Approximately 65 percent of patients with acute myocardial infarction report experiencing symptoms of depression. Major depression is present in 15 to 22 percent of these patients. Depression is an independent risk factor in the development of and mortality associated with cardiovascular disease in otherwise healthy persons. Persons who are depressed and who have pre-existing cardiovascular disease have a 3.5 times greater risk of death than patients who are not depressed and have cardiovascular disease. Physicians can assess patients for depression by using one of several easily administered and scored self-report inventories, including the SIG E CAPS + mood mnemonic. Cognitive-behavior therapy is the preferred psychologic treatment. Selective serotonin reuptake inhibitor antidepressants are the recommended pharmacologic treatment because of the relative absence of effects on the cardiovascular system. The combination of a selective serotonin reuptake inhibitor with cognitive-behavior therapy is often the most effective treatment for depression in patients with cardiovascular disease.
As many as 65 percent of patients with acute myocardial infarction report experiencing symptoms of depression; major depression is present in 15 to 22 percent of these patients.1 During the past two decades, a substantial body of evidence has established a link between depression, cardiovascular disease and mortality.2,3 Two large, community epidemiologic studies4,5 demonstrated a significant relationship between depression and mortality in patients with myocardial infarction. Results from another study6 showed that depression contributes to a greater chance of developing or dying of heart disease in persons who are otherwise healthy, even after controlling for smoking status, gender, weight, activity, blood pressure and cholesterol levels. Results from additional studies2 have supported the contribution of depression as an independent risk factor for cardiovascular disease in persons who were initially free of the disease.
Other research7,8 has shown that persons who are depressed and have pre-existing cardiovascular disease have a 3.5 times greater risk of dying of a myocardial infarction than patients with cardiac disease who are not depressed. In a recent study,9 depression was shown to be associated with an increased risk of developing coronary heart disease in men and women. Depression was shown to increase mortality related to coronary heart disease in men but had no effect on mortality in women.
Diagnosis
Depression is frequently undiagnosed and untreated in patients with cardiovascular disease. It is estimated that only 25 percent or less of cardiac patients with major depression are diagnosed with depression, and only about one half of those patients receive treatment for depression.3 Depression may be undiagnosed and untreated in patients with cardiovascular disease because of the following factors: (1) some symptoms, such as fatigue and insomnia, are common to depression and coronary heart disease; (2) physicians and patients may erroneously believe that depression is a normal reaction to cardiovascular disease; (3) patients may be reluctant to report symptoms of depression in this setting; (4) physicians may be reluctant to ask their patients about depression1; and (5) physicians may be reluctant to prescribe antidepressant medications to patients with cardiovascular disease because of potential adverse side effects.
TABLE 1 Mnemonic for Assessment of Depression: SIG E CAPS + Mood
| S | Sleep (insomnia or hypersomnia) |
| I | Interests (diminished interest in or pleasurefrom activities) |
| G | Guilt (excessive or inappropriate guilt; feelings of worthlessness) |
| E | Energy (loss of energy or fatigue) |
| C | Concentration (diminished concentration or indecisiveness) |
| A | Appetite (decrease or increase in appetite; weight loss or gain) |
| P | Psychomotor (retardation or agitation) |
| S | Suicide (recurrent thoughts of death, suicidal ideation or suicide attempt) |
| + | Mood |
Assessment
A diagnosis of major depressive disorder requires the presence of five or more of the following symptoms as recommended by The Diagnostic and Statistical Manual for Mental Disorders, 4th ed.10 (DSM-IV): (1) depressed mood; (2) markedly diminished interest or pleasure in all or nearly all activities; (3) significant weight loss or gain, or a decrease or increase in appetite; (4) insomnia or hypersomnia; (5) psychomotor agitation or retardation; (6) fatigue or loss of energy; (7) feelings of worthlessness or excessive or inappropriate guilt; (8) diminished ability to think or concentrate, or indecisiveness; and (9) recurrent thoughts of death, suicidal ideation or suicide attempt.
