Common genitourinary health issues that arise in the care of male patients include prostatitis, benign prostatic hyperplasia, urogenital cancers, premature ejaculation and erectile dysfunction. Bacterial infections are responsible for only 5 to 10 percent of prostatitis cases. Benign prostatic hyperplasia is present in 90 percent of men by the age of 85. Common urogenital cancers include prostate cancer, transitional cell carcinoma of the bladder and testicular cancer. Although an estimated 10 percent of men eventually develop prostate cancer, screening for this malignancy is one of the most controversial areas of health prevention. Premature ejaculation occurs in as many as 40 percent of men. Treatment with tricyclic antidepressants, selective serotonin reuptake inhibitors, counseling or behavioral therapy may be helpful. Erectile dysfunction affects up to 30 percent of men between 40 and 70 years of age. Stepped therapy is a useful approach to this common malady. Good treatment results have been obtained with orally administered sildenafil and intraurethrally administered alprostadil.
Early genitourinary symptoms or problems in men may not be brought to the family physician's attention unless direct questions are asked to assess sexual and urinary function. Common genitourinary health issues in men include prostatitis, benign prostatic hyperplasia, urogenital cancer, premature ejaculation and erectile dysfunction. Screening questions directed at identifying these health problems should be included in the comprehensive health evaluation.
Prostatitis
Prostatitis has traditionally been divided into four clinical categories: acute bacterial prostatitis, chronic bacterial prostatitis, non-bacterial prostatitis and prostatodynia. Only 5 to 10 percent of prostatitis cases have a bacterial etiology.1
ACUTE BACTERIAL PROSTATITIS
Acute bacterial prostatitis typically presents as an acute febrile illness accompanied by rectal, perineal or lower back pain. Patients may also have chills, myalgias, arthralgias, dysuria and irritative urinary symptoms. An enlarged, warm, tender prostate might be noted on digital rectal examination (note, however, that this examination generally is not recommended because of concern about precipitating bacteremia).
Escherichia coli is identified as the causative organism in 80 percent of cases.1 Acute bacterial prostatitis can also be caused by Pseudomonas aeruginosa, Neisseria gonorrhoeae, Mycobacterium tuberculosis, Serratia, Klebsiella and Proteus species, and other enteric organisms.
Depending on the severity of the infection, patients with acute bacterial prostatitis may need to be hospitalized for intravenous antibiotic therapy. Outpatient oral antibiotic therapy can be used in patients with less severe illness. Effective agents include the fluoroquinolones (e.g., ciprofloxacin [Cipro], levofloxacin [Levaquin] and ofloxacin [Floxin]), ampicillin, doxycycline (Vibramycin) and trimethoprim-sulfamethoxazole (Bactrim, Septra). Typically, treatment is continued for four to six weeks to prevent the development of chronic prostatitis and prostatic abscess.1
CHRONIC BACTERIAL PROSTATITIS
Chronic bacterial prostatitis is caused by the same organisms as acute bacterial prostatitis. It occurs more frequently in older men and is described as a relapsing disease with acute exacerbations.
In the absence of symptoms, chronic bacterial prostatitis is sometimes discovered by the presence of bacteriuria. Typically, the condition is detected on the basis of recurring urinary tract infections, dysuria, urinary urgency and hesitancy, or suprapubic, perineal, rectal and low back pain. Fever is typically absent. In the physical examination, a normal prostate is usually found, although the gland can sometimes be boggy and tender. The ultimate diagnosis is based on the patient's symptoms and, subsequent to prostatic massage, examination of prostatic secretions for white blood cells.
