
Am Fam Physician. 2001;64(5):769-774
Over the past decade, the conceptual understanding of heart failure has changed significantly. Several large clinical trials have demonstrated that pharmacologic interventions can dramatically reduce the morbidity and mortality associated with heart failure. These trials have extended the therapeutic paradigm for treating heart failure beyond the goal of limiting congestive symptoms of volume overload. This two-part article presents an evidence-based guideline to assist primary care physicians in evaluating and treating patients with heart failure. Part I describes the new paradigm of heart failure and offers guidance for diagnostic testing. Part II presents a treatment guideline.
Approximately 5 million Americans currently have heart failure,1 and an additional 400,000 develop heart failure annually.2 Each year, more than 800,000 patients with heart failure are hospitalized, and 250,000 die.3 Nearly 50 percent of patients die within five years of the onset of symptoms.4 The incidence of heart failure and associated morbidity and mortality is expected to increase in the future.
Until the past decade, only symptomatic therapy with diuretics and digoxin (Lanoxin) was available for patients with heart failure. In the early 1990s, angiotensin-converting enzyme (ACE) inhibitors and direct-acting vasodilators (e.g., isosorbide dinitrate [Isordil] and hydralazine [Apresoline]) were found to improve mortality in patients with heart failure caused by left ventricular systolic dysfunction. Recently, beta blockers and spironolactone (Aldactone) were also found to improve mortality in appropriate patients, and these agents have revolutionized the management of heart failure. However, primary care physicians, who manage the majority of patients with heart failure, have been given little guidance on how to assemble the new data into a form that can be applied in the clinical setting.
Guideline Development
In 1994, the Agency for Health Care Policy and Research (AHCPR), now the Agency for Healthcare Research and Quality (AHRQ), published a clinical practice guideline for the management of heart failure.5 This guideline is now outdated.
In 1999, the Advisory Council to Improve Outcomes Nationwide in Heart Failure published recommendations for the management of chronic heart failure.6 The Heart Failure Society of America also published practice guidelines in 1999.7 Both of these guidelines are long, comprehensive reviews of the literature that were published in cardiology journals and never circulated to primary care audiences. Furthermore, neither has a succinct, clinically based summary that would be relevant to primary care physicians.
The guideline presented in this two-part article was developed by a multidisciplinary panel at the University of Michigan. The goal was to provide an easy-to-use primary care guideline for the management of heart failure caused by left ventricular systolic dysfunction. Since the guideline was released, it has been updated and modified to fit formatting requirements for publication.8
The literature search for the University of Michigan project started with the results of the literature search performed and published by the AHCPR in 1994.5 Subsequent literature was searched in Medline to February 1, 1998. The details of the search are available elsewhere.8 The literature search was supplemented by the results of large clinical trials that were completed during the course of the guideline's development. Furthermore, in the preparation of this article, the results of more recently completed clinical trials were included, and the guidelines published by the Advisory Council to Improve Outcomes Nationwide in Heart Failure6 and the Heart Failure Society of America7 were reviewed. The recommendations in the University of Michigan guideline are weighted by the level of the available evidence, as described in Table 1.8
Etiology
Coronary artery disease producing ischemic cardiomyopathy is the most frequent cause of left ventricular systolic dysfunction. Non-ischemic cardiomyopathies can also produce systolic dysfunction; they may be inherited as genetic disorders or occur sporadically. Patients with both ischemic and nonischemic dilated cardiomyopathies develop similar symptoms of heart failure, suffer frequent hospitalization and are at increased risk for premature death.
Progressive pump failure and malignant arrhythmias are the most frequent causes of death in patients with dilated cardiomyopathy. Thromboembolic events, principally strokes, can complicate the course of heart failure secondary to systolic dysfunction.
In the past, physicians attempted to relieve the symptoms of congestive heart failure by using rotating tourniquets and diuretics. More recently, left ventricular systolic dysfunction was conceptualized in terms of derangements of hemodynamic parameters such as contractility and afterload. Although concerns about fluid status and hemodynamic parameters are still valid, interventions targeted to these issues alone provide primarily symptomatic relief and have little impact on mortality.
| Level of evidence | Description |
|---|---|
| A | Randomized, controlled trials |
| B | Nonrandomized, controlled trials |
| C | Observational trials |
| D | Opinion of expert panel |
The conceptual basis of progressive left ventricular dysfunction has now turned to neurohormones such as norepinephrine, angiotensin and aldosterone. Elevated levels of these hormones have been correlated with poor prognosis.9,10 Interventions targeted at decreasing the impact of these hormones have significantly improved mortality in several recent large mortality trials (described in part II of this article). Thus, the new conceptualization of heart failure has resulted in new approaches to treatment.
