Hypercholesterolemia is one of the major contributors to atherosclerosis and coronary heart disease in our society. The National Cholesterol Education Program of the National Institutes of Health has created a set of guidelines that standardize the clinical assessment and management of hypercholesterolemia for practicing physicians and other professionals in the medical community. In May 2001, the National Cholesterol Education Program released its third set of guidelines, reflecting changes in cholesterol management since their previous report in 1993. In addition to modifying current strategies of risk assessment, the new guidelines stress the importance of an aggressive therapeutic approach in the management of hypercholesterolemia. The major risk factors that modify low-density lipoprotein goals include age, smoking status, hypertension, high-density lipoprotein levels, and family history. The concept of “CHD equivalent” is introduced—conditions requiring the same vigilance used in patients with coronary heart disease. Patients with diabetes and those with a 10-year cardiac event risk of 20 percent or greater are considered CHD equivalents. Once low-density lipoprotein cholesterol is at an accepted level, physicians are advised to address the metabolic syndrome and hypertriglyceridemia.
Coronary heart disease (CHD) is the leading cause of morbidity and mortality in the United States, accounting for approximately 500,000 deaths per year and an associated annual morbidity cost of more than $200 billion.1 In the past three decades, numerous clinical and epidemiologic studies have shown repeatedly that an elevated blood cholesterol level is one of the major modifiable risk factors associated with the development of CHD.2 In particular, these studies have demonstrated that low-density lipoprotein (LDL) cholesterol is the primary lipoprotein mediating atherosclerosis. Other risk factors such as cigarette smoking, hypertension, diabetes, and a low level of high-density lipoprotein (HDL) cholesterol also have been implicated in CHD.3
In an effort to address this public health issue, the National Institutes of Health established the National Cholesterol Education Program in 1985. In 1988, the National Cholesterol Education Program, Adult Treatment Panel I (NCEP–ATP I) developed its first set of guidelines, establishing clear goals for patients with lipid abnormalities. In 1993, the NCEP–ATP II revised its initial recommendations and developed a second set of guidelines; in addition to emphasizing CHD risk status, this report placed even more emphasis on HDL levels, weight loss, and physical activity. In May 2001, the NCEP–ATP III released its third set of guidelines, reflecting changes in calculating coronary risk and in the management of hypercholesterolemia. According to the new guidelines, the number of patients with cholesterol levels that can be classified as abnormal has now tripled.
The NCEP–ATP III guidelines are similar to those in the second report in identifying LDL as a primary target of cholesterol-lowering therapy. Risk stratification continues to determine LDL goals and the intensity of LDL-lowering therapy. Dietary therapy remains the first line of treatment, with drug therapy reserved for use in patients at high risk for CHD or patients who do not respond to nonpharmacologic therapy. The differences between the third report and the previous report are summarized in Table 14 and discussed throughout this article.
TABLE 1 New Aspects of ATP III Guidelines
| Focus on multiple risk factors |
| Raises persons with diabetes but without CHD to the risk level of CHD risk equivalent |
| Uses Framingham projections of 10-year absolute CHD risk to identify patients with multiple (2 or more) risk factors for more intensive treatment |
| Identifies persons with metabolic syndrome as candidates for intensified therapeutic lifestyle changes |
| Modification of lipid/lipoprotein classification |
| Optimal LDL cholesterol level is now <100 mg per dL (2.60 mmol per L) |
| Increases categorical low HDL cholesterol level to <40 mg per dL (1.05 mmol per L) |
| Lowers triglyceride classification cut points |
| Support for implementation |
| Recommends complete lipoprotein profile (total, LDL, HDL, triglycerides) as preferred screening for assessing CHD risk status |
| Encourages use of plant sterols/stanols as a therapeutic dietary option to lower LDL cholesterol levels |
| Presents strategies for adherence to therapeutic lifestyle changes and drug therapies |
| Recommends treatment beyond LDL lowering for triglyceride levels >200 mg per dL (2.26 mmol per L) |
| One diet recommended for the entire population |
| 12-week trial of diet alone before adding pharmacotherapy |
ATP = Adult Treatment Panel; CHD = coronary heart disease; LDL = low-density lipoprotein; HDL = high-density lipoprotein.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2487.