Chronic bacterial prostatitis is difficult to treat because of the poor penetration of antibiotics into an uninflamed prostate. The fluoroquinolones can be effective, with cephalexin (Keflex), doxycycline and carbenicillin (Miostat) as good alternatives. Treatment duration is debatable, but symptoms often improve after three months of therapy. Reported treatment success rates have varied from 30 to 100 percent.1
NONBACTERIAL PROSTATITIS AND PROSTATODYNIA
Nonbacterial prostatitis and prostatodynia are eight times more common than acute and chronic bacterial prostatitis.1
Nonbacterial prostatitis is described as pain and inflammation of the prostate with no apparent causative organism. Secretions elicited by prostatic massage reveal numerous white blood cells, but cultures are negative. Although organisms such as Chlamydia trachomatis, Ureaplasma urealyticum, Mycoplasma hominis and Trichomonas vaginalis have been postulated as etiologies, conclusive evidence is lacking.1
Prostatodynia is described as pain and other symptoms localized to the prostate but with no obvious inflammation of the gland and no white blood cells in prostatic secretions.
Nonbacterial prostatitis and prostatodynia typically present with pain, urinary urgency, weak stream, nocturia, urinary hesitancy and sexual dysfunction (e.g., painful ejaculation, postejaculatory pain and hematospermia). The conditions are easily confused with benign prostatic hyperplasia. Physical examination of the prostate is normal.
Although bacteria have not been shown to cause these syndromes, antimicrobial therapy directed at C. trachomatis and U. urealyticum can be used empirically. Agents such as tetracycline, doxycycline or erythromycin are reasonable choices. Treatment should be continued for a minimum of six weeks in responding patients.1 Alpha1-adrenergic blocking agents and nonsteroidal anti-inflammatory drugs may relieve symptoms. Nonbacterial prostatitis and prostatodynia are difficult to cure, but symptoms improve in up to 50 percent of treated patients.1
Benign Prostatic Hyperplasia
Benign prostatic hyperplasia, the most common benign tumor in men, is responsible for more than 1.7 million office visits every year.2 This condition is identifiable in 50 percent of men at 60 years of age and in 90 percent of men by 85 years of age.3
Treatment is necessary in only about 50 percent of men who develop hyperplasia.3 The symptoms of benign prostatic hyperplasia are obstructive or irritative. Obstructive symptoms include weak urinary stream, hesitancy, terminal dribbling, sensation of incomplete bladder emptying and acute urinary retention. Irritative symptoms include nocturia (more than once a night), frequency of urination (more than eight times a day during normal daytime hours), urgency of urination and dysuria.3 The American Urological Association Symptom Index for Benign Prostatic Hyperplasia has been developed to help quantify urinary symptoms and assess treatment success4 (Figure 1).3
FIGURE 1. AUA Symptom Index
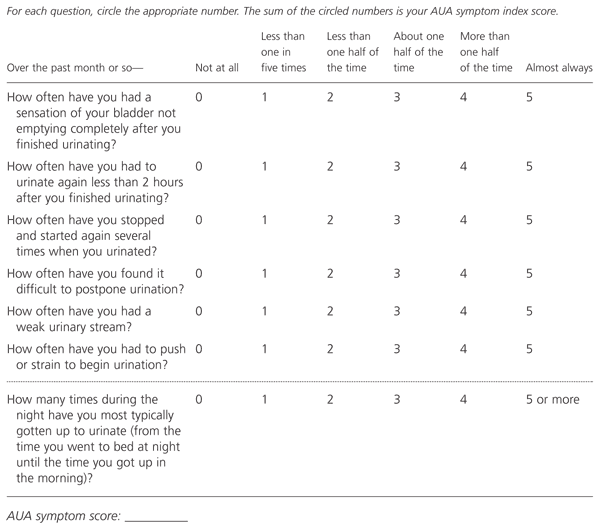
American Urological Association (AUA) symptom index for benign prostatic hyperplasia.
Adapted with permission from Madsen FA, Bruskewitz RC. Clinical manifestations of benign prostatic hyperplasia. Urol Clin North Am 1995;22:291–8.
Patients with mild symptoms often require no intervention, but those with moderate to severe symptoms can benefit dramatically from therapy. Treatment options include medical therapy, transurethral incision of the prostate, transurethral resection of the prostate, prostatectomy and balloon dilation.