Diagnosis
Most clinical trials of therapeutic interventions have included only patients with heart failure confirmed as being caused by left ventricular systolic dysfunction. Although diastolic dysfunction is also a common cause of heart failure, consensus is lacking on the optimal techniques to quantify the degree of dysfunction or reliably make the diagnosis.
| Classification | Description |
|---|---|
| NYHA class I | Asymptomatic |
| NYHA class II | Symptoms with moderate exertion |
| NYHA class III | Symptoms with minimal exertion |
| NYHA class IV | Symptoms at rest |
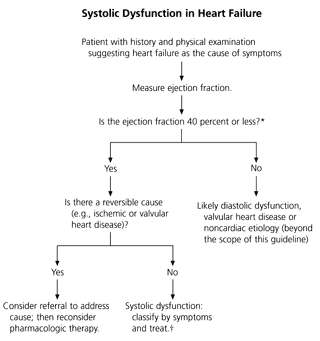
The University of Michigan guideline applies only to patients with left ventricular systolic dysfunction, defined as an ejection fraction of less than 40 percent. In managing patients with symptoms of heart failure, it is critical to measure the ejection fraction to determine whether the symptoms are due to systolic dysfunction or another cause. Widely available techniques can quantify the left ventricular ejection fraction and estimate the degree of systolic dysfunction.
PRESENTING SIGNS AND SYMPTOMS
Heart failure often presents initially as dyspnea with exertion or recumbency. Patients also commonly have dependent edema, rapid fatigue, cough and early satiety. These symptoms are sometimes attributed to other causes. Arrhythmias causing palpitations, dizziness or aborted sudden death may also be the initial manifestations of the disease.
The symptoms of heart failure can mimic those of many other conditions, sometimes making accurate diagnosis difficult. The University of Michigan guideline assumes that heart failure has already been suspected or diagnosed. Guidance is then given on the studies that ought to be considered or performed (Figure 1).8
CLASSIFICATION
Heart failure limits exercise capacity. In general, patients with more severe functional limitations have poorer survival. Physicians commonly use the four-tier New York Heart Association (NYHA) classification of functional capacity to estimate prognosis in clinical practice and to selectively define study populations in clinical trials10 (Table 2).
Because the definitions of the NYHA classes are subjective, standardization of the classification scheme has eluded both clinical investigators and practicing physicians. Cohorts of patients with the same NYHA class according to investigators have quite different prognoses in different trials, despite similar demographics and therapy. Furthermore, physicians often find it difficult to assign patients to one NYHA class. Thus, patients may be assigned to multiple classes (e.g., NYHA class II/III or III/IV).
We believe that primary care physicians will find a less subjective classification of heart failure easier to apply than the NYHA scheme. Therefore, we recommend that they consider using the new classification scheme shown in Table 3.8 We believe that by using the new scheme, physicians will be able to classify individual patients less ambiguously and will find it easier to apply the results of recent trials.
Although this new scheme has not been tested prospectively, we consider it to be practical and valid for the following reasons:
NYHA class I is identical to our description of an “asymptomatic” class.
NYHA class IV is identical to our classification of “symptomatic with dyspnea at rest.”
NYHA class II and III are combined into our classification of “symptomatic.” Physicians frequently classify patients as NYHA class II/III. There is no evidence that patients in NYHA class II and III should be treated differently. It may be helpful to continue to categorize these patients separately in research studies, as the prognosis differs for the two classes.
Our other class, described as “symptomatic with recent history of dyspnea at rest,” is based in part on a classification strategy used for patient recruitment in the Randomized Aldactone Evaluation Study (RALES).11 Based on comparisons between the outcomes of RALES and other mortality trials, patients in this class appear to have a worse prognosis than other symptomatic patients without a history of dyspnea at rest, but a better prognosis than patients who continue to have dyspnea at rest. In addition, therapy based on currently available trial data is distinct for this class of patients. This class most closely correlates to NYHA class IIIb, a subclassification of NYHA class III used by some cardiologists.
