 RNA levels in a minority of HIV-infected persons. However, there is no evidence that adverse clinical outcomes are associated with this transient increase. Most experts believe that the benefit of pneumococcal vaccination outweighs the potential risk.
The duration of the protective effect of primary pneumococcal vaccination is unknown. Periodic revaccination may be considered; an interval of five years has been recommended for persons not infected with HIV and also might be appropriate for persons infected with HIV.37 In addition, revaccination one time should also be considered if the initial vaccination was given when the CD4+ T-lymphocyte count was less than 200 per mm3 and if the CD4+ T-lymphocyte count has increased to greater than 200 per mm3 as a result of HAART (CIII).
The incidence of Haemophilus influenzae type b infection in adults is low. Therefore, H. influenzae type b vaccine is not generally recommended for adult use (DIII). TMP-SMZ, when administered daily for PCP prophylaxis, reduces the frequency of bacterial respiratory infections; this should be considered in the selection of an agent for PCP prophylaxis (AII). However, indiscriminate use of this drug (when not indicated for PCP prophylaxis or other specific reasons) might promote the development of TMP-SMZ–resistant organisms. Thus, TMP-SMZ should not be prescribed solely to prevent bacterial respiratory infection (DIII). Similarly, clarithromycin administered daily and azithromycin administered weekly for MAC prophylaxis might be effective in preventing bacterial respiratory infections; this possibility should be considered in the selection of an agent for prophylaxis against MAC disease (BII). However, these drugs should not be prescribed solely for preventing bacterial respiratory infection (DIII).
An absolute neutrophil count that is depressed because of HIV disease or drug therapy is associated with an increased risk for bacterial infections, including pneumonia. To reduce the risk for such bacterial infections, providers may consider taking steps to reverse neutropenia, by stopping myelosuppressive drugs (CII) or by administering granulocyte–colony-stimulating factor (G–CSF) (CII).
Children. Children who have HIV infection should be administered H. influenzae type b vaccine in accordance with the guidelines of the Advisory Committee on Immunization Practices38 and the American Academy of Pediatrics39 (AII). Children more than two years of age also should be administered 23-valent polysaccharide pneumococcal vaccine (BII). Revaccination with pneumococcal vaccine generally should be offered after three to five years to children less than 10 years of age and after five years to children 10 years of age or older (BIII).
To prevent serious bacterial infections in HIV-infected children who have hypogammaglobulinemia (IgG less than 400 mg per dL), clinicians should use intravenous immunoglobulin (IVIG) (AI). Respiratory syncytial virus (RSV) IVIG (in a dosage of 750 mg per kg), not monoclonal RSV antibody, may be substituted for IVIG during the RSV season to provide broad anti-infective protection, if RSV IVIG is available.
Pregnant Women. Pneumococcal vaccination is recommended during pregnancy for HIV-infected patients who have not been vaccinated during the previous five years (BIII). Among nonpregnant adults, vaccination has been associated with a transient burst of HIV replication. Whether the transient viremia can increase the risk for perinatal HIV transmission is unknown. Because of this concern, vaccination may be deferred when feasible, until after antiretroviral therapy has been initiated to prevent perinatal HIV transmission (CIII).
BACTERIAL ENTERIC INFECTIONS
Prophylactic antimicrobial agents are not generally recommended for travelers (DIII). The effectiveness of these agents depends on local antimicrobial-resistance patterns of gastrointestinal pathogens, which are seldom known. Moreover, these agents can elicit adverse reactions and promote the emergence of resistant organisms. However, for HIV-infected travelers, antimicrobial prophylaxis may be considered, depending on the level of immunosuppression and the region and duration of travel (CIII). The use of flu-oroquinolones such as ciprofloxacin (in a dosage of 500 mg per day) can be considered when prophylaxis is deemed necessary (BIII). As an alternative (e.g., in children, pregnant women and persons already taking TMP-SMZ for PCP prophylaxis), TMP-SMZ might offer some protection against traveler's diarrhea (BIII). The risk of toxicity should be considered before treatment with TMP-SMZ is initiated solely because of travel.
