Up to 40 percent of postmenopausal women have symptoms of atrophic vaginitis. Because the condition is attributable to estrogen deficiency, it may occur in pre-menopausal women who take antiestrogenic medications or who have medical or surgical conditions that result in decreased levels of estrogen. The thinned endometrium and increased vaginal pH level induced by estrogen deficiency predispose the vagina and urinary tract to infection and mechanical weakness. The earliest symptoms are decreased vaginal lubrication, followed by other vaginal and urinary symptoms that may be exacerbated by superimposed infection. Once other causes of symptoms have been eliminated, treatment usually depends on estrogen replacement. Estrogen replacement therapy may be provided systemically or locally, but the dosage and delivery method must be individualized. Vaginal moisturizers and lubricants, and participation in coitus may also be beneficial in the treatment of women with atrophic vaginitis.
Because of declining estrogen levels, women who are in mid-life or beyond often present with symptoms of atrophic vaginitis. An estimated 10 to 40 percent of postmenopausal women have symptoms of atrophic vaginitis, also referred to as urogenital atrophy.1 Despite the prevalence of symptoms, only 20 to 25 percent of symptomatic women seek medical attention.2,3 Therefore, physicians have an opportunity to improve the urogenital health and quality of life of a large patient population through identification of and intervention in this often overlooked and underdiagnosed condition.
Throughout a woman's life cycle, the vaginal epithelium undergoes changes in response to the level of circulating estrogen. Stimulated by maternal estrogen, the vaginal epithelium is rugated and rich in glycogen in the newborn. During childhood, the epithelium remains thin until puberty, when it again thickens as a result of estrogen stimulation. Estrogen stimulation produces copious amounts of glycogen. Döderlein's lactobacilli depend on glycogen from sloughed vaginal cells.3 Lactic acid produced by these bacteria lowers vaginal pH levels to 3.5 to 4.5; this is essential for the body's natural defense against vaginal and urinary tract infections.4 Increased vaginal pH levels predispose the vagina to infection by streptococci, staphylococci, coliforms and diphtheroid.3 After menopause, circulating estrogen levels (mainly estradiol), are dramatically reduced from greater than 120 pg per mL to around 18 pg per mL.3 Numerous cytologic transformations follow estrogen reduction, including proliferation of connective tissue, fragmentation of elastin and hyalinization of collagen. These changes may result in granulation, fissures, ecchymoses, telangiectases and ulcerations.5 Postmenopausal changes in tissue composition are not limited to the genital tract but also include the urinary tract because of the shared common embryologic origin. Vaginal and urethral epithelia are estrogen dependent and adversely change in an estrogen-deprived environment.
Predisposing Factors
Menopause is the leading cause of decreased levels of circulating estrogen; therefore, it is the etiology in almost all cases of atrophic vaginitis. In nonmenopausal women, production of ovarian estrogen can be interrupted by radiation therapy, chemotherapy, immunologic disorders and oophorectomy. The postpartum decline in estrogen levels accompanies the loss of placental estrogen and the antagonistic action of prolactin on estrogen production during lactation. Side effects of antiestrogen medications, including medroxyprogesterone (Provera), tamoxifen (Nolvadex), danazol (Danocrine), leuprolide (Lupron) and nafarelin (Synarel), are also implicated as causes of atrophic vaginitis.6 An increase in the severity of symptoms occurs in women who are naturally premenopausally estrogen deficient, smoke cigarettes, have not given birth vaginally or exhibit nonfluctuating levels of estrogen.3,7,8 Milder atrophy occurs in postmenopausal women who participate in coitus, have higher androgen levels and have not undergone vaginal surgery (Table 1).3,6–9
TABLE 1 Factors That Increase the Risk of Developing Atrophic Vaginitis
| Menopause | |
| Decreased ovarian functioning | |
| Radiation therapy | |
| Chemotherapy | |
| Immune disorder | |
| Oophorectomy | |
| Postpartum loss of placental estrogen | |
| Elevated prolactin level during lactation | |
| Medications containing antiestrogen properties6 | |
| Tamoxifen (Nolvadex) | |
| Danazol (Danocrine) | |
| Medroxyprogesterone (Provera) | |
| Leuprolide (Lupron) | |
| Nafarelin (Synarel) | |
| Natural estrogen deficiency before menopause3 | |
| Cigarette smoking7 | |
| Vaginal nulliparity | |
| Nonfluctuating estrogen levels8 | |
| Cessation of coital activity9 | |
Presenting Signs and Symptoms
A long-term decrease in estrogen stimulation is generally required before symptoms of atrophic vaginitis arise. A decrease in vaginal lubrication is an early hallmark of hormone insufficiency.10 Genital symptoms include dryness, burning, dyspareunia, loss of vaginal secretions, leukorrhea, vulvar pruritus, feeling of pressure, itching and yellow malodorous discharge.3,6,11 Urinary symptoms of urethral discomfort, frequency, hematuria, urinary tract infection, dysuria and stress incontinence may be later symptoms of vaginal atrophy (Table 2).3,6,10,11 All atrophic vaginitis symptoms can be exacerbated by a simultaneous infection of candidiasis, trichomoniasis or bacterial vaginosis. Over time, the lack of vaginal lubrication often results in sexual dysfunction and associated emotional distress.