Risk Assessment
In accordance with the new guidelines, optimal cholesterol screening now includes a lipoprotein profile (total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides), preferably using blood drawn in a fasting state. Screening is still recommended for all patients 20 years or older and every five years thereafter. The lipoprotein profile cannot be interpreted without knowledge of the patient's risk factors. Risk factor counting remains an important part of the guidelines (Table 2).4 In ATP III, diabetes is no longer on this risk factor list but is now included in a new category termed “CHD risk equivalent.”
TABLE 2 Major Risk Factors That Modify LDL Goals
| Positive risk factors |
| Age (men ≥ 45 years; women ≥ 55 years) |
| Low HDL cholesterol (<40 mg per dL [1.05 mmol per L]) |
| Cigarette smoking |
| Hypertension (blood pressure >140/90 mm Hg or taking antihypertensive medication) |
| Family history of premature CHD (CHD in male first-degree relative <55 years; |
| CHD in female first-degree relative <65 years) |
| Negative risk factor |
| High HDL cholesterol (> 60 mg per dL [1.55 mmol per L]); presence of this risk factor removes one risk factor from the total count |
LDL = low-density lipoprotein; HDL = high-density lipoprotein; CHD = coronary heart disease.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2487.
As reflected in ATP III, an additional step in the determination of CHD risk involves the calculation of the Framingham risk score (FRS) for persons with two or more risk factors. The addition of this step, along with the identification of major risk factors, allows physicians to recognize those patients at greatest risk for CHD.
Risk Factors
Since 1993, additional evidence for age, gender, and HDL importance has emerged, reinforcing the need to address these factors. The NCEP stance on smoking status, hypertension, and family history has remained essentially unchanged.
AGE AND GENDER
Recent studies have shown that the identification and treatment of dyslipidemia in patients 65 years and older can decrease the risk of first and recurrent coronary events. [Evidence level A, randomized controlled trials (RCTs)/meta-analyses] The Scandinavian Simvastatin Survival Study (4S), the Cholesterol and Recurrent Events (CARE) study, and the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS) were further analyzed for the population older than 65 years. In each treatment group, morbidity and mortality from cardiovascular disease was decreased by at least 29 percent.5–7 Although data are limited for patients older than 85 years, the elderly are candidates for cholesterol-lowering therapy. Initiation of therapy should be carefully examined in the context of comorbidities and the increased use of medications within this population.
Primary and secondary cardiovascular risk reduction is also important in women. Cardiovascular disease is the primary cause of death among American women, far exceeding the rate of all cancers combined.1 Women's mortality following myocardial infarction is three times that of men,8 suggesting that women may not be active participants in cardiovascular risk reduction strategies. By demonstrating cardiovascular event reduction through the use of statins, the 4S, CARE, and AFCAPS/TexCAPS trials support cholesterol-lowering therapy in women. [Evidence level A, RCTs/meta-analyses]
The recent Heart and Estrogen/progestin Replacement Study (HERS) has cast some doubt on the use of hormone replacement therapy (HRT) in the secondary prevention of CHD risk in postmenopausal women.9 Although several primary prevention studies support the use of HRT to prevent heart disease, the studies that support the use of statins to prevent heart disease in women are much stronger. Therefore, the ATP III prefers the initial use of a cholesterol-lowering agent to HRT for CHD risk reduction in postmenopausal women.
Although women have, on average, higher HDL levels than men, the ATP III guidelines do not distinguish between genders in regard to choosing a threshold level for HDL.
