Bilious vomiting in newborns is an urgent condition that requires the immediate involvement of a team of pediatric surgeons and neonatologists for perioperative management. However, initial detection, evaluation and treatment are often performed by nurses, family physicians and general pediatricians. Bilious vomiting, with or without abdominal distention, is an initial sign of intestinal obstruction in newborns. A naso- or orogastric tube should be placed immediately to decompress the stomach. Physical examination should be followed by plain abdominal films. Dilated bowel loops and air-fluid levels suggest surgical obstruction. Contrast radiography may be required. Duodenal atresia, midgut malrotation and volvulus, jejunoileal atresia, meconium ileus and necrotizing enterocolitis are the most common causes of neonatal intestinal obstruction.
When a neonate develops bilious vomiting, one should suspect a surgical condition. After a focused physical examination, a nasogastric or orogastric catheter should be placed for gastric decompression to prevent further vomiting and aspiration. This should be done before any diagnostic or therapeutic maneuvers are performed. Establishment of an intravenous line should follow for administration of fluid, electrolytes and nutrition. When the patient is hemodynamically stabilized, appropriate imaging studies of the abdomen should be performed. These would include plain abdominal films and/or contrast studies. When dilated bowel loops and air-fluid levels are demonstrated, the diagnosis of a surgical abdomen is suggested, and urgent consultation with a pediatric surgeon is indicated, preferably in a pediatric surgical center. Gastric decompression, hydration and secured airway must be completed before initiating transport of the patient. Intestinal obstruction with bilious vomiting in neonates can be caused by duodenal atresia, malrotation and volvulus, jejunoileal atresia, meconium ileus, and necrotizing enterocolitis (Table 1).
TABLE 1 Causes of Intestinal Obstruction with Bilious Vomiting in Neonates
| Type of obstruction | Cause and incidence | Age of onset and presentations | Diagnostic procedure and findings | Preoperative management interval before surgery | Treatment | Postoperative management | Prognosis |
|---|---|---|---|---|---|---|---|
| Duodenal atresia | Embryogenic; occurs in 1 per 5,000 live births; 25% have Down syndrome | Few hours after birth; bilious vomiting, no distention | Abdominal film, “double-bubble” sign | Nasogastric suction, IV fluids; 24 to 48 hours | Diamond-shaped duodenoduodenostomy | No oral intake, nasogastric suction; feeding at 2 to 3 days after surgery | Good unless associated with serious anomalies |
| Malrotation with volvulus | Incomplete bowel rotation occurring during 7th to 12th weeks of gestation | At 3 to 7 days; bilious vomiting, rapid deterioration with volvulus | Upper GI spiral sign on ultrasound; abnormal location of the superior mesenteric vessels | Nasogastric suction, IV fluid; STAT surgery for symptomatic patients, within daysfor others | Ladd's procedure; may require a second laparotomy | No oral intake; nasogastric suction | Good without bowel resection, difficult with short-gut syndrome after bowel resection |
| Jejunoileal atresia | Mesenteric vascular accident during fetal life in 1 per 3,000 live births | Within 24 hours of birth; vomiting, abdominal distention | Air-fluid levels on abdominal film | Nasogastric suction, IV fluids; 12 to 24 hours | Resection(s) and anastomosis(es) | No oral intake, nasogastric suction; feeding at 2 to 4 days after surgery | Good unless excessive loss of bowel |
| Meconium ileus | Genetic, occurs in 15% of newborns with cystic fibrosis, and in 1 per 5,000 to 10,000 live births | Immediately after birth; abdominal distention, bilious vomiting | Abdominal film; distention, air-fluid levels, sweat test, “ground-glass” sign | Decompression | Enterostomy if complicated; Gastrografin enema plus IV fluids | Acetylcysteine (Mucomyst), pancreatic enzymes | Depends on the systemic problems |
| Necrotizing ileus | Cause unknown in 2.4 per 1,000 live births | 10 to 12 days after birth; distention, vomiting, bloody stools | Abdominal film; distention, pneumatosis, air in the aortal vein | Nasogastric suction, IV fluids, nutrition, antibiotics for 10 days. When perforated, immediate surgery | Resection of necrotic bowel and enterostomy | Same as preoperative management | 25% need surgery (65% survival rate) 75% can be treated medically (95% survival rate) |
IV = intravenous; GI = gastrointestinal.
