
Am Fam Physician. 2002;65(2):217-227
Patient information handouts on preventing heart disease in women and lowering cholesterol levels, written by the author of this article, are provided on the AFP Web site.
Coronary heart disease, the leading cause of death in women, is largely preventable. Lifestyle modifications (e.g., diet and exercise) are the cornerstone of primary and secondary prevention. Elevated levels of low-density lipoprotein cholesterol and triglycerides and low levels of high-density lipoprotein cholesterol are significant risk factors for coronary heart disease. Abundant data show inadequate utilization of lipid-lowering therapy in women. Even when women are given lipid-lowering agents, target levels often are not achieved. Recent guidelines from the American Heart Association and the American College of Cardiology encourage a more aggressive approach to lipid lowering in women. The National Cholesterol Education Program Adult Treatment Panel III also supports this strategy and significantly expands the number of women who qualify for intervention.
Cardiovascular disease is the leading cause of death among men and women in the United States, with most deaths resulting from coronary heart disease. Although women become at risk for death from coronary heart disease approximately 10 years later than men, each year more women than men die from cardiovascular disease.1 Currently, cardiovascular disease kills more women than the next 14 causes of death combined. Further, the mortality rate for coronary heart disease is higher in black women than white women.1
Although cardiovascular disease in women is known to be a major threat to life, women are still more afraid of breast cancer. In a recent survey conducted by the American Heart Association (AHA),2 only 8 percent of women cited heart disease as their leading health threat, compared with more than 50 percent who cited cancer. In reality, however, only one in 28 women will die of breast cancer, whereas one in two women will die of cardiovascular disease.1 The gap between the perceived and actual risk of heart disease in women may impede the adoption of effective preventive strategies.
Substantial data support aggressive risk factor management in both the primary and secondary prevention of cardiovascular disease. In women for whom treatment is indicated, established therapies directed at risk reduction need to be more widely implemented in the primary care setting.
Gender Differences in Risk Factors
The identification of risk factors and comorbid conditions helps to determine the aggressiveness of lipid-lowering therapy and the type of treatment that is required. Many cardiovascular risk factors are similar in men and women. However, gender-specific differences in risk factors exist for women, particularly in relation to diabetes mellitus, isolated hypertension, and dyslipidemia.7 Premature menopausal status also further increases the risk of cardiovascular disease in women.
CHOLESTEROL AND TRIGLYCERIDES
A high blood cholesterol level, specifically an elevated low-density lipoprotein (LDL) cholesterol level, is a significant risk factor for coronary heart disease in both men and women.8,9 The National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) classification system highlights the graded risk for coronary heart disease over a broad range of cholesterol levels (Table 1).9 LDL cholesterol remains the primary target of therapy; however, treatment beyond LDL cholesterol lowering is recommended for men and women with triglyceride levels at or above 200 mg per dL (2.26 mmol per L).
A low high-density lipoprotein (HDL) cholesterol level (at or below 40 mg per dL [1.05 mmol per L]) is a strong predictor of coronary heart disease mortality in women older than 65 years, compared with men of the same age.10 Elevated triglyceride levels may also be a significant risk factor in women, particularly elderly women.
HYPERTENSION
High blood pressure (above 140/90 mm Hg) is present in 52 percent of women older than 45 years, and approximately 30 percent of women older than 65 years have isolated systolic hypertension.7,11 Because hypertension increases the risk of both coronary heart disease and stroke in these women, monitoring and treatment are required.
