Effective pain management in the terminally ill patient requires an understanding of pain control strategies. Ongoing assessment of pain is crucial and can be accomplished using various forms and scales. It is also important to determine if the pain is nociceptive (somatic or visceral pain) or neuropathic (continuous dysesthesias or chronic lancinating or paroxysmal pain). Nociceptive pain can usually be controlled with nonsteroidal anti-inflammatory drugs or corticosteroids, whereas neuropathic pain responds to tricyclic antidepressants or anticonvulsants. Relief of breakthrough pain requires the administration of an immediate-release analgesic medication. If a significant amount of medication for breakthrough pain is already being given, the baseline dose of sustained-release analgesic medication should be increased. If pain does not respond to one analgesic medication, physicians should use an equianalgesic dose chart when changing the medication or route of administration. Opioid rotation can be used if pain can no longer be controlled on a specific regimen. The impact of unresolved psychosocial or spiritual issues on pain management may need to be addressed.
Pain control is one of the most challenging tasks that family physicians face when providing care for patients at the end of life. Despite recent advances in the understanding of pain management, pain is often untreated or undertreated.1 Consequently, a significant number of patients needlessly suffer physical pain and mental distress at the end of life.2,3 The challenge for physicians is to provide aggressive pain management and implement strategies to alleviate suffering in patients with pain that is difficult to control.
Three principles should be followed in providing pain control at the end of life.4 First, pain can be controlled in most patients by following the World Health Organization's step-care approach.5 Second, acute or escalating pain is a medical emergency that requires prompt attention. A delay in responding to this pain makes it more difficult to control. Third, addiction is not an issue in patients with a terminal illness. When pain is treated appropriately, addiction problems are rare.6
Pain Assessment
The initial pain assessment should include information about the location, quality, intensity, onset, duration and frequency of pain, as well as factors that relieve or exacerbate the pain. This information can be organized using a pain assessment form (Figure 1). Pain intensity can be assessed on a 10-point scale, with zero representing no pain and 10 representing the worst pain possible. Other potential assessment tools include a pictorial assessment scale and a visual analog scale (Figure 2).
While it is important to obtain current information on pain intensity, it is also essential to assess worst, least and average pain intensity over the previous 24 hours. If necessary, patients should keep a log that documents the relationship of pain intensity to the time of day. If patients are unable to perform this task, their caregivers should be instructed in how to complete the log.
Patients with a terminal illness should be encouraged to verbalize the pain experience in their own words. Physicians should always believe what patients report about their pain.4
FIGURE 1. Hospice Initial Pain Assessment
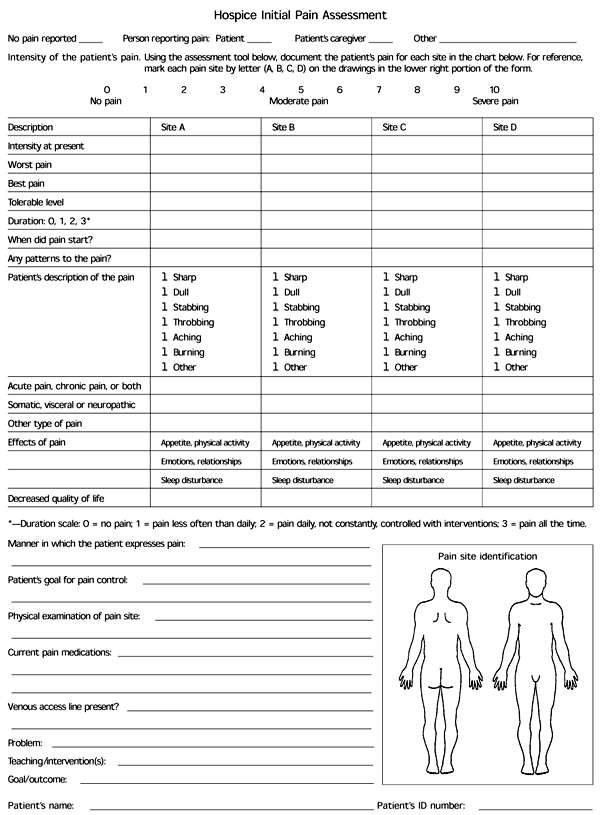
Form used for hospice initial pain assessment.
Adapted with permission from Housecall Medical Resources, Inc., Knoxville, Tenn.
