Rhabdomyolysis is a potentially life-threatening syndrome resulting from the breakdown of skeletal muscle fibers with leakage of muscle contents into the circulation. The most common causes are crush injury, overexertion, alcohol abuse and certain medicines and toxic substances. Several inherited genetic disorders, such as McArdle's disease and Duchenne's muscular dystrophy, are predisposing factors for the syndrome. Clinical features are often nonspecific, and tea-colored urine is usually the first clue to the presence of rhabdomyolysis. Screening may be performed with a urine dipstick in combination with urine microscopy. A positive urine myoglobin test provides supportive evidence. Multiple complications can occur and are classified as early or late. Early complications include severe hyperkalemia that causes cardiac arrhythmia and arrest. The most serious late complication is acute renal failure, which occurs in approximately 15 percent of patients with the syndrome. Early recognition of rhabdomyolysis and prompt management of complications are crucial to a successful outcome.
Rhabdomyolysis, which literally means striated muscle dissolution or disintegration,1 is a potentially lethal clinical and biochemical syndrome.2 Approximately 26,000 cases of rhabdomyolysis are reported annually in the United States.3 Prompt recognition and early intervention are vital. Full recovery can be expected with early diagnosis and treatment of the many complications that can develop in patients with this syndrome.
Clinical features of rhabdomyolysis may be absent initially, and its most serious complication, acute renal failure, is common. Many patients develop dialysis-dependent acute renal failure associated with the misuse of alcohol or other drugs.4 The nephrotoxicity of myoglobin is decreased by forced alkaline diuresis. Critically ill patients with acute renal failure are also likely to develop multiorgan failure syndrome, with a resultant increase in mortality.5
Pathophysiology
Muscle injury, regardless of mechanism, results in a cascade of events that leads to leakage of extracellular calcium ions into the intracellular space.6 The excess calcium causes a pathologic interaction of actin and myosin that ends in muscle destruction and fiber necrosis (Figure 1).
FIGURE 1.
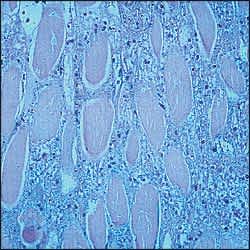
Fiber necrosis in rhabdomyolysis (hematoxylin and eosin).
With muscle injury, large quantities of potassium, phosphate, myoglobin, creatine kinase (CK) and urate leak into the circulation. Under physiologic circumstances, the plasma concentration of myoglobin is very low (0 to 0.003 mg per dL). If more than 100 g of skeletal muscle is damaged, serum haptoglobin binding capacity becomes saturated.6 The circulating myoglobin becomes “free” and is filtered by the kidneys. Myoglobin in the renal glomerular filtrate can precipitate and cause renal tubular obstruction, leading to renal damage.
Etiology and Risk Factors
Several investigators7,8 have attempted to categorize the many diverse causes and risk factors for rhabdomyolysis. The most common causes are alcohol abuse,9 muscle overexertion,10 muscle compression11 and the use of certain medications or illicit drugs.12–15 Medications and toxic substances that increase the risk of rhabdomyolysis are listed in Table 1.
TABLE 1 Medications and Toxic Substances That Increase the Risk of Rhabdomyolysis
| Direct myotoxicity | Indirect muscle damage |
| HMG-CoA reductase inhibitors, especially in combination with fibrate-derived lipid-lowering agents such as niacin (nicotinic acid; Nicolar) Cyclosporine (Sandimmune) Itraconazole (Sporanox) Erythromycin Colchicine Zidovudine (Retrovir) Corticosteroids | Alcohol Central nervous system depressants Cocaine Amphetamine Ecstasy (MDMA) LSD Neuromuscular blocking agents |
HMG-CoA = 3-hydroxy-3-methylglutaryl coenzyme A; LSD = lysergic acid diethylamide; MDMA = 3,4-methylene dioxymethamphetamine.
Other significant causes of rhabdomyolysis include electrical shock injury16 and crush injury. In crush injury, rhabdomyolysis occurs because of the release of necrotic muscle material into the circulation after compression is relieved in, for example, persons trapped in crashed cars or collapsed buildings. Heat-stroke17 and sporting activities,18 especially in previously untrained persons, are also common causes of the syndrome. Heat dissipation impairment18 from wearing heavy sports equipment or exercising in humid, warm weather increases the risk of rhabdomyolysis. Traumatic, heat-related, ischemic and exertional causes of rhabdomyolysis are listed in Table 2.