Medical therapy is attempted first, with surgical intervention reserved for use in patients who do not respond to pharmacologic treatment. Agents that have been used to treat benign prostatic hyperplasia include alpha1-adrenergic blocking agents, 5-alpha-reductase enzyme inhibitors and various herbal remedies.
The alpha1-adrenergic blocking agents include doxazosin (Cardura), tamsulosin (Flomax) and terazosin (Hytrin). These agents block alpha-adrenergic receptors and thereby prompt smooth-muscle relaxation in the prostate and the neck of the bladder.5 Side effects include postural hypotension and syncope, but the alpha1-adrenergic blocking agents are generally well tolerated if they are started in lower dosages and slowly titrated upward. Because tamsulosin has greater specificity for the receptors in the bladder and prostate, it appears to be associated with a lower incidence of orthostatic effects than its counterparts; however, use of tamsulosin increases the risk of ejaculatory dysfunction.5 Dosage ranges for alpha1-adrenergic blocking agents used in the treatment of benign prostatic hyperplasia are given in Table 1.5
Finasteride (Proscar), a 5-alpha-reductase enzyme inhibitor, exerts its effect by inhibiting the conversion of testosterone to dihydrotestosterone in the prostate. Long-term inhibition causes the gland to shrink. Unfortunately, although finasteride can shrink the prostate by 25 percent or more, this effect does not correlate with a reduction in symptoms.5 Finasteride is most useful in symptomatic men with prostate volumes greater than 40 mL.6 Side effects include impotence, decreased libido and decreased ejaculatory volume.5
Saw palmetto is commonly used to treat benign prostatic hyperplasia in Europe. This herbal remedy is also becoming increasingly popular in the United States. The mechanism of action of saw palmetto is unknown, and the U.S. Food and Drug Administration has not labeled it for the treatment of benign prostatic hyperplasia.5
Urogenital Cancer
PROSTATE CANCER
Prostate cancer, with 209,900 cases identified in 1997, is the most frequent new cancer diagnosis in men.7 Cancer of the prostate is second only to lung cancer as the leading cause of cancer deaths in men, with 41,000 deaths reported in 1997 alone.7 An estimated one in 10 men develop this malignancy at some point in life.8
Despite the frequency of prostate cancer, screening programs remain controversial because of inconclusive evidence demonstrating improved outcomes in patients diagnosed at an earlier disease stage.9 Use of the prostatic specific antigen (PSA) test for prostate cancer screening is one of the most controversial issues in disease prevention. Conflicting screening recommendations have been presented by various organization and associations.
The authors of this article believe that PSA screening should be addressed on an individual basis after 50 years of age (40 years of age in blacks and in all patients with a significant family history). Patients should be advised annually about the risk of prostate cancer, the lack of evidence supporting PSA screening as a way to modify outcome, the risk of both false-positive and false-negative screening results that can cause unnecessary testing or false reassurance, the side effects of potential treatments for prostate cancer and the limitations that comorbid illnesses may impose on treatment. If, after appropriate counseling, the patient desires PSA testing, we believe that this screening test should be made available.10
Compared with the PSA test alone, the “percent-free PSA” is thought to be more sensitive in identifying patients at risk for prostate cancer. A percent-free PSA of greater than 25 percent is 95 percent sensitive in excluding prostate cancers when PSA values are in the ambiguous range of 4 to 10 ng per mL (4 to 10 μg per L).11
Risk factors for prostate cancer include family history and black race. The presence of benign prostatic hyperplasia is not considered to be a significant risk factor. Smoking and dietary habits do not seem to contribute to the incidence of this cancer.8 Although early reports showed a slight increase in the incidence of prostate cancer among men who had vasectomies, current data do not support a causal relationship.12
Prostate cancer is an indolent disease in most men. Well-differentiated prostate cancer has a five-year survival rate approaching 98 percent and a 15-year survival rate approaching 83 percent. Conversely, poorly differentiated forms of this cancer have only a 68 percent five-year survival rate and a 25 percent 15-year survival rate. Unfortunately, men diagnosed with prostate cancer before the age of 65 have a higher likelihood of metastatic disease and subsequently poorer outcomes.8
The treatment of prostate cancer can include prostatectomy, radiation therapy, hormonal therapy and observation. Each method has its own benefits and limitations, and comprehensive discussion is beyond the scope of this review. However, the family physician must help tailor treatment to the patient's age, current disease stage, comorbid illnesses, lifestyle and life expectancy.9
TRANSITIONAL CELL CARCINOMA OF THE BLADDER
Transitional cell carcinoma of the bladder is the second most common urogenital cancer. In 1998, an estimated 54,000 cases of this malignancy were diagnosed, and 12,500 persons died of the disease. Its incidence is 2.5 times higher in men than in women, and it is found more frequently in whites than in blacks.8
The malignancy typically presents as gross or microscopic hematuria, although dysuria, urinary urgency and urinary frequency are other manifestations. The diagnosis is obtained by cystoscopy, based on visualization of abnormalities of the bladder mucosa.