HDL CHOLESTEROL
The ATP III guidelines have been expanded to recognize the importance of HDL levels by raising the threshold of low HDL cholesterol from less than 35 mg per dL (0.90 mmol per L) to less than 40 mg per dL (1.05 mmol per L). [Evidence level C, consensus/expert opinion]
The “negative” risk factor (which negates one risk point from the major risk factor list) awarded to patients with an HDL level of 60 mg per dL (1.55 mmol per L) or higher in the last set of guidelines remains in ATP III. Since the release of the second set of guidelines in 1993, the significance of HDL cholesterol in the evolution of coronary artery disease has become more evident. The AFCAPS/TexCAPS study correlated a 6 percent increase in HDL cholesterol levels with a reduction of first acute major coronary events in men and women with baseline average LDL cholesterol levels and below-average HDL cholesterol levels. [Evidence level A, RCTs/meta-analyses]
Similarly, The Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial (VA-HIT) also demonstrated the benefit of raising HDL levels.10 In this study, HDL levels increased by 6 percent, triglyceride levels decreased by 31 percent, and coronary events decreased by 22 percent with gemfibrozil therapy, compared with placebo. Analyses of the data revealed a correlation between rising HDL levels and lower coronary event rates. [Evidence level A, RCTs/meta-analyses] This was not consistently true across the spectrum of baseline triglyceride levels, suggesting that HDL was the primary element responsible for the positive outcome.
Framingham Risk Score
The FRS is a risk assessment tool that has been derived from data collected in the Framingham Heart Study.11 As mentioned previously, the new NCEP guidelines recommend that patients with two or more risk factors have their FRS calculated. [Evidence level B, retrospective data analysis]
The FRS consists of points that are allocated for the various degrees of risk associated with five categories: age, total cholesterol level, HDL cholesterol level, tobacco smoking status, and hypertension (and whether the latter condition is treated). The summation of these points results in a percent risk of having a cardiac event in the next 10 years. Figures 1 and 2 outline FRS assessment for men and women, respectively.4
FIGURE 1. Calculating 10-Year Risk in Men
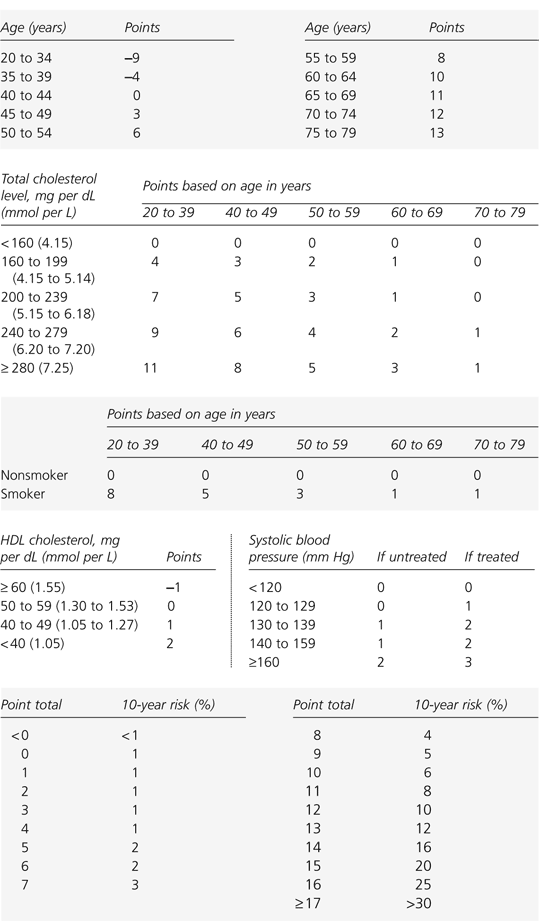
Framingham scoring system for estimating 10-year risk of coronary heart disease in men. (CHD = coronary heart disease; BP = blood pressure; HDL = high-density lipoprotein; FRS = Framingham risk scoring)
note:Risk assessment for determining the 10-year risk for developing CHD is carried out using Framingham risk scoring. The first step is to calculate the number of risk points for each risk factor from the table. For initial assessment, values for total cholesterol and HDL cholesterol are required. Total cholesterol and HDL cholesterol values should be the average of at least two measurements obtained from lipoprotein analysis. The designation smoker means cigarette smoking in the past month. The blood pressure value used is that obtained at the time of the assessment, regardless of whether the person is taking antihypertensive agents.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adults Treatment Panel III). JAMA 2001;285:2486–97.
