Thyroid disease affects up to 0.5 percent of the population of the United States. Its prevalence is higher in women and the elderly. The management of hypothyroidism focuses on ensuring that patients receive appropriate thyroid hormone replacement therapy and monitoring their response. Hormone replacement should be initiated in a low dosage, especially in the elderly and in patients prone to cardiac problems. The dosage should be increased gradually, and laboratory values should be monitored six to eight weeks after any dosage change. Once a stable dosage is achieved, annual monitoring of the thyroid-stimulating hormone (TSH) level is probably unnecessary, except in older patients. After full replacement of thyroxine (T4) using levothyroxine, the addition of triiodothyronine (T3) in a low dosage may be beneficial in some patients who continue to have mood or memory problems. The management of patients with subclinical hypothyroidism (a high TSH in the presence of normal free T4 and T3 levels) remains controversial. In these patients, physicians should weigh the benefits of replacement (e.g., improved cardiac function) against problems that can accompany the excessive use of levothyroxine (e.g., osteoporosis).
Hypothyroidism is second only to diabetes mellitus as the most common endocrine disorder in the United States, and its prevalence may be as high as 18 cases per 1,000 persons in the general population.1 The disorder becomes increasingly common with advancing age, affecting about 2 to 3 percent of older women.2 Because hypothyroidism is so common, family physicians need to know how to diagnose the disorder and select appropriate thyroid hormone replacement therapy.
Etiology
A number of conditions can lead to hypothyroidism (Table 1).3 Of noniatrogenic causes, Hashimoto's thyroiditis, or chronic lymphocytic thyroiditis, is the most common inflammatory thyroid disorder and the most frequent cause of goiter in the United States.4 For an unknown reason, the prevalence of Hashimoto's thyroiditis has been increasing dramatically in this country over the past 50 years.5 Other common causes of hypothyroidism include irradiation of the thyroid subsequent to Graves' disease and surgical removal of the thyroid gland.
Long-term thyroid dysfunction after subacute granulomatous thyroiditis (de Quervain's thyroiditis) or subacute lymphocytic thyroiditis (silent or painless thyroiditis) is fairly rare. Full thyroid function is regained in 90 percent of patients with these conditions.6
Hypothyroidism can also develop secondary to hypothalamic and pituitary disorders. These endocrine conditions occur primarily in patients who have undergone intracranial irradiation or surgical removal of a pituitary adenoma.
Signs and Symptoms
The signs and symptoms of hypothyroidism are nonspecific and may be confused with those of other clinical conditions, especially in postpartum women and the elderly. Because of the variety of possible manifestations, family physicians must maintain a high index of suspicion for the disorder, especially in high-risk groups.
Patients with severe hypothyroidism generally present with a constellation of signs and symptoms that may include lethargy, weight gain, hair loss, dry skin, forgetfulness, constipation and depression. Not all of these signs and symptoms occur in every patient, and many may be blunted in patients with mild hypothyroidism. The most common manifestations of hypothyroidism are listed in Table 2.7
TABLE 1 Causes of Hypothyroidism
| Primary hypothyroidism (95% of cases) |
| Idiopathic hypothyroidism* |
| Hashimoto's thyroiditis |
| Irradiation of the thyroid subsequent to Graves' disease |
| Surgical removal of the thyroid |
| Late-stage invasive fibrous thyroiditis |
| Iodine deficiency |
| Drug therapy (e.g., lithium, interferon) |
| Infiltrative diseases (e.g., sarcoidosis, amyloidosis, scleroderma, hemochromatosis) |
| Secondary hypothyroidism (5% of cases) |
| Pituitary or hypothalamic neoplasms |
| Congenital hypopituitarism |
| Pituitary necrosis (Sheehan's syndrome) |
*— Probably old Hashimoto's thyroiditis (i.e., a “burned out” thyroid from Hashimoto's thyroiditis).
Adapted with permission from Hueston WJ. Thyroid disease. In: Rosenfeld JA, Alley N, Acheson LS, Admire JB, eds. Women's health in primary care. Baltimore: Williams & Wilkins, 1997:617–31.
In older patients, hypothyroidism can be confused with Alzheimer's disease and other conditions that cause cognitive impairment. Because depression can be a manifestation of hypothyroidism, patients with this endocrine condition may be treated as depressed, and other signs and symptoms of the disorder may be overlooked. This is particularly true with hypothyroidism that develops or worsens during pregnancy, or with postpartum thyroiditis, which has many of the same symptoms as postpartum depression.