Treatment depends on cancer staging and varies from intravesicular chemotherapy to radical cystoprostatectomy. If left untreated, transitional cell carcinoma of the bladder can metastasize rapidly. The family physician must be aware of the possibility of this potentially lethal cancer in patients with hematuria or urinary symptoms inconsistent with other etiologies.8
TESTICULAR CANCER
Testicular carcinoma is the leading cause of solid tumors in men 20 to 34 years of age, with an incidence of approximately three cases per 1,000 men in this age group.13 Men with a history of undescended testicles or orchiopexy to descend the testicles have a 2.5 to 20 times greater risk of testicular carcinoma compared with normal men.14
This malignancy typically presents asymptomatically as a lump or mass on the testicle. Although the U.S. Preventive Health Services Task Force failed to find conclusive evidence supporting regular formal testicular cancer screening or self-examination,15 the authors of this article believe that it is important to teach adolescent males to perform testicular self-examination because this neoplasm is nearly 100 percent curable with early treatment. A delay in diagnosis can lead to more advanced disease and higher morbidity and mortality rates.
A useful approach to testicular self-examination is to advise younger male patients to examine themselves at least once a month in a warm shower. Warm water relaxes the cremaster muscle and allows scrotal lengthening. The patient is instructed to hold the testicle between the thumb and first two fingers. The thumb should be behind the testicle. With the two fingers, the patient gently feels the entire surface of the testicle, searching for a hard nodule that is clearly different in texture or consistency than the normal rubbery, homogeneous surface of the testicle. A useful guide is to describe the normal testicular surface as a hard-boiled egg without the shell. Patients should be informed of the location of the epididymis. They need to know that this structure is the only normal “lump” on the testicle. Any other mass should be promptly reported to the physician.16
Premature Ejaculation
Premature ejaculation is best defined as persistent or recurrent ejaculation with minimal stimulation before, on or shortly after penetration, and before the sexual partner wishes it. Premature ejaculation is thought to be the most common form of male sexual dysfunction, with an estimated prevalence of up to 40 percent.17
Many men are hesitant to discuss the problem of premature ejaculation with their physician. A relaxed, comfortable setting can facilitate communication. In addition, the family physician can prompt discussion by including questions about premature ejaculation in the annual health assessment.
Treatment of ejaculatory dysfunction centers on relationship counseling, behavioral therapy and pharmacologic interventions. Relationship counseling is of paramount importance because premature ejaculation can create discord and increase the tension surrounding sexual intercourse. Because many therapies involve the patient's sexual partner, it is important to educate both members of the relationship about premature ejaculation and to facilitate communication between the partners.