TABLE 2 Traumatic, Heat-Related, Ischemic and Exertional Causes of Rhabdomyolysis
| Traumatic causes |
| Lightning strike |
| Immobilization |
| Extensive third-degree burn |
| Crush injury |
| Heat-related causes |
| Heatstroke |
| Malignant hyperthermia |
| Neuroleptic malignant syndrome |
| Ischemic causes |
| Ischemic limb injury |
| Exertional causes |
| Marathon running |
| Physical overexertion in untrained athletes |
| Pathologic muscle exertion |
| Heat dissipation impairment |
| Physical overexertion in persons with sickle cell disease |
Numerous infectious and inflammatory processes can lead to rhabdomyolysis. Certain metabolic and endocrinologic disorders can also increase the risk of developing the syndrome. These processes and disorders are listed in Table 3.
TABLE 3 Infectious, Inflammatory, Metabolic and Endocrinologic Causes of Rhabdomyolysis
| Infectious causes |
| Viruses: influenza virus B, parainfluenza virus, adenovirus, coxsackievirus, echovirus, herpes simplex virus, cytomegalovirus, Epstein-Barr virus, human immunodeficiency virus |
| Bacteria: Streptococcus, Salmonella, Legionella, Staphyloccus and Listeria species |
| Inflammatory causes |
| Polymyositis |
| Dermatomyositis |
| Capillary leak syndrome |
| Snake bites (mostly in South America, Asia and Africa) |
| Metabolic and endocrinologic causes |
| Electrolyte imbalances: hyponatremia, hypernatremia, hypokalemia, hypophosphatemia, hypocalcemia |
| Hypothyroidism |
| Thyrotoxicosis |
| Diabetic ketoacidosis |
| Nonketotic hyperosmolar syndrome |
The cause of rhabdomyolysis can be obscure. In this situation, genetic etiologies should be considered (Table 4). A genetic disorder should be suspected in patients who have recurrent rhabdomyolysis after minimal to moderate exertion or after viral infections starting in childhood.
TABLE 4 Genetic Causes of Rhabdomyolysis
| Lipid metabolism |
| Carnitine palmitoyltransferase deficiency |
| Carnitine deficiency |
| Short-chain and long-chain acyl-coenzyme A dehydrogenase deficiency |
| Carbohydrate metabolism |
| Myophosphorylase deficiency (McArdle's disease) |
| Phosphorylase kinase deficiency |
| Phosphofructokinase deficiency |
| Phosphoglycerate mutase deficiency |
| Lactate dehydrogenase deficiency (characteristic elevation of creatine kinase level with normal lactate dehydrogenase level) |
| Purine metabolism |
| Myoadenylate deaminase deficiency |
| Duchenne's muscular dystrophy |
Clinical Presentation
Many clinical features of rhabdomyolysis are nonspecific, and the course of the syndrome varies depending on the underlying condition. The syndrome has local and systemic features, and early or late complications may occur. Prompt recognition of rhabdomyolysis is critical to preventing late complications.
Screening may be performed with a urine dipstick test.10 The orthotoluidine portion of the dipstick turns blue in the presence of hemoglobin or myoglobin. Positive urine “blood” can be used as a surrogate marker for myoglobin if freshly spun sediment of urine shows no red blood cells. In this setting, a serum sample with normal color indicates myoglobinuria, whereas a pigmented brown or red serum sample indicates hemoglobinuria.
In ambiguous cases, clinical suspicion of rhabdomyolysis is confirmed by a positive urine or serum test for myoglobin. Because it takes several days to obtain results, neither test should be relied on in making therapeutic decisions.
Clinical features of rhabdomyolysis are listed in Table 5. Local signs and symptoms may include muscle pain, tenderness and swelling. Systemic features may include tea-colored urine, which is usually the first sign, along with fever and malaise.
TABLE 5 Clinical Features of Rhabdomyolysis
| Local features | Systemic features |
| Muscle pain | Tea-colored urine |
| Tenderness | Fever |
| Swelling | Malaise |
| Bruising | Nausea |
| Weakness | Emesis |
| Confusion | |
| Agitation | |
| Delirium | |
| Anuria |
When a genetic disorder is suspected, forearm ischemic testing can be used to help differentiate among possible inherited causes (Table 6).19 A muscle biopsy with histochemical analysis is necessary to determine the specific cause of a genetic myopathy.
TABLE 6 Forearm Ischemic Test to Differentiate Genetic Causes of Rhabdomyolysis
Procedure
|
Interpretation
|
Information from Sinkeler SP, Wevers RA, Joosten EM, Binkhorst RA, Oei LT, Van't Hof MA, et al. Improvement of screening in exertional myalgia with a standardized ischemic forearm test. Muscle Nerve 1986;9:731–7.
Complications
The complications of rhabdomyolysis can be classified as early or late (Table 7). Severe hyperkalemia may occur secondary to massive muscle breakdown, causing cardiac arrhythmia and, possibly, cardiac arrest. Hypocalcemia is another early complication that can be potentiated by the release of large amounts of phosphate from the lysed muscle cells. Hepatic dysfunction occurs in approximately 25 percent of patients with rhabdomyolysis.20 Proteases released from injured muscle may be implicated in hepatic inflammation.