TABLE 2 Common Signs and Symptoms of Hypothyroidism*
| Sign or symptom | Affected patients (%) |
|---|---|
| Weakness | 99 |
| Skin changes (dry or coarse skin) | 97 |
| Lethargy | 91 |
| Slow speech | 91 |
| Eyelid edema | 90 |
| Cold sensation | 89 |
| Decreased sweating | 89 |
| Cold skin | 83 |
| Thick tongue | 82 |
| Facial edema | 79 |
| Coarse hair | 76 |
| Skin pallor | 67 |
| Forgetfulness | 66 |
| Constipation | 61 |
*— Only signs and symptoms that occur in 60 percent or more of patients with hypothyroidism are listed in this table.
Adapted with permission from Larsen PR, Davies TF, Hay ID. The thyroid gland. In: Wilson JD, Foster DW, Kronenberg HM, Larsen PR, eds. Williams Textbook of endocrinology. 9th ed. Philadelphia: Saunders, 1998:461.
Physical findings in patients with hypothyroidism are also nonspecific. These findings can include lowered blood pressure with bradycardia, nonpitting edema, generalized hair loss (especially along the outer third of the eyebrows), dry skin and a diminished relaxation phase of reflexes.
A primary challenge is to differentiate the generalized symptoms of early hypothyroidism from the similar symptoms of fatigue and depression that occur in many other conditions.
Diagnosis
The evaluation of patients with new-onset hypothyroidism is quite limited. In patients with primary hypothyroidism, the thyroid-stimulating hormone (TSH) level is elevated, indicating that thyroid hormone production is insufficient to meet metabolic demands, and free thyroid hormone levels are depressed. In contrast, patients with secondary hypothyroidism have a low or undetectable TSH level.
TSH results have to be interpreted in light of the patient's clinical condition. A low TSH level should not be misinterpreted as hyperthyroidism in the patient with clinical manifestations of hypothyroidism. When symptoms are nonspecific, a follow-up assessment of the free thyroxine (T4) level can help distinguish between primary and secondary hypothyroidism. A guide to the laboratory diagnosis of hypothyroidism and the interpretation of TSH, T4 and triiodothyronine (T3) levels is provided in Table 3.
Once the diagnosis of primary hypothyroidism is made, additional imaging or serologic testing is unnecessary if the thyroid gland is normal on examination. In patients with secondary hypothyroidism, further investigation with provocative testing of the pituitary gland can be performed to determine if the underlying cause is a hypothalamic or pituitary disorder. In patients with pituitary dysfunction, imaging is indicated to detect microadenomas, and levels of other hormones that depend on pituitary stimulation should also be measured. In general, evidence of decreased production of more than one pituitary hormone is indicative of panhy-popituitary problems.
Thyroid Hormone Replacement
SELECTING THE APPROPRIATE AGENT
Thyroid medications were once prepared from desiccated samples of ground thyroid glands from cows, and standardization was based on the iodine content of the extract rather than its T3 or T4 content. The actual thyroid hormone content of the products varied considerably from manufacturer to manufacturer, and even within products from the same manufacturer, depending on the thyroid status of the cows. Fortunately, this method of preparing thyroid hormone has been abandoned, and replacement is now accomplished primarily with synthetic thyroid hormones.
TABLE 3 Laboratory Values in Hypothyroidism
| TSH level | Free T4 level | Free T3 level | Likely diagnosis |
|---|---|---|---|
| High | Low | Low | Primary hypothyroidism |
| High (>10 μU per mL [10 mU per L]) | Normal | Normal | Subclinical hypothyroidism with high risk for future development of overt hypothyroidism |
| High (6 to 10 μU per mL [6 to 10 mU per L]) | Normal | Normal | Subclinical hypothyroidism with low risk for future development of overt hypothyroidism |
| High | High | Low | Congenital absence of T4-T3–converting enzyme; amiodarone (Cordarone) effect on T4-T3 conversion |
| High | High | High | Peripheral thyroid hormone resistance |
| Low | Low | Low | Pituitary thyroid deficiency or recent withdrawal of thyroxine after excessive replacement therapy |
TSH = thyroid-stimulating hormone; T4 = thyroxine; T3 = triiodothyronine.
A recent analysis8 of four levothyroxine preparations, including two brand-name products (Synthroid and Levoxyl) and two generic preparations, demonstrated relative bioequivalence. Patients switched from any one of the four preparations to another showed insignificant variations in their thyroid function tests. Among the four products, the only difference noted was that Synthroid produced a more rapid and higher rise in the T3 level after administration. However, the difference was not statistically significant and is of questionable clinical importance.