Antimicrobial agents such as fluoroquinolones should be given to patients before their departure, with directions to be taken empirically (e.g., 500 mg of ciprofloxacin twice a day for three to seven days) should traveler's diarrhea develop (BIII). Fluoroquinolones should be avoided in children less than 18 years of age and in pregnant women, and alternative antibiotics should be considered (BIII).
Travelers should consult a physician if the diarrhea is severe and does not respond to empiric therapy, if the stools contain blood, if fever is accompanied by shaking chills or if dehydration develops. Antiperistaltic agents (e.g., diphenoxylate and loperamide) can be used to treat mild diarrhea. However, the use of these drugs should be discontinued if symptoms persist beyond 48 hours. Moreover, these agents should not be administered to patients who have a high fever or who have blood in the stool (AII).
Some experts recommend that HIV-infected persons who have Salmonella gastroenteritis be administered antimicrobial therapy to prevent extraintestinal spread of the pathogen. However, no controlled study has demonstrated a beneficial effect of such treatment, and some studies of immunocompetent persons have suggested that antimicrobial therapy can lengthen the shedding period. The fluoroquinolones—primarily ciprofloxacin (in a dosage of 750 mg twice a day for 14 days)—can be used when antimicrobial therapy is chosen (CIII).
Children. Like HIV-infected adults, HIV-infected children should wash their hands after handling pets (especially before eating) and should avoid contact with pets' feces. Hand washing should be supervised (BIII).
HIV-exposed infants less than three months of age and all HIV-infected children who have severe immunosup-pression should be administered treatment for Salmonella gastroenteritis to prevent extraintestinal spread of the pathogen (CIII). Choices of antibiotics include TMP-SMZ, ampicillin, cefotaxime, ceftriaxone or chloramphenicol; fluoroquinolones should be used with caution and only if no alternatives exist.
HIV-infected children who have Salmonella septicemia should be offered long-term therapy to prevent recurrence (CIII). TMP-SMZ is the drug of choice; ampicillin or chloramphenicol can be used if the organism is susceptible. Fluoroquinolones should be used with caution and only if no alternative exists. Antiperistaltic drugs are not recommended for children (DIII).
Pregnant Women. Because pregnancy and HIV infection confer a risk for listeriosis, pregnant HIV-infected women should heed recommendations regarding listeriosis (BII).
Because extraintestinal spread of Salmonella during pregnancy might lead to infection of the placenta and amniotic fluid and result in pregnancy loss similar to that seen with Listeria monocytogenes infection, pregnant women with Salmonella gastroenteritis should receive treatment (BIII). Choices for treatment include ampicillin, cefotaxime, ceftriaxone or TMP-SMZ. Fluoroquinolones should be avoided.
Fluoroquinolones should not be used during pregnancy. TMP-SMZ might offer some protection against traveler's diarrhea.
INFECTION WITH BARTONELLA (FORMERLY ROCHALIMAEA)
No data support chemoprophylaxis for Bartonella-associated disease (CIII).
Children. The risks of cat ownership for HIV-infected children who are severely immunocompromised should be discussed with parents and caretakers (CIII).
Pregnant Women. If long-term suppression of Bartonella infection is required, erythromycin should be used. Tetracyclines should not be used during pregnancy.
CANDIDIASIS
Data from prospective controlled trials indicate that fluconazole can reduce the risk for mucosal (oropharyngeal, esophageal and vaginal) candidiasis and cryptococcosis as well in patients with advanced HIV disease.40–42 However, routine primary prophylaxis is not recommended because of the effectiveness of therapy for acute disease, the low mortality associated with mucosal candidiasis, the potential for resistant Candida organisms to develop, the possibility of drug interactions and the cost of prophylaxis (DIII).