TABLE 2 Presenting Symptoms of Atrophic Vaginitis
Diagnosis
PHYSICAL EXAMINATION
It is important not to assume a diagnosis of atrophic vaginitis (or solely a diagnosis of atrophic vaginitis) in the postmenopausal patient who presents with urogenital complaints. A patient history should include attention to exogenous agents that may cause or further aggravate symptoms. Perfumes, powders, soaps, deodorants, panty liners, spermicides and lubricants often contain irritant compounds.6 In addition, tight-fitting clothing and long-term use of perineal pads or synthetic materials can worsen atrophic symptoms12 (Table 3).6,12
TABLE 3 Differential Diagnosis of Atrophic Vaginitis
| Candidiasis | |
| Bacterial vaginosis | |
| Trichomoniasis | |
| Contact irritation or reaction to: | |
| Perfumes | |
| Powders | |
| Deodorants | |
| Panty liners | |
| Perineal pads | |
| Soaps | |
| Spermicides | |
| Lubricants | |
| Tight-fitting or synthetic clothing | |
Information from Beard MK. Atrophic vaginitis. Can it be prevented as well as treated? Postgrad Med 1992;91:257–60, and Beard MK, Curtis LR. Libido, menopause, and estrogen replacement therapy. Postgrad Med 1989;86:225–8.
On examination, several signs of vaginal atrophy will be evident. Atrophic epithelium appears pale, smooth and shiny. Often, inflammation with patchy erythema, petechiae and increased friability may be present. External genitalia should be examined for diminished elasticity, turgor of skin, sparsity of pubic hair, dryness of labia, vulvar dermatoses, vulvar lesions and fusion of the labia minora3,6 (Figure 1). Introital stenosis to a width less then two fingers and decreased vaginal depth will be apparent; if these conditions are not diagnosed before insertion of the speculum, the pelvic examination will cause considerable pain. Friable and poorly rugated vaginal epithelium is more prone to traumatic damage. Ecchymoses and minor lacerations at periintroital and posterior fourchette may also recur after coitus or during a speculum examination. Vaginal examination or sexual activity can result in vaginal bleeding or spotting. Vulvar signs of irritation caused by urinary incontinence may also be identified on pelvic examination. Cystocele, urethral polyps, urethral caruncle, eversion of urethral mucosa, pelvic organ prolapse and rectocele often accompany atrophic vaginitis3 (Table 4).3,6
FIGURE 1.
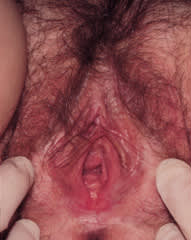
External genitalia of a 67-year-old woman who is naturally menopausal for two years and is not on estrogen replacement therapy. Note loss of labial and vulvar fullness, pallor of urethral and vaginal epithelium, and decreased vaginal moisture.