TABLE 7 Complications of Rhabdomyolysis
| Early complications |
| Hyperkalemia |
| Hypocalcemia |
| Hepatic inflammation |
| Cardiac arrhythmia |
| Cardiac arrest |
| Late complications |
| Acute renal failure |
| Disseminated intravascular coagulation |
| Early or late complication |
| Compartment syndrome |
Acute renal failure and diffuse intravascular coagulation are late complications of rhabdomyolysis (i.e., past 12 to 24 hours). Acute renal failure, the more serious complication, develops in up to 15 percent of patients21 and is associated with high morbidity and mortality. Renal damage results from the mechanical obstruction of tubules by myoglobin precipitation, the direct toxic effect of free chelatable iron on tubules, and hypovolemia. In addition, the release of vasoactive kinins from muscle may interfere with renal hemodynamics. There is a loose predictive correlation between CK levels and the development of acute renal failure, with levels higher than 16,000 units per L more likely to be associated with renal failure.21 The rate at which serum creatinine levels increase is typically faster in patients with myoglobinuric renal failure (up to 2.5 mg per dL per day [220 μmol per L]) than in those with other causes of acute renal failure.
Disseminated intravascular coagulation may develop in patients with rhabdomyolysis. This complication is usually worse on the third to fifth day of presentation. Prompt recognition and vigorous treatment of the underlying cause is necessary.
Compartment syndrome may be an early or late complication, resulting mainly from direct muscle injury or vigorous muscle activity. This complication occurs primarily in muscles whose expansion is limited by tight fascia, such as the anterior tibial muscles. Peripheral pulses may still be palpable, in which case nerve deficits (mainly sensory) are more important findings. A delay of more than six hours in diagnosing this complication can lead to irreversible muscle damage or death. Decompressive fasciotomy should be considered if the compartment pressure is greater than 30 mm Hg.22
Treatment
The treatment of rhabdomyolysis is primarily directed at preserving renal function. Up to 12 L of fluid may be sequestered in the necrotic muscle tissues, thereby contributing to hypovolemia, which is one cause of renal failure in patients with rhabdomyolysis.23
Intravenous (IV) hydration must be initiated as early as possible. In the patient with a crush injury, IV fluids should be started even before the trapped limb is freed and decompressed, and certainly no later than six hours after decompression. The longer it takes for rehydration to be initiated, the more likely it is that oliguric renal failure (less than 500 mL of urine per day) or anuric renal failure (less than 50 mL of urine per day) will be established.23 Investigators in one study24 found that forced diuresis within the first six hours of admission prevented all episodes of acute renal failure.
Initially, normal saline should be given at a rate of 1.5 L per hour. Urine output should be maintained at 300 mL per hour until myoglobinuria has ceased. High rates of IV fluid administration should be used at least until the CK level decreases to or below 1,000 units per L. If these measures successfully thwart the development of oliguria, the patient can be switched to 0.45 percent saline with the addition of one or two ampules of sodium bicarbonate (40 mEq) and 10 g per L of mannitol. Diuretics (loop or other types) should not be used because they do not improve, and may actually compromise, the final renal outcome.
The objectives are to alkalinize urine to a pH of greater than 6.5 (thereby decreasing the toxicity of myoglobin to the tubules) and to enhance the flushing of myoglobin casts from renal tubules by means of osmotic diuresis. However, these measures should not be employed if oliguria is established despite initial generous hydration with normal saline. The use of mannitol remains controversial as it is mostly supported by experimental animal studies and retrospective clinical studies.25,26 In one study,27 mannitol did not confer additional protection compared with normal saline alone. There are also some concerns about the use of sodium bicarbonate, because it may worsen hypocalcemia or precipitate calcium phosphate deposition on various tissues.28
Elderly patients should be treated in an intensive care unit so that vital signs, intake and hourly output can be closely monitored and fluid overload can be quickly detected. Invasive hemodynamic monitoring is critical to fine-tune treatment in patients with comorbid cardiovascular disorders or preexisting chronic renal dysfunction.
Hemodialysis may be a therapeutic modality. Despite treatment, patients with rhabdomyolysis often develop oliguric acute tubular necrosis. In this situation, hemodialysis should be started and carried on aggressively, frequently on a daily basis. If given enough time, many patients partially or completely recover renal function. The chances of recovery are obviously much higher in the absence of preexisting renal insufficiency.
Finally, initial hypocalcemia should not be corrected unless a patient is symptomatic. It is important to avoid further aggravating the hypercalcemia that commonly develops during the recovery phase of rhabdomyolysis, when calcium deposited in the injured muscles is mobilized back to the extracellular space.29