Children. Primary prophylaxis of candidiasis in HIV-infected infants is not indicated (DIII).
Pregnant Women. Experience is limited with the use of systemic antifungal drugs during human pregnancy. Four cases of infants born with craniofacial and skeletal abnormalities following prolonged in utero exposure to fluconazole have been reported.34,43 In addition, itraconazole is embryotoxic and teratogenic in animal systems.44 These same potential risks of teratogenicity are presumed to apply to other systemically absorbed azole antifungals, such as ketoconazole. Therefore, chemoprophylaxis against oropharyngeal, esophageal or vaginal candidiasis using systemically absorbed azoles should not be initiated during pregnancy (DIII), and therapy with azoles should be discontinued in HIV-infected women who become pregnant (DIII). Effective birth-control measures should be recommended to all HIV-infected women on azole therapy for candidiasis (AIII).
CRYPTOCOCCOSIS
Routine testing of asymptomatic persons for serum cryptococcal antigen is not recommended because of the low probability that the results will affect clinical decisions (DIII).
Prospective controlled trials indicate that fluconazole and itraconazole can reduce the frequency of cryptococcal disease among patients who have advanced HIV disease. However, most experts recommend that antifungal prophylaxis not be used routinely to prevent cryptococcosis because of the relative infrequency of cryptococcal disease, the lack of survival benefits associated with prophylaxis, the possibility of drug interactions, the potential development of antifungal drug resistance and cost. The need for prophylaxis or suppressive therapy for other fungal infections (e.g., candidiasis, histoplasmosis or coccidioidomycosis) should be considered in making decisions about prophylaxis for cryptococcosis. If used, fluconazole at dosages of 100 to 200 mg daily is reasonable in patients whose CD4+ T-lymphocyte counts are less than 50 per mm3 (CI).40–42
Pregnant Women. Prophylaxis with fluconazole or itraconazole should not be initiated during pregnancy because of the low incidence of cryptococcal disease, the lack of a recommendation for primary prophylaxis against cryptococcosis in nonpregnant adults and potential teratogenic effects of these drugs during pregnancy (DIII).43–45 In patients who conceive while being administered primary prophylaxis and who elect to continue their pregnancy, prophylaxis should be discontinued.
HISTOPLASMOSIS
Routine skin testing with histoplasmin and serologic testing for antibody or antigen in histoplasmosis-endemic areas are not predictive of disease and should not be performed (DII).
Data from a prospective randomized, controlled trial indicate that itraconazole can reduce the frequency of histoplasmosis among patients who have advanced HIV infection and who live in areas where Histoplasma capsulatum is endemic.46 However, no survival benefit was observed among persons receiving itraconazole. Prophylaxis with itraconazole may be considered in patients with CD4+ T-lymphocyte counts less than 100 per mm3 who are at especially high risk because of occupational exposure or who live in a community with a hyperendemic rate of histoplasmosis (10 or more cases per 100 patient-years) (CI).
Children. Because primary histoplasmosis can lead to disseminated infection in children, a reasonable option is to administer lifelong suppressive therapy after an acute episode of the disease (AIII).
Pregnant Women. Because of the embryotoxicity and teratogenicity of itraconazole in animal systems, primary prophylaxis against histoplasmosis should not be offered during pregnancy (DIII).
COCCIDIOIDOMYCOSIS
Routine skin testing with coccidioidin (spherulin) in coccidioidomycosis-endemic areas is not predictive of disease and should not be performed (DII). Within the endemic area, a positive serologic test might indicate an increased risk for active infection; however, routine testing does not appear to be useful and should not be performed (DIII).
Primary prophylaxis for HIV-infected persons who live in coccidioidomycosis-endemic areas is not routinely recommended.