FIGURE 2. Calculating 10-Year Risk in Women
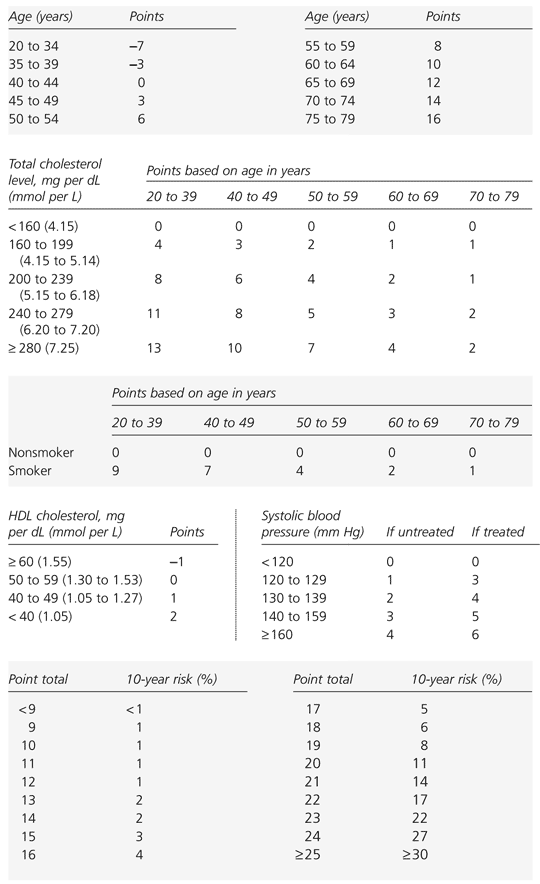
Framingham scoring system for estimating 10-year risk of coronary heart disease in women. (CHD = coronary heart disease; BP = blood pressure; HDL = high-density lipoprotein; FRS = Framingham risk scoring)
note:Risk assessment for determining the 10-year risk for developing CHD is carried out using Framingham risk scoring. The first step is to calculate the number of risk points for each risk factor from the table. For initial assessment, values for total cholesterol and HDL cholesterol are required. Total cholesterol and HDL cholesterol values should be the average of at least two measurements obtained from lipoprotein analysis. The designation smoker means cigarette smoking in the past month. The blood pressure value used is that obtained at the time of the assessment, regardless of whether the person is taking antihypertensive agents.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adults Treatment Panel III). JAMA 2001;285:2486–97.
CHD Equivalents
In the ATP II guidelines, risk categories were developed to separate patients with CHD (or its equivalent, such as abdominal aortic aneurysm, intermittent claudication, symptomatic carotid artery disease, etc.) from those without CHD. This distinction was primarily made to delineate the establishment of new target LDL levels for patients with CHD and to provide appropriate therapy to these patients based on their increased cardiovascular risk. In the ATP III guidelines, the target LDL level for patients with established CHD is still 100 mg per dL or less. Patients with diabetes and patients with an FRS of 20 percent or higher are considered CHD equivalents. Because patients with diabetes and patients with an FRS of 20 percent or higher are in the same risk category as CHD patients, they are also recommended to maintain an LDL level of 100 mg per dL. [Evidence level C, consensus/expert opinion]
The interventions for reaching the LDL goal in patients with diabetes or an FRS of 20 percent or higher are the same as those in patients with CHD.
The establishment of diabetes as a CHD risk equivalent reflects the prevalence of heart disease as a cause of death in the diabetic population. Three fourths of patients with diabetes die from heart disease–related illnesses compared with one half of the general population. Poor glycemic control has repeatedly been shown to be associated with an elevated risk of cardiovascular events.
According to the 2000 American Diabetes Association Guidelines, the primary goal of hyperlipidemia therapy in patients with type 2 diabetes (with or without vascular disease) is to reduce LDL cholesterol levels below 100 mg per dL.12 [Evidence level C, consensus/expert opinion] In addition to having LDL cholesterol particles of a more atherogenic variety, persons with diabetes have lower HDL cholesterol levels and higher triglyceride levels.13 The 4S and CARE trials included a sufficient number of diabetic patients to prove that after LDL cholesterol lowering, fewer cardiovascular events occurred in this patient population.