DIABETES MELLITUS
The risk of coronary heart disease is three to seven times higher in women with diabetes mellitus than in those without the disease.7 By comparison, diabetes is associated with a two to three times increased risk of heart disease in men. The higher risk in women is probably caused by a more deleterious effect of diabetes on lipids and blood pressure.12
SMOKING AND ORAL CONTRACEPTION
Missed Opportunities for Prevention in Women
More than 50 clinical studies have shown that lowering the total cholesterol level or the LDL cholesterol level reduces morbidity and mortality from coronary heart disease.8,15 Although the benefit of cholesterol management is supported by more than two decades of research, data from the third National Health and Nutrition Examination Survey (NHANES III) indicated that more women (51.5 million) than men (46.6 million) had total blood cholesterol levels of 200 mg per dL (5.15 mmol per L).16 NHANES III also found that 42 percent of women had LDL cholesterol levels of 130 mg per dL (3.35 mmol per L) or higher. However, in the Lipid Treatment Assessment Project (L-TAP),17 only 39 percent of female patients treated with lipid-lowering agents achieved target cholesterol levels based on the NCEP ATP II classification.8
Screening for Coronary Heart Disease
CHOLESTEROL LEVELS
The NCEP ATP III guidelines recommend that a fasting lipoprotein profile be obtained at least once every five years in adults age 20 and over. More frequent measurements are required for persons with multiple risk factors for coronary heart disease (Table 1).9 Compared with the NCEP, the American College of Physicians,18 the U.S. Preventive Services Task Force19 and the Joint Task Force of European and Other Societies on Coronary Prevention20 suggest more conservative (i.e., less frequent) screening and treatment guidelines, particularly in the setting of primary prevention; however, these guidelines were developed more than five years ago, before the results of primary prevention trials were published.
European treatment guidelines recommend initiating secondary prevention measures when the probability of developing coronary heart disease reaches 20 percent per decade.20 However, the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/Tex-CAPS)21 demonstrated that patients who were treated with “statins” (3-hydroxy-3-methy-glutaryl coenzyme A reductase inhibitors) and whose average baseline absolute risk was below 20 percent per decade had a 40 percent reduction in their risk for acute coronary events.
HISTORY
In women with hyperlipidemia, the screening examination should begin with a detailed medical history that focuses on lifestyle factors and medical conditions (e.g., diabetes mellitus) that increase the risk of coronary heart disease. In particular, women 65 years and older should be screened for a low HDL cholesterol level, high triglyceride levels, and the presence of isolated systolic hypertension. These risk factors have been shown to be particularly important in older women.14 Because numerous medications have lipid-altering effects, the use of prescription and over-the-counter medications should be documented.22 Selected causes of secondary hyperlipidemia are listed in Table 2.23
PHYSICAL EXAMINATION
The physical examination should assess cardiovascular status, including documentation of peripheral pulses, the presence of bruits, and measurements of blood pressure, height, weight, and abdominal girth. In women, an abdominal circumference greater than 88 cm (35 in) should be cause for concern,14 with European guidelines suggesting an even lower threshold (80 cm [32 in]).20
LABORATORY TESTS
To help rule out secondary causes of dyslipidemia, the laboratory evaluation should include fasting blood glucose level, assessment of liver, thyroid and endocrine function, and urinalysis. In patients with an identifiable cause of secondary dyslipidemia (e.g., diabetes mellitus, renal disease, hypothyroidism, obstructive liver disease), it is necessary to treat the underlying disorder or eliminate the causative agent (e.g., thiazide diuretics). However, secondary causes of dyslipidemia are most often related to lifestyle and include dietary factors (i.e., consumption of foods that are high in fat) and cigarette smoking. In these instances, a counseling-based lifestyle modification program should be implemented.
Lifestyle Modifications
Dietary approaches to lipid management are comprehensively described in the NCEP guidelines.8,9 In summary, women should limit their fat intake (or calories from fat) to 25 to 35 percent of total daily intake (with saturated fat intake limited to less than 7 percent) and their cholesterol intake to less than 200 mg per day. Intake of trans fatty acids should also be kept at a low level.