CYTOMEGALOVIRUS DISEASE
Prophylaxis with oral ganciclovir may be considered for HIV-infected adults and adolescents who are cytomegalovirus (CMV) seropositive and who have a CD4+ T-lymphocyte count of less than 50 per mm3 (CI).47,48 Ganciclovir-induced neutropenia, anemia, conflicting reports of efficacy, lack of proven survival benefit, the risk for developing ganciclovir-resistant CMV and cost are among the issues that should be considered when deciding to institute prophylaxis in individual patients. Acyclovir is not effective in preventing CMV disease, and valacyclovir is not recommended because of an unexplained trend toward increased deaths among persons with AIDS who were administered valacyclovir for CMV prophylaxis.49 Therefore, neither acyclovir nor valacyclovir should be used for this purpose (EI). The most important method of preventing severe CMV disease is recognition of the early manifestations of the disease. Early recognition of CMV retinitis is most likely when the patient has been educated on this topic. Patients should be made aware of the significance of increased floaters in the eye and should be advised to assess their visual acuity regularly by using simple techniques such as reading newsprint (BIII). Regular funduscopic examinations performed by an ophthalmologist are recommended by some experts for patients with low (e.g., less than 50 per mm3) CD4+ T-lymphocyte counts (CIII).
Children. Some experts recommend obtaining a CMV urine culture for all HIV-infected (or exposed) infants at birth or at an early postnatal visit to identify infants with congenital CMV infection (CIII). In addition, beginning at one year of age, CMV antibody testing on an annual basis may be considered for CMV-seronegative (and culture-negative) HIV-infected infants and children who are severely immunosuppressed (Table 10) (CIII). Annual testing will allow identification of children who have acquired CMV infection and might benefit from screening for retinitis.
HIV-infected children who are CMV-infected and severely immunosuppressed might benefit from a dilated retinal examination performed by an ophthalmologist every four to six months (CIII). In addition, older children should be counseled to be aware of floaters in the eye, as recommended for adults (BIII).
Oral ganciclovir results in reduced CMV shedding in CMV-infected children and may be considered for primary prophylaxis against CMV disease in CMV-infected children who are severely immunosuppressed (e.g., CD4+ T-lymphocyte count less than 50 per mm3) (CII).
Pregnant Women. Because of the lack of a recommendation for routine use of ganciclovir among nonpregnant adults and the lack of experience with this drug during pregnancy, ganciclovir is not recommended for primary prophylaxis against CMV disease during pregnancy (DIII). Ganciclovir therapy should be discontinued in patients who conceive while being administered primary prophylaxis.
HERPES SIMPLEX VIRUS DISEASE
Prophylaxis of initial episodes of HSV disease is not recommended (DIII).
Children. The recommendations for preventing initial disease and recurrence among adults and adolescents apply to children as well.
Pregnant Women. Oral acyclovir prophylaxis during late pregnancy is a controversial strategy recommended by some experts to prevent neonatal herpes transmission. However, such prophylaxis is not routinely recommended.
VARICELLA-ZOSTER VIRUS INFECTION
Minimal data regarding the safety and efficacy of varicella vaccine in HIV-infected adults are available, and no recommendation for its use can be made in this population. (See Children, below, for information about the use of varicella vaccine in children.)
For the prophylaxis of chickenpox, HIV-infected children and adults who are susceptible to varicella-zoster virus (VZV) (i.e., those who have no history of chickenpox or shingles or who have no detectable antibody against VZV) should be administered varicella zoster immune globulin (VZIG) as soon as possible but within 96 hours after close contact with a patient who has chickenpox or shingles (AIII). Data are lacking on the effectiveness of acyclovir in preventing chickenpox in susceptible HIV-infected children or adults. No preventive measures are currently available for shingles.
Children. HIV-infected children who are asymptomatic and not immunosuppressed (i.e., in immunologic category 1 [Table 10]) should receive live attenuated varicella vaccine at 12 to 15 months of age or later (BII). Varicella vaccine should not be administered to other HIV-infected children because of the potential for disseminated viral infection (EIII).