Duodenal Atresia
Duodenal atresia is a congenital obstruction of the second portion of the duodenum. Its etiology is believed to be failure of recanalization of this bowel segment during the early gestational stage. The pregnancy is associated with polyhydramnios, as it is in many cases where the infant is subsequently discovered to have an intestinal obstruction. Duodenal atresia occurs in 1 per 5,000 to 10,000 live births, involving male infants more commonly than female infants. Down syndrome occurs in about one-quarter of these patients; congenital heart disease occurs in about 20 percent of them.
Duodenal atresia may result in either a membranous or interrupted-type lesion that is located at the level of the papilla of Vater. In 80 percent of these patients, the papilla of Vater opens into the proximal duodenum, accounting for the bilious nature of the vomiting. Abdominal plain film shows a characteristic “double-bubble” sign, demonstrating the bubbles in the stomach and the dilated proximal duodenum; this confirms the diagnosis (Figure 1). A nasogastric tube should be placed for continuous suction of gastric contents, and intravenous fluid administration should be started. Surgery is required but is not urgent. A 24- to 48-hour delay may be allowed before operation for transport, further evaluation and fluid resuscitation.
FIGURE 1.
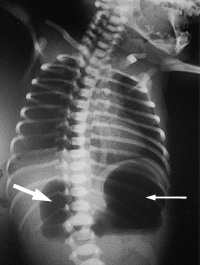
Upright abdominal film showing the characteristic “double-bubble” sign that confirms the diagnosis of duodenal atresia. Note the dilated stomach (thin arrow) and dilated proximal duodenum (thick arrow).
A diamond-shaped duodenoduodenostomy is the standard procedure for the interrupted-type lesion.1–5 The membranous-type lesion is treated simply by excision of the obstructing membrane. No gastrostomy or transanastomotic tube placement is required. The prognosis is excellent unless the patient has associated serious congenital anomalies.2–6
Midgut Malrotation and Volvulus
Malrotation of the midgut is an anatomic abnormality that allows the midgut to twist in a clockwise direction around the superior mesenteric vessels to obstruct and, perhaps, infarct the bulk of the small and large intestines. Normally, the third portion of the duodenum passes behind the superior mesenteric vessels along the lower margin of the pancreas. During embryonic life, the colon and small bowel grow rapidly and extrude from the abdominal cavity. During the seventh week of gestation, the midgut starts being reduced back into the abdominal cavity. The bowel then settles into the abdominal cavity and rotates in a counterclockwise direction (as viewed from the front), with the cecum coming to rest in the right lower quadrant of the abdomen. This process is completed by the 12th week of gestation.
For unknown reasons, however, in patients with midgut malrotation, the rotation ceases after the first 90 degrees, and the duodenum and ascending colon are juxtaposed around the superior mesenteric vessels, with the entire midgut suspended from this narrow axis. The malrotated bowel itself does not cause any significant problem. However, because of the narrow axis, the midgut can at any time twist around the axis, perhaps triggered by peristaltic action. The tighter the twist, the more the midgut suffers from obstruction of the lumen, obstruction of venous and lymphatic return from the midgut, and obstruction of arterial inflow, thus threatening midgut viability. Unless it is treated in a timely manner, bowel strangulation results in an ischemic loss of extensive bowel, causing the short-gut syndrome.
Most patients with midgut malrotation develop volvulus within the first week of life. Bilious vomiting is the initial symptom, but abdominal distention is not remarkable. The bowel can be involved in strangulation at any time and at any age. Once midgut ischemia occurs, unstable hemodynamics, intractable metabolic acidosis and necrosis with perforation develop, putting the patient at critical risk.
A malposition of the superior mesenteric vessels demonstrated by ultrasound examination is diagnostic.7,8 An upper gastrointestinal (GI) contrast study may show an obstruction at the second portion of the duodenum. The jejunum may show a spiral configuration (Figure 2). Lesser degrees of obstruction may show as a duodenojejunum that occupies the right hemi-abdomen. Urgent surgical treatment should be considered once the diagnosis is established. Midgut volvulus is associated with a mortality rate of 28 percent.9 Elective surgery is allowed in patients who were incidentally diagnosed by a contrast study performed for some other reason.10
FIGURE 2.
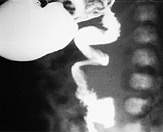
Upper gastrointestinal contrast study demonstrating a typical spiral configuration of jejunum in a patient with volvulus of the bowel.