Pain Syndromes and Management
Pain syndromes can be nociceptive (somatic or visceral pain) or neuropathic (continuous dysesthesias or chronic lancinating or paroxysmal pain). With correct identification of the pain syndrome, physicians can provide better pain control using appropriate adjuvant therapies and interventions. It is still important to apply basic pain management principles in terminally ill patients.
NOCICEPTIVE PAIN
Nociceptive pain is the result of actual or potential tissue damage and includes somatic and visceral pain. Somatic pain presents as an aching, throbbing, stabbing and/or pressure sensation, and its source is skin, muscle or bone. Visceral pain presents as a gnawing, cramping, aching, sharp and/or stabbing sensation, and it comes from internal organs.
Somatic Pain
Skin or musculoskeletal pain usually responds to nonsteroidal anti-inflammatory drugs (NSAIDs). Patients who cannot tolerate NSAIDs because of gastrointestinal side effects may be able to tolerate choline magnesium trisalicylate (Trilisate).
Compared with NSAIDs, the new selective cyclooxygenase-2 inhibitors (COX-2), which include celecoxib (Celebrex) and rofecoxib (Vioxx), are associated with less gastromucosal injury and less medication-induced dyspepsia. However, COX-2 inhibitors are more expensive than NSAIDS, and they provide no advantage with regard to analgesic effect.7
Bone pain typically cannot be completely controlled with narcotics. Therefore, adjuvant agents are added to the narcotic regimen.8 First-line adjuvant therapies for bone pain include NSAIDs and corticosteroids such as prednisone (30 to 60 mg per day taken orally), dexamethasone (Decadron; 16 mg per day taken orally) and methylprednisolone (Medrol; 120 mg per day taken orally).
FIGURE 2. Pain Assessment Tools
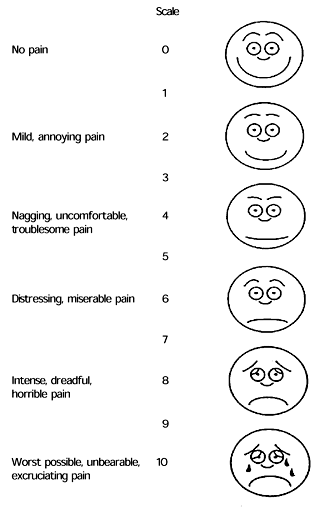
Widely used pain assessment tools: (A) pictorial assessment scale and (B) visual analog scale.
Bisphosphonates, calcitonin-salmon (Calcimar) or palliative radiotherapy may be used as adjuvant treatment in patients whose pain does not respond to NSAIDs or corticosteroids. Bisphosphonates have been shown to reduce pain from bony metastasis.9,10 The bisphosphonate pamidronate (Aredia) has been used in the treatment of bone pain; the currently recommended dosage is 90 mg given intravenously over two to four hours once a month.
Calcitonin is available for intranasal, intramuscular, intravenous or subcutaneous administration. However, this agent is less effective than other adjuvant medications, and it must be taken for several weeks before it becomes effective. Consequently, calcitonin therapy should be reserved for use in patients with refractory bone pain.11
Palliative radiotherapy using injectable radionuclides or an external beam directed at the pain site may be beneficial in patients who do not respond to other adjuvant treatments.12
Visceral Pain
The most common pain syndrome related to the visceral organs is associated with partial or total bowel obstruction. The first step in the treatment of this pain is to reduce the amount of gastrointestinal stimulation by limiting intake to clear liquids and administering antiemetics. If these measures are not successful, an anticholinergic drug may control the pain. Scopolamine, administered subcutaneously or transdermally (Transderm Scop), may alleviate symptoms by reducing peristalsis and secretions. Octreotide (Sandostatin) or corticosteroids may be used in patients with refractory visceral pain.11
NEUROPATHIC PAIN
Neuropathic pain has two distinct types. The first consists of continuous dysesthesias, which are characterized by continuous burning, electrical sensations or other abnormal sensations. The second is chronic lancinating or paroxysmal pain, which is described as a sharp, stabbing, shooting, knifelike pain, often with a sudden onset.11
Continuous Dysesthesias
Tricyclic antidepressants are currently recommended as first-line treatment for continuous dysesthesia–type pain (Table 1).11,13 Compared with other tricyclic antidepressants, amitriptyline (Elavil), doxepin (Sinequan) and imipramine (Tofranil) have greater analgesic properties, whereas clomipramine (Anafranil), desipramine (Norpramin) and nortriptyline (Pamelor) have fewer side effects.14
Systemic local anesthetics may be useful in patients who cannot tolerate tricyclic antidepressants or cannot take those medications because of cardiac disease (Table 1).11,13 In patients with refractory pain, orally or transdermally administered clonidine (Catapres, Catapres-TTS) can be used as an adjuvant to opioid therapy.11
Lancinating or Paroxysmal Pain