These symptoms must be present nearly every day for two weeks and one of the symptoms must be depressed mood or loss of interest or pleasure in activities. These symptoms must represent a change from previous functioning resulting in social, occupational or other life impairment, and they cannot be the direct result of substance use, a medical condition or bereavement. The mnemonic SIG E CAPS + Mood (Table 1) can be used to remember these assessment criteria.
Assessing for depression in patients with myocardial infarction requires an understanding of the risk factors for depression. These include female gender, previous history of depression, family history of depression, lack of social support (especially if living alone) and loss of functioning or major life role. This information can be acquired through a complete interview with the patient and his or her significant other. In addition, any of the selfreport measures presented in Table 211–15 can be used to assess for depression in patients with myocardial infarction. Several of these assessment tools are readily available and can be easily used in a medical setting. The Zung Self-Rating Depression Scale (SDS) (Figure 1),11 and the Beck Depression Inventory,12 which are 20- and 21-item self-reporting instruments, respectively, cover most of the nine DSM-IV symptom categories for a major depressive episode. The SDS is available in more than 30 languages and, therefore, has broad multicultural applications. The Geriatric Depression Inventory15 takes into account special considerations of older persons.
TABLE 2 Self-Report Instruments for Depression
| Scale | Population | Items | Format | DSM-IV coverage* |
|---|---|---|---|---|
| Beck Depression Inventory12 | Adults | 21 | Multiple choice | 8/9 |
| Center for Epidemiologic Studies–Depression Scale13 | Adults | 20 | Likert scale | 7/9 |
| General Health Questionnaire†14 | Adults | 28 | Likert scale | 3/9 |
| Geriatric Depression Inventory15 | Older adults (> 55 years) | 30 | Yes/no answers | 6/9 |
| Zung Self-Rating Depression Scale11 | Adults | 20 | Likert scale | 9/9 |
DSM-IV = The Diagnostic and Statistical Manual for Mental Disorders, 4th ed.
*—None of the self-report instruments listed above assess DSM-IV Exclusion Criteria.
†—Only the seven items from the Depression Subscale are included to determine DSM-IV coverage.
FIGURE 1. Zung Self-Rating Depression Scale
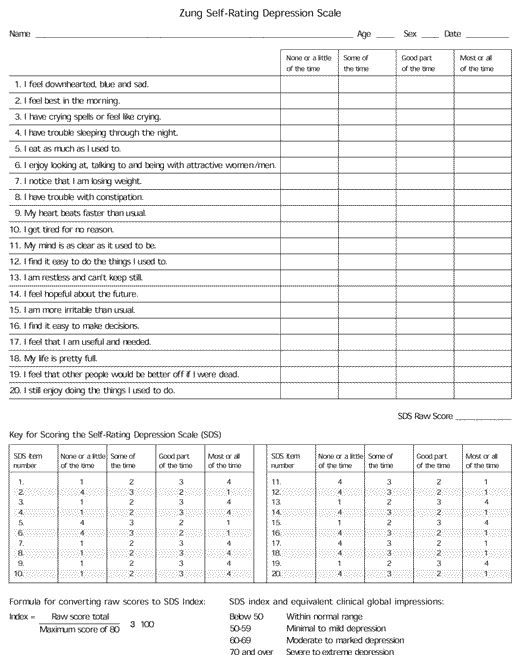
A self-rating depression scale to be used to help diagnose depression.
Adapted with permission from Zung WW. A self-rating depression scale. Arch Gen Psychiatry 1965;12:63–70.
These instruments are sensitive to changes in depression and, as a result, are also useful for monitoring treatment. Despite these benefits, the greatest risk associated with depression screening instruments is the generation of false positive results that may result in harmful labeling and unnecessary treatment or further testing. To reduce this risk, patients with a positive depression score should be interviewed more extensively to confirm or rule out depression.
Psychosocial Treatment
The most effective psychosocial treatment for depression in patients with myocardial infarction is cognitive-behavior therapy. It assumes patients are active participants in treatment and reciprocal interactions are occurring among the five key elements of therapy—environment, thoughts, emotions, behavior and physiology. Cognitive-behavior therapy emphasizes short-term, practical, problem-focused skill development in each of these key areas. Patients are encouraged to change the stressful aspects of their environment that they can control.