INITIATING TREATMENT
Most otherwise healthy adult patients with hypothyroidism require thyroid hormone replacement in a dosage of 1.7 μg per kg per day, with requirements falling to 1 μg per kg per day in the elderly. Thus, levothyroxine in a dosage of 0.10 to 0.15 mg per day is needed to achieve euthyroid status. For full replacement, children may require up to 4 μg per kg per day.9
In young patients without risk factors for cardiovascular disease, thyroid hormone replacement can start close to the target goal. [ corrected] In most healthy young adults, replacement is initiated using levothyroxine in a dosage of 0.075 mg per day, with the dosage increased slowly as indicated by continued elevation of the TSH level.
Levothyroxine should be initiated in a low dosage in older patients and those at risk for the cardiovascular compromise that could occur with a rapid increase in resting heart rate and blood pressure.9 In these patients, the usual starting dosage is 0.025 mg per day. This dosage can be increased in increments of 0.025 to 0.050 mg every four to six weeks until the TSH level returns to normal.
Thyroid hormone is usually given once daily, but some evidence suggests that weekly dosing may also be effective. In a small study10 of 12 patients with hypothyroidism, a bolus dose of thyroid hormone equal to seven times the usual daily dose was well tolerated. Before weekly replacement can be recommended, however, more investigation is required, including definitions of the populations in which this approach is indicated.
In a study11 of 33 middle-aged patients (mostly women) with stable hypothyroidism who were already receiving levothyroxine, small improvements in mood, memory and cold tolerance occurred after triiodothyronine was added, in a dosage of 0.0125 mg per day, with a concomitant 0.05-mg decrease in the usual levothyroxine dosage. Although this study was small, it suggests that some patients who are chemically euthyroid but have lingering neuropsychiatric problems might benefit from triiodothyronine. Further investigation is required to determine the role of triiodothyronine in these patients, as well as the long-term consequences of its use.
MONITORING THYROID FUNCTION
In patients with an intact hypothalamic-pituitary axis, the adequacy of thyroid hormone replacement can be followed with serial TSH assessments. However, changes in the TSH level lag behind serum thyroid hormone levels. Thus, the TSH level should be evaluated no earlier than four weeks after an adjustment in the levothyroxine dosage. The full effects of thyroid hormone replacement on the TSH level may not become apparent until after eight weeks of therapy.12
In patients with pituitary insufficiency, measurements of free T4 and T3 levels can be performed to determine whether patients remain euthyroid. In these patients, the goal is to maintain free thyroid hormone levels in the middle to upper ranges of normal to ensure adequate replacement.
TSH or free T4 levels are monitored annually in most patients with hypothyroidism, although no data support this practice. Generally, once a stable maintenance dosage of levothyroxine is achieved, that dosage will remain adequate until patients are 60 to 70 years of age. With age, thyroid binding may decrease, and the serum albumin level may decline. In this setting, the levothyroxine dosage may need to be reduced by up to 20 percent.13,14 Although less frequent than annual monitoring could be justified in younger adult patients whose weight is stable, annual monitoring in older patients is necessary to avoid overreplacement.15
A guideline for initiating and monitoring thyroid hormone replacement therapy is provided in Figure 1.
INTRAVENOUS REPLACEMENT
Because thyroid hormone has a large volume of distribution and long half-life, parenteral replacement is unnecessary in patients who are unable to take medication orally for a few days to a week. However, some patients may be unable to take oral medications for much longer periods. Intravenous administration is advised in these patients and in those who need to begin thyroid hormone replacement but cannot take oral medications. Only about 70 to 80 percent of an oral dose of replacement medication is absorbed. Therefore, parenteral replacement should be initiated at 70 to 80 percent of the usual oral dose.16
SUBCLINICAL HYPOTHYROIDISM
The TSH level can be mildly elevated when the free T4 and T3 levels are normal, a situation that occurs most often in women and becomes increasingly common with advancing age. This condition has been termed “subclinical hypothyroidism,” based on the supposition that it reflects early failure of the thyroid hormone and eventual development of hypothyroidism.17 However, it appears that patients with a TSH level between 6 and 10 μU per mL(6 to 10 mU per L) are not at risk for subsequent hypothyroidism.1 In contrast, patients with a higher TSH level (above 10 μU per mL) progress to overt hypothyroidism at a rate of 1 to 20 percent per year.1
FIGURE 1. Treatment of Hypothyroidism
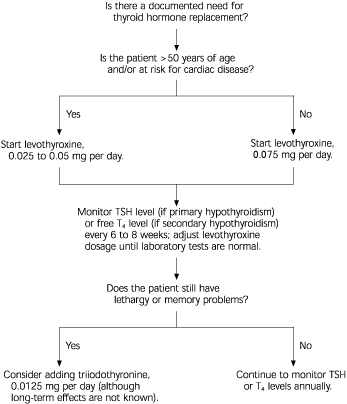
Initiation and monitoring of treatment for hypothyroidism. (TSH = thyroid-stimulating hormone; T4 = thyroxine)
Thyroid hormone replacement may have some benefits in patients with subclinical hypothyroidism, but there is also a potential for adverse effects, particularly in older patients. Some studies have shown that supplementation of thyroid hormone accelerates bone mineral loss in older women with subclinical hypothyroidism, and that estrogen replacement therapy does not counteract this effect.17 Bone-wasting effects have not been observed in patients who are clinically hypothyroid and receive adequate thyroid hormone replacement therapy.18
Thyroid hormone replacement has also been reported to decrease serum homocysteine levels.19 Along with changes in lipids, hyperhomocysteinemia may be one of the mechanisms through which hypothyroidism is associated with an increased risk for cardiovascular disease.20
At this time, the approach to patients with subclinical hypothyroidism must be individualized. In patients at higher risk for osteoporosis or fractures, the deleterious effects of excessive thyroid hormone can be avoided by withholding replacement until the free T4 and T3 levels drop below normal. In patients with hyperhomocysteinemia, existing cardiac disease or risk factors for heart disease, early thyroid hormone replacement may offer more advantages. Right now, consensus is lacking on how to manage patients with subclinical hypothyroidism. More research is needed to sort out the most appropriate management.