Anticonvulsants are first-line options for the treatment of lancinating or paroxysmal neuropathic pain11 (Table 2).13 Patients with trigeminal neuralgia have been shown to respond well to carbamazepine (Tegretol).14 Because carbamazepine has potential bone marrow side effects, other anticonvulsants are better options in patients with a history of bone marrow suppression.14
Gabapentin (Neurontin) has advantages over other anticonvulsants because of its safety profile and lack of drug interactions.15 In addition, no monitoring of plasma concentration is required. Baclofen (Lioresal) can be used as a first-line drug in the treatment of lancinating or paroxysmal neuropathic pain.11 In patients with refractory pain, oral local anesthetics, tricyclic antidepressants, clonidine and topical capsaicin 0.025 percent (Zostrix) may be used.11
Special Situations
BREAKTHROUGH PAIN
Sustained-release preparations for pain control have provided physicians with multiple therapeutic options. However, breakthrough pain can be expected to occur when a sustained-release analgesic medication becomes less effective in controlling pain. In one study,16 hospice patients experienced an average of about three episodes of breakthrough pain per day, with the pain having a mean intensity of 7 on a 10-point scale.
Because of the high incidence of breakthrough pain, it is important for patients to have immediate-release analgesic medication available. Each dose of this medication is usually 10 to 30 percent of the total daily dose of sustained-release analgesic medication. For example, the patient who is receiving 60 mg of sustained-released morphine twice daily should have 15-mg doses (range: 12 to 36 mg) of immediate-release morphine available to manage breakthrough pain. The frequency of use can be 15 mg every hour until pain is controlled, or more frequently than every hour if a health care professional is present while the patient is taking the doses.
In patients who are using a significant amount of medication for breakthrough pain, the increase in the dose of sustained-release analgesic medication should reflect the total breakthrough dose taken in 24 hours. One strategy for this increase is to use 25 percent of the total dose of immediate-release medication when slight reduction of pain is needed, 50 percent when moderate reduction is needed and 100 percent when severe reduction is needed.17 For instance, a patient reports taking a total of 60 mg of immediate-release morphine for breakthrough pain in the past 24 hours. The assessment reveals that the patient is in moderate pain, so the baseline dose of sustained-release morphine, when given twice daily, should be increased by 15 mg per dose (for a total increase of 30 mg per day). When the baseline dose of sustained-released analgesic medication is increased, the dose of the immediate-release medication also needs to be increased accordingly.
TABLE 2 Adjuvant Therapy for Lancinating or Paroxysmal Neuropathic Pain
| Medication | Starting dosage* | Maximum per day* |
|---|---|---|
| Carbamazepine (Tegretol)† | 200 mg twice daily | 1.6 g |
| Clonazepam (Klonopin)‡ | 0.5 mg three times daily | 20 mg |
| Divalproex (Depakote)‡ | 10 mg per kg per day | 60 mg per kg |
| Gabapentin (Neurontin)§ | 100 mg three times daily | 3.6 g |
| Lamotrigine (Lamictal)† | 50 mg once daily | 500 mg |
| Phenytoin (Dilantin)‡ | 100 mg three times daily | 600 mg |
| Baclofen (Lioresal)§ | 5 mg three times daily | 80 mg |
*—Reduce dosage in children and the elderly.
†—Reduce dosage in patients with hepatic and/or renal impairment.
‡—Reduce dosage in patients with hepatic impairment.
§—Reduce dosage in patients with renal impairment.
Product information from Physicians' desk reference. 54th ed. Montvale, N.J.: Medical Economics, 2000.
ACUTE OR CRESCENDO PAIN
A variant of breakthrough pain occurs in patients who are under good pain control but develop acute onset of new pain or crescendo of established pain. Pain management in these patients should be considered a medical emergency. Acute or crescendo pain can be controlled with the aggressive use of breakthrough pain medication (e.g., immediate-release morphine) every 15 minutes until the patient is comfortable. This medication should be taken in the presence of a health care professional who understands pain management. Once the crisis has resolved, the baseline sustained-release analgesic medication should be adjusted in an attempt to prevent the recurrence of acute or crescendo pain. However, this pain persists in some patients, and arrangements must then be made to provide parenterally administered pain medications.