The new classification scheme is the basis for the classification of patients in this guideline.
COMMON DIAGNOSTIC STUDIES
Accurate diagnosis of heart failure is important.
Electrocardiography
In most instances, left ventricular systolic dysfunction is caused by ischemic heart disease. Standard 12-lead electrocardiography should be used to determine whether ischemic heart disease is likely, so that appropriate interventions can be initiated. In addition, electrocardiography can provide information about rhythm abnormalities (level of evidence: D).
Assessment of Ejection Fraction
The management of heart failure is based on the clinical presentation, the findings of the physical examination and the determination of systolic dysfunction by measurement of the left ventricular ejection fraction. Documentation of systolic dysfunction is critical to guide therapy.
Transthoracic Echocardiography and Radio-nuclide Ventriculography
Transthoracic (standard surface) echocardiography is noninvasive and provides diagnostic information readily and safely. It gives information about ventricular function, chamber size and shape, wall thickness and valvular function. All such information is helpful in the management of patients with heart failure. Transthoracic echocardiography is inexpensive, reliable and widely available.
Radionuclide ventriculography also may be used to assess left ventricular and right ventricular ejection fractions. Although this modality provides reproducible quantification of the ejection fraction, it does not yield information about valvular function or wall thickness.
Echocardiography should be performed to guide management in patients with a presumed diagnosis of heart failure (level of evidence: D). The results can help to differentiate systolic from diastolic dysfunction and clarify relevant valvular dysfunction, as these disorders may be managed quite differently from systolic dysfunction.
Exercise Stress Testing and Cardiac Catheterization
Exercise stress testing is useful for evaluating active and significant concomitant coronary artery disease, and it may have a role in assessing the degree of cardiac disability. Thus, it may be helpful in the evaluation of some patients with heart failure. The decision to perform exercise stress testing should be individualized (level of evidence: D). Blanket application in all patients is not indicated. Consultation with a cardiologist may be helpful in deciding when and exactly how to perform stress testing in appropriate patients.
Cardiac catheterization is useful in the management of heart failure when the discovery of significant coronary artery disease or valvular heart disease would affect medical treatment or provide the necessary information to proceed to surgery. Coronary artery bypass grafting in multivessel disease with depressed systolic function decreases mortality and significantly improves symptoms of angina.12
The decision to proceed to cardiac catheterization should be determined by the clinical presentation, particular features in the patient, the results of noninvasive tests and a substantial weighing of the risks and benefits of the procedure. The decision to perform cardiac catheterization should be individualized (level of evidence: D) and is best made in collaboration with a cardiologist.
Ambulatory Rhythm Monitors
Sudden death, presumably because of arrhythmias, is a major cause of mortality in patients with heart failure. Over the past few years, studies13–15 have shown that implantable cardioverter-defibrillators provide a major survival advantage in patients who have symptomatic or inducible ventricular arrhythmias and ischemic heart disease, with or without heart failure (level of evidence: A). Ambulatory monitoring should be performed when rhythm disturbance is suspected in a patient with heart failure. If a ventricular arrhythmia is present, the patient should be referred for further evaluation.
Other Studies
Serum levels of atrial natriuretic peptide (ANP), brain natriuretic peptide (BNP), norepinephrine and other neurohormones are elevated in patients with heart failure. ANP and BNP levels may predict prognosis and are currently used in some centers to monitor patients with heart failure.16 However, clinical application of the use of these hormones in the management of heart failure is controversial and is not recommended in primary care at this time (level of evidence: D).
| New classification based on symptoms | Corresponding NYHA class |
|---|---|
| Asymptomatic | NYHA class I |
| Symptomatic | NYHA class II/III |
| Symptomatic with recent history of dyspnea at rest | NYHA class IIIb* |
| Symptomatic with dyspnea at rest | NYHA class IV |
Heart failure also may be caused by conditions such as thyroid disease, alcohol use or myocarditis; in such instances, heart failure may be reversible. These conditions should be considered, but a full discussion is beyond the scope of this article.