Conditions Affecting Thyroid Hormone Replacement Requirements
Because thyroid hormone is highly protein bound, medical conditions that alter the amount of binding hormones and drugs that compete for binding may change the amount of available free thyroid hormone. The thyroid replacement dosage must be changed in response to alterations in binding status.
With conditions that cause an increase in serum binding proteins, such as high estrogen states (e.g., pregnancy), oral contraceptive use or postmenopausal estrogen replacement, the dosage of levothyroxine must be increased. In contrast, androgens decrease levels of thyroid binding proteins, necessitating a reduction in the dosage. Older patients also have lower serum protein levels and may require reductions in their maintenance dosage over time. Nephrosis, protein-losing enteropathies and cirrhosis are other conditions that require a reduced thyroid hormone dosage.
A number of medications reduce the absorption of thyroid hormone from the intestines, necessitating an increase in the replacement dosage (Table 4).21 Other drugs accelerate the metabolism of thyroid hormone, and an increase in the replacement dosage is then required. When these medications are started or adjusted, the TSH value should be monitored to determine whether additional thyroid hormone replacement is indicated.
Persistently Elevated TSH Despite Thyroid Hormone Replacement
Poor compliance is the most common reason for continued elevation of the TSH level in patients receiving presumably adequate thyroid hormone replacement. Patients who do not regularly take their replacement medication and then try to “catch up” just before a physician visit may restore their free T4 levels to normal but continue to have an elevated TSH level.
Very rarely, patients have tissue-level unresponsiveness to thyroid hormone. This condition reflects a mutation in the gene that controls a receptor for T3, rendering it unable to bind with the hormone. The genetic mutation has been identified in only 300 families.22 In these patients, adequate amounts of thyroid hormone are produced but are ineffective. Consequently, the TSH level remains elevated, and the patients continue to have symptoms of hypothyroidism. These patients should be referred to an endocrinologist for further evaluation and management.
TABLE 4 Drugs Potentially Altering Thyroid Hormone Replacement Requirements
| Increase replacement requirements | |
| Drugs that reduce thyroid hormone production | |
| Lithium | |
| Iodine-containing medications | |
| Amiodarone (Cordarone) | |
| Drugs that reduce thyroid hormone absorption | |
| Sucralfate (Carafate) | |
| Ferrous sulfate (Slow Fe) | |
| Cholestyramine (Questran) | |
| Colestipol (Colestid) | |
| Aluminum-containing antacids | |
| Calcium products | |
| Drugs that increase metabolism of thyroxine | |
| Rifampin (Rifadin) | |
| Phenobarbital | |
| Carbamazepine (Tegretol) | |
| Warfarin (Coumadin) | |
| Oral hypoglycemic agents | |
| Increase thyroxine availability and may decrease replacement requirements | |
| Drugs that displace thyroid hormone from protein binding | |
| Furosemide (Lasix) | |
| Mefenamic acid (Ponstel) | |
| Salicylates | |
Adapted with permission from Surks MI, Sievert R. Drugs and thyroid function. N Engl J Med 1995; 333:1688–94.
Screening for Hypothyroidism
The U.S. Preventive Services Task Force23 does not recommend routine screening for hypothyroidism in asymptomatic persons. Recently, some expert panels24 noted that screening may be beneficial in high-risk populations such as elderly women. However, widespread screening is not likely to be cost-effective. Because of the nonspecific symptoms of hypothyroidism, many patients would be tested because of their symptoms. This practice should not be confused with asymptomatic screening.