INCIDENT PAIN
Incident pain is another type of pain that patients may experience at the end of life. This pain occurs in conjunction with certain activities, such as rolling over in bed, riding in a car or being bathed. Incident pain can be managed by giving patients their breakthrough dose of immediate-release medication 30 minutes before the activity is performed. Premedication can reduce the amount of pain that occurs during the activity. It is also important to assess patients for underlying causes of the pain, and to correct those causes if possible.
CHANGING MEDICATIONS AND ROUTES
One of the most difficult challenges physicians face in providing pain management at the end of life is how to change from one medication to another, from one route of administration to another, or both. An equianalgesic dose chart can facilitate these changes (Table 3).18 The conversions in the chart are based on studies of the effects of single doses of different pain medications, usually in comparison with the effects of 10 mg of parenterally administered morphine.
In using the equianalgesic chart to change from one route of administration to another, for example, equianalgesic doses of morphine are 10 mg for intravenous administration and 30 mg for oral administration. Simply put, the equivalence of intravenously administered to orally administered morphine is 1 to 3. Hence, the patient who has been receiving 60 mg per day of intravenously administered morphine would be given 180 mg per day of orally administered morphine.
In some instances, both the medication and the route of administration must be changed. For example, a patient who has been controlled on 30 mg per day of orally administered hydromorphone (Dilaudid) progresses to the point where the oral route is no longer tolerated. The decision is made to change to a continuous infusion of morphine. The equianalgesic dose chart shows that 7.5 mg of orally administered hydromorphone is equivalent to 10 mg of parenterally administered morphine. Therefore, the total daily dose of intravenously administered morphine would be 40 mg, or 1.7 mg per hour.
The fentanyl patch (Duragesic) can provide pain control in patients who are unable to swallow medications. Because no parenteral access is required, the patch can be applied by the patient, a family member or other caregiver. In one study,19 the fentanyl patch provided pain control equivalent to that achieved with subcutaneously administered morphine. In another study,20 patient compliance with the patch and acceptance of the treatment were excellent, and side effects were similar to those with other opioids. In the recommended conversion, 1 mg of the fentanyl patch is equivalent to 2 mg per day of orally administered morphine. Thus, a patient who has been taking 50 mg per day of orally administered morphine would start with a 25-mg fentanyl patch. Because the patch takes approximately 12 hours to become effective, the morphine should be continued during that period. The fentanyl patch is difficult to titrate, and a steady state may not be achieved for three to four days. Consequently, some form of immediate-release analgesic medication should be available to manage breakthrough pain.
OPIOID TOLERANCE
In most terminally ill patients, escalating pain is related to progression of their disease. Tolerance of orally or parenterally administered opioids develops in some of these patients. When pain is no longer controlled on a specific regimen, opioid rotation is a possible solution. Opioid rotation includes changing to a different medication using the same route of administration, maintaining the current medication but changing the route of administration, or changing both the medication and the route of administration.21
If opioid rotation does not control pain, epidural analgesia is an effective option. The equivalence conversion for parentally to epidurally administered morphine is 3 to 1.18
COGNITIVELY IMPAIRED PATIENTS
The assessment of pain is based on the patient's description of the pain and rating of its intensity. Patients who, for whatever reason, are cognitively impaired have lost some of their ability to communicate. As a result, their pain tends to be undertreated.22–24 Because of their pain, these patients are more likely to become aggressive.24
The assumption that cognitively impaired patients cannot use a pain scale is inaccurate. In one study,25 65 percent of patients with significant cognitive impairment were able to use a word-anchored pain scale (i.e., with zero indicating no pain and 5 indicating the worst pain ever felt). The patients responded best when they were given sufficient time to process information. In addition, nonverbal signs of pain, such as a furrowed brow, agitation or moaning, are excellent pain indicators in patients who are unable to articulate their pain level.
PSYCHOSOCIAL OR SPIRITUAL PAIN
Successful pain management requires a holistic approach to the broad spectrum of problems in patients who are at the end of life.26 When providing pain management to these patients, physicians need to recognize the impact that unresolved psychosocial and spiritual issues can have on pain management. A multidiscipline hospice team can provide support for terminally ill patients.
UNCONTROLLED PAIN
Despite receiving the best treatment family physicians can provide, some terminally ill patients continue to have pain. In these instances, a pain subspecialist can provide advanced pain management options such as epidural catheters and nerve blocks.