TABLE 4 Physical Signs of Atrophic Vaginitis
| Genital | |
| Pale, smooth or shiny vaginal epithelium | |
| Loss of elasticity or turgor of skin | |
| Sparsity of pubic hair | |
| Dryness of labia | |
| Fusion of labia minora | |
| Introital stenosis | |
| Friable, unrugated epithelium | |
| Pelvic organ prolapse | |
| Rectocele | |
| Vulvar dermatoses | |
| Vulvar lesions | |
| Vulvar patch erythema | |
| Petechiae of epithelium | |
| Urethral | |
| Urethral caruncle | |
| Eversion of urethral mucosa | |
| Cystocele | |
| Urethral polyps | |
| Ecchymoses | |
| Minor lacerations at peri-introital and posterior fourchette | |
Information from Pandit L, Ouslander JG. Post-menopausal vaginal atrophy and atrophic vaginitis. Am J Med Sci 1997;314:228–31, and Beard MK. Atrophic vaginitis. Can it be prevented as well as treated? Postgrad Med 1992;91:257–60.
LABORATORY FINDINGS
Laboratory diagnostic testing, including serum hormone levels and Papanicolaou smear, can confirm the presence of urogenital atrophy (Figures 2 and 3; Table 5).3,13 Cytologic examination of smears from the upper one third of the vagina show an increased proportion of parabasal cells and a decreased percentage of superficial cells. An elevated pH level (postmenopausal pH levels exceeding 5),3 monitored by a pH strip in the vaginal vault, may also be a sign of vaginal atrophy. In addition, a vaginal ultrasonogram of the uterine lining that demonstrates a thin endometrium measuring between 4 and 5 mm signifies loss of adequate estrogenic stimulation.13 On microscopic evaluation, loss of superficial cells is obvious with atrophy, but there may also be evidence of infection with Trichomonas, candida or bacterial vaginitis.
FIGURE 2.
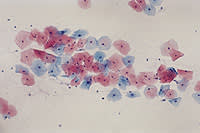
Normal cervical Papanicolaou smear demonstrating squamous cells from the superficial and intermediate layers of the epithelium. The cells have abundant cytoplasm and a low nuclear-cytoplasmic ratio. As the cells mature toward the surface, the cytoplasm becomes keratinized, acquiring a pink color, and the nucleus turns small and condensed, reflecting pyknosis.
FIGURE 3.
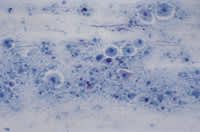
Papanicolaou smear demonstrating atrophic vaginitis with immature (parabasal) squamous epithelial cells with enlarged nuclei in a background of basophilic granular debris and inflammatory exudate. Characteristic round-shaped, amorphous basophilic structures (“blue blobs”) are present.
TABLE 5 Laboratory Diagnosis of Atrophic Vaginitis
| Laboratory test | Positive indication |
|---|---|
| Wet preparation/cytologic smear of cells from upper one third of vagina | Atrophic cytologic changes including increase in proportion of parabasal cells |
| Ultrasonograhy of uterine lining | Uterine lining demonstrating endometrial thinness between 4 and 5 mm13 |
| Serum hormone concentration | Low level of circulating estrogen ≤ 4.5 |
| Vaginal pH | pH elevation above normal postmenopausal levels (pH exceeding 5)3 |
| Microscopy | Elimination of diagnosis of trichomoniasis, candidiasis and bacterial vaginosis |
Information from Pandit L, Ouslander JG. Postmenopausal vaginal atrophy and atrophic vaginitis. Am J Med Sci 1997;314:228–31, and Osmers R, Volksen M, Schauer A. Vaginosonography for early detection of endometrial carcinoma? Lancet 1990;335:1569–71.
Treatment
ESTROGEN REPLACEMENT
Because the lack of circulating, natural estrogens is the primary cause of atrophic vaginitis, hormone replacement therapy is the most logical choice of treatment and has proved to be effective in the restoration of anatomy and the resolution of symptoms. Estrogen replacement restores normal pH levels and thickens and revascularizes the epithelium. Adequate estrogen replacement therapy increases the number of superficial cells.3 Estrogen therapy may alleviate existing symptoms or even prevent development of urogenital symptoms if initiated at the time of menopause. Contraindications to estrogen therapy include estrogen-sensitive tumors, end-stage liver failure and a past history of estrogen-related thromboembolization. Adverse effects of estrogen therapy include breast tenderness, vaginal bleeding and a slight increase in the risk of an estrogen-dependent neoplasm.14 An increased risk of developing endometrial carcinoma and hyperplasia is conclusively related to unopposed, exogenous estrogen intake.15 Factors that determine the degree of increased risk include duration, dosage and method of estrogen delivery. Routes of administration include oral, transdermal and intravaginal. Dose frequency may be continuous, cyclic or symptomatic. The amount of estrogen and the duration of time required to eliminate symptoms depend greatly on the degree of vaginal atrophy and vary among patients.