Treatment
The extent of LDL-lowering therapy depends on the patient's CHD risk. Two major modalities for lowering the LDL level advocated by the ATP III are therapeutic lifestyle changes (TLC) and drug therapy. Table 34 outlines LDL cholesterol goals and cut points for initiation of TLC and prescription medication. Patients are classified in one of three categories of risk: (1) CHD and CHD equivalents, (2) two or more risk factors (further delineated by an FRS of 10 to 20 percent versus 10 percent or less), or (3) zero or one risk factor.
TABLE 3 LDL Cholesterol Goals and Cut Points for Therapeutic Lifestyle Changes (TLC) and Drug Therapy in Different Risk Categories
| Risk category | LDL goal | LDL level at which to initiate TLC | LDL level at which to consider drug therapy |
|---|---|---|---|
| CHD or CHD risk equivalent (10-year risk >20 percent) | <100 mg/dL(2.60 mmol/L) | ≥ 100 mg/dL | ≥ 130 mg/dL (at 100 to 129 mg/dL, drug optional)* |
| 2 or more risk factors (10-year risk <20 percent) | <130 mg/dL (3.35 mmol/L) | ≥ 130 mg/dL | ≥ 130 mg/dL for 10-year risk of 10 to 20 percent; 160 mg/dL for 10-year risk of <10 percent |
| 0 to 1 risk factor† | <160 mg/dL (4.15 mmol/L) | ≥ 160 mg/dL | ≥ 190 mg/dL (at 160 to 189 mg/dL, LDL-lowering drug optional) |
LDL = low-density lipoprotein; CHD = coronary heart disease; HDL = high-density lipoprotein.
*—If an LDL cholesterol level of <100 mg per dL cannot be achieved by therapeutic lifestyle changes, some authorities recommend use of LDL-lowering drugs in this category. Others prefer using drugs that primarily modify triglycerides and HDL (i.e., nicotinic acid or fibrate). Clinical judgment also may call for deferring drug therapy in this subcategory.
†—People with zero to one risk factor almost always have a 10-year risk <10 percent; thus, 10-year risk assessment is not necessary in this group.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2486–97.
Therapeutic Lifestyle Changes
TLC encompasses diet, physical activity, and weight loss. ATP III continues to stress the importance of nonpharmacologic treatment but recognizes its limitations by reducing the trial of these modalities from six months to 12 weeks before considering the use of medications to assist in achieving recommended LDL goals. Furthermore, the composition of the diet has been modified, as noted in Table 4.4 One diet is recommended for all patients, as opposed to the two-step diet recommended in the previous guidelines.
TABLE 4 Nutrient Composition of the Therapeutic Lifestyle Changes Diet
| Nutrient | Recommended intake |
|---|---|
| Saturated fat* | <7 percent of total calories |
| Polyunsaturated fat | Up to 10 percent of total calories |
| Monounsaturated fat | Up to 20 percent of total calories |
| Total fat | 25 to 35 percent of total calories |
| Carbohydrates† | 50 to 60 percent of total calories |
| Fiber | 20 to 30 g per day |
| Protein | Approximately 15 percent of total calories |
| Cholesterol | <200 mg per day |
| Total calories‡ | Balance energy intake and expenditure to maintain desirable body weight |
LDL = low-density lipoprotein; HDL = high-density lipoprotein.
*—Avoid trans fatty acids as well because they increase LDL and lower HDL cholesterol levels.
†—Carbohydrates should be derived from foods rich in complex carbohydrates, including whole grains, fruits, and vegetables.
‡—Daily energy expenditure should include at least moderate physical activity.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) JAMA 2001;285:2486–97.