Physicians can be extremely helpful when addressing legitimate and erroneous beliefs and expectations about postmyocardial infarction care. For example, it is common for patients and their families to believe that physical activity following myocardial infarction can place the patient at risk of sudden death. As a result, activity may be restricted, which can hinder recovery, maintain depression and increase the risk for developing other health problems. Identifying these misconceptions and providing supportive education can lead to better adherence to treatment by patients and a more positive approach to their behavior, mood and health.
Behaviorally, physicians should encourage reasonable participation in exercise, hobbies and social activities as well as formal cardiac rehabilitation programs. They also can support patients' efforts to use assertive communication skills to set limits. Physiologically, regular use of breathing, relaxation or visual imagery techniques can help reduce anxiety.
Finally, it is crucial to involve family members in the rehabilitation process of patients with myocardial infarction. Family members often experience feelings of helplessness, loss and depression similar to those of the patient. The patients' partners may be fearful about the return to activity, including sexual activity, based on misconceptions. Educating patients' families and partners and providing them with appropriate resource information is crucial to their ability to provide a supportive role in recovery.
Pharmacologic Treatment
Efficacy and adverse effects are important considerations when selecting a pharmacologic agent for treatment of depression following myocardial infarction. Overall, there is little difference in efficacy among the antidepressants in treating mild to moderate depression.16 However, there are significant differences in their adverse effect profiles. Table 317–21 outlines the cardiac effects of many commonly prescribed antidepressants.
During the past two decades, selective serotonin reuptake inhibitors (SSRIs) have emerged as first-line agents in the treatment of mild to moderate depression. Unlike their tricyclic antidepressant (TCA) predecessors, SSRIs have repeatedly been demonstrated to be safe16 and to have a negligible effect on the cardiovascular system, even in cases of overdose. SSRIs have virtually no effect on fast sodium channels or conduction, and are not noted to have any proarrhythmic or antiarrhythmic effects. In a study22 comparing the cardiovascular effects of paroxetine with nortriptyline, which is one of the most tolerable TCAs because it has the least cholinergic blocking activity, the rate of discontinuation secondary to a cardiovascular event was significantly greater in the nortriptyline group (7 in 40) than in the paroxetine group (1 in 41). In addition, this study showed a statistically significant increase in supine and standing pulse rates in the subjects receiving nortriptyline.
TABLE 3 Cardiac Effects of Commonly Used Agents for Treatment of Depression
| Agent | Hypotensive effects | Cardiac conduction | Heart rate | Proarrhythmic effects | |
|---|---|---|---|---|---|
| Tricyclic antidepressants | |||||
| Amitriptyline (Elavil) | +++ | +++ | ++ | ++++ | |
| Imipramine (Tofranil) | +++ | +++ | ++ | ++++ | |
| Nortriptyline (Pamelor) | + | + | ++ | +++ | |
| Atypical antidepressants | |||||
| Bupropion (Wellbutrin) | 0/+ | 0/+ | + | 0 | |
| Trazadone (Desyrel) | ++ | 0 | ++ | ||
| Selective serotonin reuptake inhibitors | |||||
| Fluoxetine (Prozac) | 0 | 0 | — | 0 | |
| Sertraline (Zoloft) | 0 | 0 | — | 0 | |
| Paroxetine (Paxil) | 0 | 0 | 0 | 0 | |
| Fluvoxamine (Luvox) | 0 | 0 | — | 0 | |
| Citalopram (Celexa) | 0/+ | — | 0 | 0/+* | |
| Phenethylamine | — | — | — | — | |
| Venlafaxine (Effexor) | 0† | + | 0/+ | 0/+ | |
0 = negligible, 0/+ = minimal, + = mild, ++ = moderate, +++ = marked, ++++ = severe.
*—Fatalities associated with suicide-implicated arrhythmias as a possible cause of death.
†—Can produce a sustained increase in blood pressure.