Systemic administration of estrogen has been shown to have a therapeutic effect on symptoms of atrophic vaginitis. Additional advantages of systemic administration include a decrease in postmenopausal bone loss and alleviation of vasomotor dysfunction (hot flushes). Standard dosages of systemic estrogen, however, may not eliminate the symptoms of atrophic vaginitis in 10 to 25 percent of patients.16 Systemic estrogen in higher dosages may be necessary to alleviate symptoms. Some women require coadministration of a vaginal estrogen product that is applied locally. Up to 24 months of therapy may be necessary to totally eradicate dryness; however, some patients do not fully respond even to this treatment regimen.10
Other treatment options include transvaginal delivery of estrogen in the form of creams, pessaries or a hormone-releasing ring (Estring). Treatment with a low-dose transvaginal estrogen has proved effective in relieving symptoms without causing significant proliferation of the vaginal epithelium.2,12,14,17 The genitourinary pH level is also lowered, leading to a decreased incidence of urinary tract infections. Absorption rates increase with treatment duration because of the enhanced vascularity of the treated epithelium. The advantage of transvaginal treatment may be a decreased risk of endometrial carcinoma because a lower hormone amount is required to eliminate urogenital symptoms. Negative effects of transvaginal treatment include patient dislike of vaginal manipulation, less prevention of postmenopausal bone loss and vasomotor dysfunction, decreased control of absorption with vaginal creams compared to oral and transdermal delivery, and irregular treatment intervals that may cause patients to forget to administer the treatment.6
Transvaginal rings offer convenience, constancy of hormonal concentration in the blood stream and a therapeutic value equivalent to creams without the need for frequent application. Control of hormone dosage is manipulated by changing the surface area of the ring. Atrophic vaginitis symptoms are relieved (with a dosage of 5 to 10 μg per 24 hours) without stimulation of endometrial proliferation, thereby eliminating the need to add opposing progestogen to the regimen.18 Rings may be removed and reinserted by most patients with little difficulty and can be worn during coitus.
MOISTURIZERS AND LUBRICANTS
Moisturizers and lubricants may be used in conjunction with estrogen replacement therapy or as alternative treatments.17 Some patients choose not to take hormone replacement, or they may have medical contraindications or experience hormonal side effects. Patients who wish to avoid using estrogen should not use moisturizers that contain ginseng because they may have estrogenic properties.19 Moisturizers help maintain natural secretions and coital comfort. The length of effectiveness is generally less than 24 hours.
Sexual Activity
Sexual activity is a healthful prescription for postmenopausal women who have a substantially estrogenized vaginal epithelium. It has been shown to encourage vaginal elasticity and pliability, and the lubricative response to sexual stimulation. Women who participate in sexual activity report fewer symptoms of atrophic vaginitis and, on vaginal examination, have less evidence of stenosis and shrinkage in comparison with sexually inactive women. A negative relationship exists between coital activity, including masturbation, and symptoms of vaginal atrophy.9
Because no positive relationship has been shown to exist between estrogen levels and sexual activity, coitus is not hypothesized to restore or maintain estrogen in postmenopausal women. The existence of a positive relationship between coital activities and both gonadotropins and androgens indicates the importance of these compounds to healthy vaginal epithelium when estrogen levels are decreased.9 All sexually active women should take appropriate precautions against sexually transmitted diseases, including the human immunodeficiency virus.
Final Comment
Vaginal atrophy need not be an inevitable consequence of menopause or other events that result in long-term estrogen loss. Active diagnosis and intervention may prevent development of atrophic vaginitis or eliminate existing symptoms. Awareness of the many choices for delivery of estrogen replacement, as well as alternative therapies, greatly increases a physician's ability to prescribe treatment that is compatible with a patient's physical needs and lifestyle. In the appropriate circumstances, encouragement of sexual activity is also an important source of nonpharmacologic treatment about which many patients may not be informed. Ironically, continued coital relations may enhance a woman's ability to enjoy a healthy sex life after menopause by encouraging maintenance of a physiologic environment defensive to atrophic changes.