The distribution of the fat allowance has been altered to recognize the value of monounsaturated and polyunsaturated fatty acids. By replacing saturated fats (cheese, whole milk, red meat) with monounsaturated fats (olive, canola oil) and polyunsaturated fats (corn oil, peanuts), LDL is reduced. Although replacing saturated fats with a high-carbohydrate diet results in lower LDL levels, it has the adverse effect of raising triglycerides and lowering HDL. Saturated and trans-unsaturated fatty acids should be avoided.
The ATP III suggests the addition of plant stanols (hydrogenated phytosterols) to the patient's diet when initial attempts to alter the diet have not resulted in reaching the LDL goal. [Evidence level C, consensus/expert opinion] Plant stanols interfere with small-intestine absorption of intestinal and biliary cholesterol. While they lower LDL levels, they have no significant effect on HDL or triglyceride levels.14 Phytosterols can be found in many products, including margarine spreads. Other sources of phytosterols include sesame seeds and peanuts; soybeans are a natural source of phytosterols.
Physical inactivity is an independent risk factor, raising the risk of a cardiovascular event twofold.15 [Evidence level A, RCTs/meta-analyses] Aerobic exercise raises HDL levels and lowers triglyceride levels. When it results in weight loss, it contributes to LDL reduction. Weight loss also improves insulin sensitivity and serum glucose uptake, reducing the risk of diabetes. Cigarette smoking remains a cardiovascular risk factor. Patients who stop smoking can expect an increase of up to 30 percent in their HDL levels.16
Drug Therapy
As indicated by ATP III, failure of TLC to modify LDL cholesterol levels or the presence of high CHD risk levels warrants the use of drug therapy. Despite its use, particular attention to TLC should always be maintained and reinforced by the physician. Several drugs have specific effects on lipoprotein metabolism. Table 5 lists current classes of drugs and their associated lipid-altering effects.17
Benefits of LDL-lowering therapy initially should be monitored at six-week intervals. If the LDL goal based on established risk is not achieved, therapy should be intensified with an increase in drug dosage or the addition of another LDL-lowering drug. Even if the LDL goal is attained, other lipid risk factors should always be addressed. Once LDL levels are within normal range, a patient's lipoprotein profile should be monitored every six to 12 months. Compliance with medication remains an obstacle in the treatment of hypercholesterolemia.
TABLE 5 Characteristics of Drugs Affecting Lipoprotein Metabolism
| Agents | Cost | Effects on LDL | Effects on HDL | Effects on triglycerides |
|---|---|---|---|---|
| Bile acid sequestrants* | $$ | ↓↓↓ | ↑/minimal | None |
| Fibric acids† | $$$ | ↓ | ↑↑↑ | ↓↓↓↓ |
| Nicotinic acid‡ | $ | ↓↓ | ↑↑↑↑ | ↓↓↓ |
| Statins§ | $$$$ | ↓↓↓↓ | ↑↑ | ↓↓ |
LDL = low-density lipoprotein; HDL = high-density lipoprotein.
note :Dollar signs indicate relative costs among drugs (Red Book, Montvale, N.J. Medical Economics Data, 1999). Arrows indicate relative effects on lipoprotein levels, with four arrows denoting the greatest effect.
*—Cholestyramine: 4 to 16 g; colestipol: 5 to 30 g (suspension); colesevelam: 2.6 to 3.8 g.
†—Gemfibrozil: 600 mg twice daily; fenofibrate: 200 mg.
‡—Immediate-release (crystalline) nicotinic acid: 1.5 to 3 g; extended-release nicotinic acid (Niaspan): 1 to 2 g; sustained-release nicotinic acid: 1 to 2 g.
§—Lovastatin: 20 to 80 mg; pravastatin: 10 to 40 mg; simvastatin: 20 to 80 mg; fluvastatin: 20 to 80 mg; atorvastatin: 10 to 80 mg; cerivastatin: 0.4 to 0.8 mg.
Information from Safeer RS, Lacivita CL. Choosing drug therapy for patients with hyperlipidemia. Am Fam Physician 2000;61:3374–5.