Weight loss and exercise (30 minutes or more of moderate-intensity physical activity on most days of the week) are essential components of lipid management24 because they correlate with decreased triglyceride levels, increased HDL cholesterol levels, decreased blood pressure, and decreased risk for the development of diabetes.8,9 In postmenopausal women with an LDL cholesterol level between 125 and 210 mg per dL (3.25 to 5.45 mmol per L) and an HDL cholesterol level of less than 59 mg per dL (1.55 mmol per L), even a fat- and cholesterol-restricted diet may be ineffective in improving lipid profiles in the absence of regular aerobic exercise.25
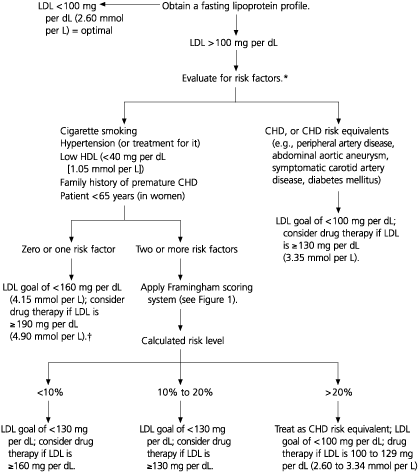
For primary prevention of coronary heart disease, the NCEP ATP III9 recommends an adequate trial of diet therapy (approximately six to 12 weeks) before drug therapy is initiated. In women with two or more risk factors for coronary heart disease and a 10-year risk of 10 to 20 percent, drug therapy should be considered when the LDL cholesterol level is 130 mg per dL or higher (Figures 1 and 2).9 In women with a 10-year risk of less than 10 percent, drug therapy is not necessary unless the LDL cholesterol level is 160 mg per dL (4.15 mmol per L) or greater. In women with established coronary heart disease and LDL cholesterol levels at or above 130 mg per dL, diet and drug therapy should be initiated immediately; adding drug therapy to lifestyle modifications may be considered in these women if their LDL cholesterol level is between 100 and 129 mg per dL (2.60 to 3.34 mmol per L). Recently the results of the MRC/BHF Heart Protection Study26 were released, showing that treatment with simvastatin in a dosage of 40 mg daily was, compared with placebo, associated with a 24 percent reduction in major vascular events among persons at high risk for a coronary event even if LDL cholesterol was less than 130 mg per dL. These data suggest that current guidelines are conservative.
In clinical practice, lipid-lowering goals can generally be achieved when lifestyle modifications are combined with appropriate pharmacotherapy.9
Pharmacologic Therapy
The selection of a lipid-lowering agent in an individual patient depends on the lipids targeted for altering and the degree of intervention needed. Efficacy, side effect profile, and cost also should be considered.
STATINS
The well-established benefits of statin therapy include reduced morbidity and mortality from coronary heart disease, decreased progression of atherosclerosis (and the development of fewer new lesions), regression of atherosclerotic lesions, and decreased coronary artery revascularization.
Statins also have lipid-independent effects that may account for the dramatic and early risk reductions observed in clinical trials. These beneficial effects include plaque stabilization, improvement of coronary endothelial function, inhibition of platelet thrombus formation, and anti-inflammatory activity.27
The currently approved statins include atorvastatin (Lipitor), fluvastatin (Lescol), lovastatin (Mevacor), pravastatin (Pravachol), and simvastatin (Zocor). These drugs have been shown to lower LDL cholesterol levels by 22 to 60 percent.28
Results of four pivotal randomized, placebo-controlled trials of statin therapy21,29–31 (three in secondary prevention, one in primary prevention) have shown that the benefit of statins in lowering lipid levels in women is equivalent to the benefit in men. In these studies, the relative risk reduction for coronary heart disease ranged from 11 to 46 percent. These trials did not include enough women to provide definitive data regarding the efficacy of statins in women. However, the Heart Protection Study26 enrolled 20,536 persons with established cardiovascular disease, diabetes and treated hypertension, and included a substantial number of women. Statin therapy significantly reduced mortality, stroke, and vascular events in both men and women.