At surgery, a fibrous band, called “Ladd's band,” is found to extend from the retroperitoneum to the malpositioned cecum, across the anterior aspect of the second portion of the duodenum. This band is divided to release the duodenal obstruction. The fused intestinal mesentery must be dissected to widen its axis and prevent recurrence of volvulus. During this dissection, the superior mesenteric vessels are clearly exposed and must not be injured.
When the operation is performed in a timely manner, the prognosis is excellent.9 If extensive bowel is ischemic because of the volvulus, the bowel is untwisted and simply reduced into the abdominal cavity, and the abdominal wound is closed. Twenty-four hours later, the abdomen is reentered for a “second look.” At this time, a demarcation is visible between necrotic and viable bowel, which allows the surgeon to resect the necrotic bowel and create an enterostomy at the distal end of the normal bowel. Bowel reconstruction is performed in a later operation.
This approach can salvage the maximal length of bowel and avoid development of the short-gut syndrome.11 Very occasionally, malrotation of the bowel can be identified in older patients who have had multiple episodes of abdominal pain of undetermined origin. The upper GI contrast study is helpful in making this diagnosis. Prophylactic surgical treatment should be recommended in this situation to avoid a potential catastrophic strangulation of bowel later.9
Jejunoileal Atresia
Jejunoileal atresia is caused by a mesenteric vascular accident during fetal life. Volvulus, intussusception or internal hernia may cause infarction of a segment of fetal intestine, as shown experimentally in animals.12 Jejunoileal atresia is classified anatomically into four types: (1) membranous, (2) interrupted (Figure 3), (3) apple-peel and (4) multiple.13 The symptoms and signs are identical regardless of the type of lesion. Abdominal distention with bilious vomiting is observed within the first 24 hours after birth. The more proximal the lesion, the earlier and more serious is the bile-stained vomiting.14 Abdominal films show air-fluid levels proximal to the lesion, confirming the diagnosis of bowel obstruction (Figure 4). In the past, barium enema was the classic diagnostic study for jejunoileal atresia, but it is not necessarily required today for diagnosis, because the results of the barium enema may appear normal in patients whose mesenteric vascular accident occurred shortly before birth, thereby confusing the diagnosis. In our experience, only 10 percent of patients required this study for diagnosis.15
FIGURE 3.
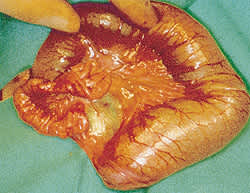
Interrupted-type bowel atresia. Continuity of the bowel is interrupted between the enlarged proximal bowel (right side of photo) and the atrophic distal bowel (left side of photo).
FIGURE 4.
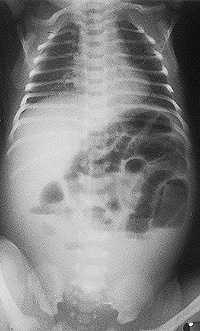
Upright abdominal film showing distention of the bowel with multiple air-fluid levels suggesting lower intestinal atresia.
Preoperatively, stomach decompression, intravenous hydration and correction of any electrolyte disturbance must be achieved. An interval of 12 to 24 hours is allowed for pre-operative preparation. At surgery, a different surgical strategy is indicated for each type of lesion. In general, bowel reconstruction is achieved by an end-to-end (or end-to-side) anastomosis (or anastomoses in multiple atresias).15,16 The prognosis is usually good unless excessive bowel resection is needed.
Meconium peritonitis is an aseptic peritonitis caused by spillage of meconium into the abdominal cavity during the development of jejunoileal atresia. Extravasation of meconium causes an intense chemical and foreign body reaction with characteristic calcifications, vascular fibrous proliferation and cyst formation. Once the diagnosis has been established, preparation is required for possible excessive blood loss during the operation. Meconium peritonitis occurs frequently in association with meconium ileus.17
Meconium Ileus
Meconium ileus is characterized by retention of thick tenacious meconium in the bowel (ileum, jejunum or colon), which results in bowel obstruction. In one half of patients with meconium ileus, the bowel is intact, and its continuity is preserved.18 The other patients have associated volvulus, jejunoileal atresia, bowel perforation and/or meconium peritonitis. Meconium ileus occurs in 15 percent of newborns with cystic fibrosis, and only 5 to 10 percent of patients with meconium ileus do not have cystic fibrosis.19,20
The involved bowel is distended by meconium retention during fetal life. After a few hours of postnatal life, bowel distention becomes remarkable because of swallowed air and causes bilious vomiting. In some cases, meconium retention may cause dystociaproducing abdominal distention. On physical examination, the thickened bowel loops are often visible and palpable through the abdominal wall. Remarkable abdominal distention, tenderness and/or erythema of the abdominal skin indicates perforation, which requires immediate surgery.