Special Considerations
METABOLIC SYNDROME
The ATP III panel recognizes the importance of metabolic syndrome (also known as syndrome X) as a secondary target of therapy after recommended LDL levels are achieved. Metabolic syndrome, or insulin resistance syndrome, is defined as a cluster of abnormalities that include obesity, hypertension, dyslipidemia, and type 2 diabetes; it is associated with insulin resistance and compensatory hyperinsulinemia.18 It is estimated that this syndrome affects 70 to 80 million Americans.19
In particular, insulin resistance has been found in persons with low levels of HDL cholesterol and high levels of very low-density lipoprotein (VLDL) cholesterol and triglycerides.20 Because insulin resistance is often a precursor to the development of this syndrome, identification and potential treatment of insulin-resistant patients has been suggested as a means of preventing some or all components of the syndrome. However, measurement of fasting insulin levels is not standard practice at this time; criteria for normal and abnormal values have not yet been established.
As discussed in the new guidelines, the diagnosis of metabolic syndrome can be made when three or more of the risk determinants are present, as outlined in Table 6.4 These determinants can be measured readily in clinical practice. The treatment of metabolic syndrome is twofold: (1) reduce the underlying causes (i.e., obesity and physical inactivity), and (2) treat the associated lipid and nonlipid risk factors.
TABLE 6 Clinical Diagnosis of Metabolic Syndrome
| Risk factor | Defining level | |
|---|---|---|
| Abdominal obesity (waist circumference) | ||
| Men | >102 cm (> 40 in) | |
| Women | >88 cm (> 35 in) | |
| Triglyceride level | ≥ 150 mg/dL (170 mmol/L) | |
| HDL cholesterol | ||
| Men | <40 mg/dL (1.05 mmol/L) | |
| Women | <50 mg/dL (1.30 mmol/L) | |
| Blood pressure | ≥ 130 / 85 mm Hg | |
| Fasting glucose | ≥ 110 mg/dL | |
HDL = high-density lipoprotein.
Adapted with permission from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001; 285:2486–97.
HYPERTRIGLYCERIDEMIA
The ATP III recognizes the increasing number of studies correlating elevated triglyceride levels with increased coronary artery disease risk. By lowering the acceptable triglyceride level from the previous set of guidelines, the ATP III encourages a more aggressive approach to hypertriglyceridemia (Table 7).4 Diet and exercise are the primary modes of treating hypertriglyceridemia. If indicated, nicotinic acid and fibric acid derivatives are the most efficacious in lowering triglyceride levels. Triglyceride reduction is a secondary benefit of statins (the primary benefit being LDL cholesterol reduction).
TABLE 7 ATP III Classification of Triglyceride Levels and Treatment Strategies
| Classification | Serum level | Treatment strategy |
|---|---|---|
| Normal | <150 mg/dL (170 mmol/L) | None |
| Borderline-high | 150 to 199 mg/dL (170 to 2.25 mmol/L) | Achieve target goal for LDL cholesterol; emphasize weight reduction and physical activity |
| High | 200 to 499 mg/dL (2.26 to 5.64 mmol/L) | Achieve target goal for LDL cholesterol; institute weight reduction and physical activity; use drug therapy to achieve non-HDL goal* |
| Very high | ≥ 500 mg/dL (5.65 mmol/L) | Primary goal is triglyceride lowering followed by LDL lowering† |
ATP = Adult Treatment Panel; LDL = low-density lipoprotein; HDL = high-density lipoprotein; VLDL = very low-density lipoprotein.
*—There are two approaches to drug therapy: (1) intensify therapy with LDL-lowering drug or (2) nicotinic acid or fibrate can be added. Non-HDL = LDL + VLDL. The non-HDL goal is 30 mg per dL higher than the LDL goal.
†—The approach to triglyceride lowering is a diet very low in fat (15 percent of calorie intake), weight reduction, increased physical activity and, usually, a triglyceride-lowering drug (fibrate or nicotinic acid).
Information from Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2486–97.
The major lipid component of VLDL is the triglyceride. The NCEP mentions VLDL levels as part of a secondary treatment goal in patients with hypertriglyceridemia. VLDL levels can be monitored as part of the lipoprotein profile, and respond similarly to the lifestyle changes and to the medicines used to treat triglyceride levels.