Pregnant Women. VZIG is recommended for VZV-susceptible, HIV-infected pregnant women within 96 hours after exposure to VZV (AIII). If oral acyclovir is used, VZV serology should be performed so the drug can be discontinued if the patient is seropositive for VZV (BIII).
HUMAN HERPESVIRUS 8 INFECTION
Because clinical use of routine serologic testing to identify human herpesvirus 8 (HHV-8) infection has not been established, no recommendation for serologic testing can be made at this time.
Lower rates of Kaposi's sarcoma have been observed among AIDS patients treated with ganciclovir or foscarnet for CMV retinitis.50 HHV-8 replication in vitro is inhibited by ganciclovir, foscarnet and cidofovir. However, because the efficacy and clinical use of these drugs in preventing Kaposi's sarcoma have not been established, no recommendation can be made concerning the use of these or other drugs to prevent Kaposi's sarcoma in persons coinfected with HIV and HHV-8.
Potent antiretroviral drug combinations that suppress HIV replication reduce the frequency of Kaposi's sarcoma in HIV-infected persons and should be considered for use in all persons who qualify for such therapy (BII).
Children. In parts of the world where HHV-8 is endemic, horizontal transmission might occur among young children, possibly through saliva. However, no recommendations are currently available for preventing HHV-8 transmission from child to child.
HUMAN PAPILLOMAVIRUS INFECTION
Human Papillomavirus-Associated Genital Epithelial Cancers in HIV-Infected Women. After a complete history of previous cervical disease has been obtained, HIV-infected women should have a pelvic examination and a Papanicolaou (Pap) smear. In accordance with the recommendation of the Agency for Health Care Policy and Research, the Pap smear should be obtained twice in the first year after diagnosis of HIV infection and, if the results are normal, annually thereafter (AII).
If the results of the Pap smear are abnormal, care should be provided according to the Interim Guidelines for Management of Abnormal Cervical Cytology published by a National Cancer Institute Consensus Panel and briefly summarized in the following recommendations.51
In patients whose Pap smears are interpreted as atypical squamous cells of undetermined significance (ASCUS), several management options are available; the choice depends in part on whether the interpretation of ASCUS is qualified by a statement indicating that a neoplastic process is suspected. Follow-up by Pap tests without colposcopy is acceptable, particularly when the diagnosis of ASCUS is not qualified further or the cytopathologist suspects a reactive process. In such situations, Pap tests should be repeated every four to six months for two years until three consecutive smears have been negative. If a second report of ASCUS occurs in the two-year follow-up period, the patient should be considered for colposcopic evaluation (BIII).
Women who have a diagnosis of unqualified ASCUS associated with severe inflammation should be evaluated for an infectious process. If specific infections are identified, re-evaluation should be performed after appropriate treatment, preferably after two to three months (BIII).
If the diagnosis of ASCUS is qualified by a statement indicating that a neoplastic process is suspected, the patient should be managed as if a low-grade squamous intraepithelial lesion (LSIL) were present (see a following recommendation) (BIII). If a patient who has a diagnosis of ASCUS is at high risk (i.e., previous positive Pap tests or poor adherence to follow-up), the option of colposcopy should be considered (BIII).
Several management options are available for patients who have LSIL. Follow-up with Pap tests every four to six months is used by many clinicians and is currently used in countries outside the United States as an established method of management. Patients managed in this way must be carefully selected and considered reliable for follow-up. If repeat smears show persistent abnormalities, colposcopy and directed biopsy are indicated (BIII). Colposcopy and directed biopsy of any abnormal area on the ectocervix constitute another appropriate option (BIII).
Women who have cytologic diagnosis of high-grade squamous intraepithelial lesions (HSILs) or squamous cell carcinoma should undergo colposcopy and directed biopsy (AII).
No data are available to suggest that these guidelines to prevent cervical disease should be modified in women receiving HAART.