Rectal examination is often difficult, the small caliber of the rectum not allowing the examiner's finger to enter. Mucus plugs may be evacuated after withdrawal of the examination finger. The plain abdominal films show distended loops of intestine with thickened bowel walls. A large amount of meconium mixed with swallowed air produces the so-called “ground-glass” sign, which is typical of meconium ileus. Calcification, free air or very large air-fluid levels suggest bowel perforation, which requires urgent surgery. A contrast enema demonstrates a microcolon (Figure 5). Reflux of contrast into the ileum demonstrates the plugs, which are located in the distal small intestine. The small bowel is of narrow caliber distal to the meconium plug and dilated proximal to the meconium plugs.
FIGURE 5.
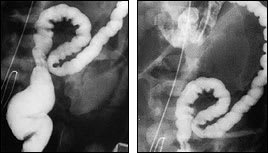
Meconium ileus. Contrast enema demonstrates a microcolon.
Patients with uncomplicated meconium ileus may be successfully treated with a diatrizoate maglumine (Gastrografin) enema performed while adequate intravenous fluid is being administered. The hypertonicity of the radiopaque agent (1,900 mOsm per L) draws fluid into the bowel to facilitate passage and expulsion of the tenacious meconium.21 This treatment is successful in 16 to 50 percent of patients.
When a Gastrografin enema is unsuccessful, laparotomy is indicated to evacuate the obstructing meconium by enterotomy irrigation. Immediate surgery is indicated in patients with complicated meconium ileus. Bowel resection for perforation and/or obstruction related to kinking of the bowel is indicated, usually requiring a temporary enterostomy.
Necrotizing Enterocolitis
Necrotizing enterocolitis is a rapidly progressing catastrophic disease producing extensive bowel necrosis, infection and perforation in newborns. The etiology is unknown, although several likely causes have been reported.22 Prematurity and pulmonary disorders are common predisposing factors.23,24 The usual onset is 10 to 12 days of age, with presenting symptoms of gastric retention, bilious vomiting, ileus, abdominal distention and bloody stools. Bradycardia, hypothermia, apneic spells and hypotension are later signs of progressive deterioration. Abnormal hemorrhage, hyperbilirubinemia and oliguria are late findings.
Physical findings include erythema and edema of the abdominal wall, absence of bowel sounds, abdominal distention, visible and/or palpable loops of the bowel, guarding and lethargy. It should be kept in mind that physical findings do not always or accurately reflect an intestinal catastrophe, especially in a weak premature infant.
Abdominal plain films taken at an interval of six to eight hours initially and daily in the following seven to 10 days are helpful in diagnosis and evaluation of the clinical progress. Distended loops with thickened bowel wall, pneumatosis cystoides intestinalis, air in the portal vein and/or free air are the radiographic findings (Figure 6). Daily change in bowel gas pattern is a good prognostic sign because it excludes ileus and bowel necrosis. Once the diagnosis is made, gastric decompression and intravenous administration of fluid and antibiotics should be started. Complete blood cell count, arterial pH and blood gas determination, and electrolyte assays are mandatory. Surgery is indicated when free peritoneal gas or clinical deterioration develop.24–28 Intractable metabolic acidosis and thrombocytopenia are also critical signs of bowel ischemia, indicating surgical treatment.29
FIGURE 6.
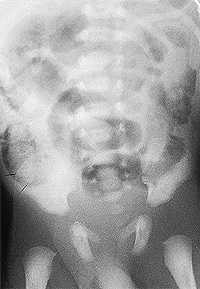
Abdominal film showing pneumatosis cystoides intestinalis in the right lower quadrant in a neonate with necrotizing enterocolitis.
At our institution, 422 patients with necrotizing enterocolitis (studied between 1978 and 1993) were treated with a uniform protocol of gastric decompression and intravenous administration of fluid, nutrition and antibiotics for 10 days. Seventy-five percent of patients were managed medically, and 95 percent survived. The remaining 25 percent of patients underwent surgery to divert the fecal stream by creation of an enterostomy, with a survival rate of 65 percent (authors' unpublished data).