Behavioral therapy has been considered the gold standard of treatment, with reported success rates ranging from 60 to 95 percent.18 Techniques include the Seman's pause maneuver, the Masters and Johnson pause-squeeze technique and the Kaplan stop-start method. These techniques are directed at the induction of male sexual arousal to the point of ejaculation followed by relaxation before orgasm is allowed to occur. The methods can be self-applied, although with suboptimal outcomes; hence, involvement of the sexual partner is essential. Short-term gains can be substantial, but long-term outcomes are less favorable and depend on continued partner involvement.17
Because of the limitations of behavioral therapy, pharmacologic interventions are often used to treat premature ejaculation. Anorgasmia and delayed ejaculatory response are well-known side effects of tricyclic antidepressants and selective serotonin reuptake inhibitors. Recent studies have shown that these drugs modify the ejaculatory response in men with premature ejaculation.17,18 In one of these studies,18 clomipramine (in a dosage of 50 mg per day) and sertraline (in a dosage of 100 mg per day) were found to increase ejaculatory latency from 46 seconds to 5.7 and 4.2 minutes, respectively. Sexual satisfaction was higher with clomipramine, but sertraline was better tolerated and had a better safety profile.
Erectile Dysfunction
Erectile dysfunction can be defined as the inability to achieve and maintain penile erection sufficient to perform satisfactory sexual activity. Between 20 to 30 million men in the United States have some form of erectile dysfunction. The disorder is present in approximately 30 percent of men 40 to 70 years of age.19,20
Erectile dysfunction can be categorized as organic, nonorganic (psychogenic), or mixed. Organic causes account for 70 percent of cases, and about 30 percent of cases are purely psychogenic.20 Common causes of erectile dysfunction are listed in Table 2.20 A thorough history (including a drug history and a sexual history), a physical examination and appropriate targeted laboratory studies (urinalysis, complete blood count, routine blood chemistries, thyroid-stimulating hormone level and testosterone level) are usually sufficient to categorize a patient's erectile dysfunction.
Step 1 and step 2 treatment options, which include orally administered drugs and the use of vacuum devices to induce and sustain an erection, can be initiated based on the results of the general assessment, without the need for a more extensive diagnostic work-up. If the patient fails to respond to step 1 and step 2 therapy and desires more invasive treatment, consideration should be given to urologic referral and the use of step 3 medications and step 4 surgical procedures (Table 3). The keys to the success of this stepped approach include awareness of the problem, categorization of the etiology, initiation of therapy, and follow-up to determine if the patient requires further testing, treatment and referral.
TABLE 3. A Stepped Approach to the Treatment of Erectile Dysfunction
| Step 1—oral agents | |
| Sildenafil (Viagra): 25, 50 or 100 mg orally 1 hour before coitus (usual initial dose is 50 mg)* | |
| Investigational agents (e.g., L-arginine) | |
| Step 2—vacuum or constriction devices | |
| Step 3—penile therapies | |
| Intracavernosal injections: prostaglandin E (alprostadil [Caverject, Prostin VR]), paperverine (Pavabid), phentolamine (Regitine) | |
| Intraurethral insertion: prostaglandin E (alprostadil [Muse]) | |
| Step 4—penile surgery | |
| Penile prostheses: semirigid or malleable rod, hydraulic inflatable device | |
| Penile vascular surgery | |
*—Because potentially fatal hypotensive effects can occur when sildenafil is used concurrently with nitrates, this drug should not be prescribed to patients who are taking nitrate medications or who may take nitroglycerin after exertional angina.
It is encouraging that many effective treatments are available for patients with erectile dysfunction. Reported success rates for these measures are presented in Table 4.20 In particular, the success rates of sildenafil (Viagra) and alprostadil (Caverject, Prostin VR) are approximately 80 percent.20 A word of caution must be given about the use of sildenafil. Because potentially fatal hypotensive side effects can occur when this agent is used concurrently with nitrates, it should not be prescribed to patients who are taking nitrate medications or who may take nitroglycerin after exertional angina.
TABLE 4. Success Rates for Erectile Dysfunction Therapy
| Treatment | Treatment success rate (%) | |
|---|---|---|
| Psychogenic cause | All causes | |
| Sildenafil (Viagra) | 80 | 56 to 85 |
| Vacuum constriction | 80 | |
| Penile injections | 89 | |
| Penile prostheses | 95 | |
| Penile surgery | Not reported | |
Information from Lawless C, Cree J. Oral medications in the management of erectile dysfunction. J Am Board Fam Pract 1998;11:307–14.