HPV-Associated Anal Intraepithelial Neoplasia and Anal Cancer in HIV-Infected Men Who Have Sex With Men. Evidence from several studies shows that HPV-positive men who have sex with men are at increased risk for anal HSILs and might be at increased risk for anal cancer. In view of this evidence, coupled with a recent cost-effectiveness analysis projecting that screening and treatment for anal HSILs provide clinical benefits comparable to other measures to prevent opportunistic infections in HIV-infected persons,52 anal cytology screening of HIV-infected men who have sex with men might become a useful preventive measure in the near future. However, further studies of screening and treatment programs for anal HSILs need to be carried out before recommendations for routine anal cytology screening can be made.
Pregnant Women. Use of intravaginal 5-fluorouracil to prevent recurrent dysplasia is not recommended during pregnancy.
HEPATITIS C VIRUS INFECTION
HIV-infected patients should be screened for hepatitis C virus (HCV) infection by using enzyme immunoassays (EIAs) licensed for detection of antibody to HCV (anti-HCV) in blood (BIII). Positive anti-HCV results should be verified with additional testing (i.e., recombinant immunoblot assay [RIBA] or reverse transcriptase polymerase chain reaction for HCV RNA). The presence of HCV RNA in blood might also be assessed in HIV-infected persons with undetectable antibody but other evidence of chronic liver disease (e.g., unexplained elevated liver-specific enzymes) or when acute HCV infection is suspected (CIII).
Persons coinfected with HIV and HCV should be advised not to drink excessive amounts of alcohol (AII). Avoiding alcohol altogether might be prudent because it is unclear whether even occasional moderate alcohol use (e.g., less than 12 oz of beer or less than 10 g of alcohol per week) increases the incidence of cirrhosis among HCV-infected persons (CIII).
Patients with chronic hepatitis C should be vaccinated against hepatitis A because (1) the risk for fulminant hepatitis associated with hepatitis A appears to be increased in HCV-coinfected persons; (2) hepatitis A vaccine is safe for HIV-infected persons; and (3) although immunogenicity is reduced in patients with advanced HIV infection, more than two thirds of patients develop protective antibody responses (BIII). Prevaccination screening for antibody to hepatitis A virus is cost-effective and therefore recommended when a greater than 30 percent prevalence of hepatitis A virus antibody is expected in the population being screened (e.g., persons more than 40 years of age)53 (BIII).
HIV-HCV-coinfected patients have a higher incidence of chronic liver disease than patients infected with HIV alone54 and should be evaluated for chronic liver disease and for the possible need for treatment.55 However, limited data exist regarding the safety and efficacy of antiviral treatment of patients coinfected with HIV and HCV. Moreover, because the optimal means of treating coin-fected patients has not been established and many HIV-infected patients have conditions that complicate therapy (e.g., depression or illicit drug use), this care should occur in a clinical trial or be coordinated by providers with experience treating HIV and HCV infections (BIII).
In some studies, the incidence of antiretroviral-associated liver enzyme elevations has been increased in patients coinfected with HIV and HCV56; such increases might not require treatment modifications. Thus, although liver enzymes should be carefully monitored, HAART should not be routinely withheld from patients coinfected with HIV and HCV (DIII). However, coinfected patients initiating antiretroviral therapy might have an inflammatory reaction that mimics an exacerbation of underlying liver disease. In this situation, careful monitoring of liver function is required.
Children. Children born to women coinfected with HIV and HCV should be tested for HCV infection57 (BI). In children with perinatal HCV infection, maternal HCV antibody can persist for up to 18 months, and HCV RNA can be intermittently undetectable. Thus, testing should be performed at or after two years of age. If earlier diagnosis is needed, HCV RNA should be assessed in more than one infant blood specimen obtained after one month of age. The average rate of HCV infection among infants born to coinfected women is approximately 15 percent (range: 5 to 36 percent).57 Data are limited on the natural history and treatment of HCV infection in children.