Of the newer agents, venlafaxine (Effexor) may be the least desirable for use in patients with pre-existing cardiovascular disease. Statistically significant increases in heart rate and blood pressure have been noted, prompting the manufacturer to recommend periodic blood pressure monitoring.17 In contrast, there have been only scattered reports of bradycardia secondary to SSRIs; however, one report was profound.18,23,24
TCAs can have a negative effect on the cardiovascular system. The tertiary amine tricyclics have a dramatic impact on blood pressure, even among persons without cardiac disease. The hypotensive responses that sometimes occur may have a profound effect on perfusion of previously compromised myocardial tissue. Orthostatic hypotension may become even more pronounced in patients who are taking commonly used agents such as beta blockers and angiotensinconverting enzyme inhibitors following myocardial infarction, or in patients taking diuretics who have hypertension or congestive heart failure.25
The influence of TCAs on cardiac conduction is similar to that of quinidine (Quinaglute) and they are, in fact, effective as class I antiarrhythmics. It was once thought to be a beneficial side effect of TCAs, but the Cardiac Arrhythmia Suppression Trial26 (CAST) report demonstrated that class I antiarrhythmic therapy caused an increase in mortality rates. Approximately 20 percent of patients with significant conduction disturbances, such as bundle branch block, exhibit complications while on TCA therapy.27 Given these disturbing side effects and drug interactions, TCAs must be considered to be contraindicated in the setting of an acute coronary event and used very cautiously, if at all, in patients with a history of significant heart disease.
Bupropion (Wellbutrin) has been studied in cardiac patients. Statistically significant changes in blood pressure were observed with no detrimental effect on cardiac conduction or contractility.19 These patients were given an average daily dosage of 455 mg, which is well above the dosage commonly prescribed for depression or smoking cessation.
The significance of potential interactions between antidepressant and cardiovascular medications must be considered when treating depression in cardiac patients. A number of these agents impact the enzyme substrates of the cytochrome P450 system, and these interactions may be particularly troublesome in patients who are slow metabolizers (an estimated 8 percent of the white population). Fluoxetine (Prozac), an inhibitor of the 2D6 isoform, may decrease the rate of metabolism of various beta blockers, thereby leading to bradycardia. Interactions relevant to medications commonly used by patients with cardiovascular disease are presented in Table 4.21,24,28,29
General Treatment Considerations
Use of cognitive-behavior therapy should be considered if any of the following conditions exist: (1) depression is not severe; (2) depression is not chronic; (3) psychotic features are absent; (4) previous response to cognitive-behavior therapy was positive; (5) cognitive-behavior therapy services are available; (6) a medical contraindication to medications exists; (7) recovery has not been achieved with medications alone; and (8) complicated psychosocial circumstances exist.
TABLE 4 Clinically Relevant Drug Interactions
| Interacting drug | Antidepressant | Potential effect and management |
|---|---|---|
| Clonidine (Catapres-TTS) | TCAs | Attenuated antihypertensive effect of clonidine, exaggerated hypertensive response to clonidine withdrawal. Avoid co-administration. |
| Quinidine (Quinaglute), flecainide (Tambocor), encainide (Enkaid) | TCAs | Prolonged QT interval. Avoid co-administration. |
| Warfarin (Coumadin) | TCAs, fluoxetine (Prozac), paroxetine (Paxil), sertraline (Zoloft) | Elevates prothrombin time. Monitor INR. |
| Aspirin, fluoxetine | TCAs | Elevates TCA blood levels. Monitor for signs and symptoms of toxicity. |
| Metoprolol tartrate (Lopressor), propranolol (Inderal) | Fluoxetine | Bradycardia |
TCA = tricyclic antidepressant, INR = International Normalized Ratio.
Antidepressant medications should be considered in the following circumstances: (1) depression is severe; (2) depression is chronic or recurrent; (3) psychotic features are present; (4) prior response to medication was positive; (5) family history of depression exists; and (6) patient is unable to do the work required in psychotherapy.30 When selecting an antidepressant, considering the points represented by the STEPS acronym can be helpful: Safety, Tolerability, Efficacy, Payment and Simplicity.31 A combination of cognitive-behavior therapy and medication may be most appropriate when symptoms of depression are severe, chronic, fail to resolve with either therapy alone, and if significant psychosocial issues are present.