The AHA and the American College of Cardiology recommend statins as first-line therapy for hyperlipidemia in women.14 This recommendation represents an updating of the 1993 NCEP ATP II guidelines,8 in which hormone replacement therapy was recommended as first-line therapy, and is further supported by the updated ATP III guidelines.9
The initial pharmacologic management of women with hypercholesterolemia should include a statin if triglyceride levels are less than 200 mg per dL. In women of childbearing potential and in those with mild hypercholesterolemia or intolerance to statin therapy, treatment with a bile acid sequestrant is an alternative. If triglyceride levels are between 200 and 500 mg per dL (2.26 to 5.64 mmol per L), the non–HDL cholesterol level (the sum of LDL cholesterol and very-low-density lipoprotein cholesterol levels) should be reduced by maximizing therapy with an LDL cholesterol–lowering agent (e.g., a statin). Niacin or a fibrate may then be added with caution to achieve a lower non–HDL cholesterol level. If triglyceride levels exceed 500 mg per dL, they must be lowered quickly to prevent acute pancreatitis. This can be achieved using a fibrate or niacin, with subsequent attention given to lowering the LDL cholesterol level.
OTHER LIPID-ALTERING AGENTS
Bile acid sequestrants (resins) and niacin exert significant LDL cholesterol–lowering effects, although the benefits are less than those observed with statins. Colesevelam (Welchol), a newly available resin, reportedly has fewer gastrointestinal side effects than previous bile acid sequestrants. Because it does not have the triglyceride-raising effect of traditional resins and is not systemically absorbed, it may be a good option in women especially among those of childbearing age.32
Fibrates (e.g., gemfibrozil [Lopid], fenofibrate [Lipidil]) and niacin are particularly effective in lowering triglyceride levels and raising HDL cholesterol levels. One recent study33 in men with coronary heart disease and normal LDL cholesterol levels showed that gemfibrozil raised HDL cholesterol levels, lowered triglyceride levels, and reduced mortality by 22 percent. The value of fibrates in women whose primary lipid abnormality is a low HDL cholesterol level remains to be established.
Treatment with combined statins and fibrates may increase the risk of myositis and rhabdomyolysis. The use of niacin is limited by its poor tolerability and the fact that it may worsen glucose intolerance. Newer formulations, such as an extended-release form (Niaspan), may be better tolerated.
HORMONE REPLACEMENT
Estrogen replacement using unopposed conjugated equine estrogens has been shown to decrease LDL cholesterol and lipoprotein(a) levels and to increase HDL cholesterol, HDL2, and apolipoprotein A-I levels.34 Estrogen replacement therapy has also been purported to have a cardioprotective effect via beneficial vascular effects, antioxidant properties, fibrinogen and plasminogen activator inhibitor down-regulation, and calcium channel antagonism.34 In a recent meta-analysis35 based primarily on observational studies, postmenopausal replacement therapy was associated with a 35 percent reduction in the risk of coronary heart disease.
The Heart and Estrogen/progestin Replacement Study (HERS)36 was the first large, randomized clinical trial to assess the effect of hormone replacement therapy (conjugated equine estrogens plus medroxyprogesterone acetate) on cardiac outcomes in postmenopausal women with coronary heart disease. No overall reduction in the risk of nonfatal myocardial infarction or coronary heart disease mortality was found after 4.1 years of treatment, and a significant increase in cardiovascular events occurred in the test group during the first year. Hormone replacement therapy was also associated with an increased incidence of thromboembolic events and gallbladder disease. An angiographic study37 of women with coronary heart disease showed that conjugated equine estrogens alone or in combination with medroxyprogesterone acetate did not reduce the progression of coronary heart disease after three years. On the basis of these and other findings, the initiation or continuation of hormone replacement therapy for the sole purpose of secondary prevention of coronary heart disease is not recommended.38
The ongoing Women's Health Initiative should provide more definitive recommendations on the role of hormone replacement therapy in the primary prevention of coronary heart disease.
Final Comment
Optimal management of lipids is an important component of a comprehensive cardiovascular disease prevention program. Suggested checklists to use in evaluating preventive interventions in women with and without coronary heart disease, based on consensus recommendations for coronary heart disease risk factor management, are presented in Figures 314 and 4.14,39