Family physicians frequently treat bacterial skin infections in the office and in the hospital. Common skin infections include cellulitis, erysipelas, impetigo, folliculitis, and furuncles and carbuncles. Cellulitis is an infection of the dermis and subcutaneous tissue that has poorly demarcated borders and is usually caused by Streptococcus or Staphylococcus species. Erysipelas is a superficial form of cellulitis with sharply demarcated borders and is caused almost exclusively by Streptococcus. Impetigo is also caused by Streptococcus or Staphylococcus and can lead to lifting of the stratum corneum resulting in the commonly seen bullous effect. Folliculitis is an inflammation of the hair follicles. When the infection is bacterial rather than mechanical in nature, it is most commonly caused by Staphylococcus. If the infection of the follicle is deeper and involves more follicles, it moves into the furuncle and carbuncle stages and usually requires incision and drainage. All of these infections are typically diagnosed by clinical presentation and treated empirically. If antibiotics are required, one that is active against gram-positive organisms such as penicillinase-resistant penicillins, cephalosporins, macrolides, or fluoroquinolones should be chosen. Children, patients who have diabetes, or patients who have immunodeficiencies are more susceptible to gram-negative infections and may require treatment with a second- or third-generation cephalosporin.
Bacterial skin infections are the 28th most common diagnosis in hospitalized patients.1 Cellulitis, impetigo, and folliculitis are the most common bacterial skin infections seen by the family physician. The percentage of office visits for cellulitis was 2.2 percent, and for impetigo, it was 0.3 percent, in a cohort of almost 320,000 health plan members (data taken from primary physician diagnosis codes from January 1, 1999 to December 1, 1999 for Intermountain Health Care, Salt Lake City). Knowledge of the presentation, histopathology, and microbiology for each type of infection is important for proper care of the patient. The presentation, etiology, and current management of these diseases are presented.
Cellulitis
Cellulitis is a painful, erythematous infection of the dermis and subcutaneous tissues that is characterized by warmth, edema, and advancing borders (Table 1). Cellulitis commonly occurs near breaks in the skin, such as surgical wounds, trauma, tinea infections (Figure 1), or ulcerations, but occasionally presents in skin that appears normal. Patients may have a fever and an elevated white blood cell count. Cellulitis can occur on any part of the body. Among the patients in the cohort above, the most common sites of cellulitis were the legs and digits, followed by the face, feet, hands, torso, neck, and buttocks (data taken from primary physician diagnosis codes from January 1, 1999 to December 1, 1999 for health plan members of Intermountain Health Care, Salt Lake City).
TABLE 1 Descriptions of Bacterial Skin Infections
| Disease | Description |
|---|---|
| Carbuncle | A network of furuncles connected by sinus tracts |
| Cellulitis | Painful, erythematous infection of deep skin with poorly demarcated borders |
| Erysipelas | Fiery red, painful infection of superficial skin with sharply demarcated borders |
| Folliculitis | Papular or pustular inflammation of hair follicles |
| Furuncle | Painful, firm or fluctuant abscess originating from a hair follicle |
| Impetigo | Large vessicles and/or honey-crusted sores |
FIGURE 1.
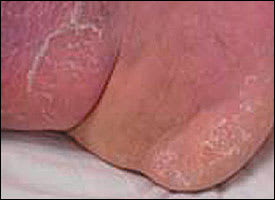
Cellulitis secondary to tinea infection.
In otherwise healthy adults, isolation of an etiologic agent is difficult and unrewarding. If the patient has diabetes, an immunocompromising disease, or persistent inflammation, blood cultures or aspiration (some physicians inject sterile nonpreserved saline before aspiration) of the area of maximal inflammation may be useful.2–4 For infection in patients without diabetes, empiric treatment with a penicillinase-resistant penicillin, first-generation cephalosporin, amoxicillin-clavulanate (Augmentin), macrolide, or fluoroquinolone (adults only) is appropriate.5 Limited disease can be treated orally, but more extensive disease requires parenteral therapy. Marking the margins of erythema with ink is helpful in following the progression or regression of cellulitis (Figure 2). Outpatient therapy with injected ceftriaxone (Rocephin) provides 24 hours of parenteral coverage and may be an option for some patients. The patient should be seen the following day to reassess disease progression.
FIGURE 2.
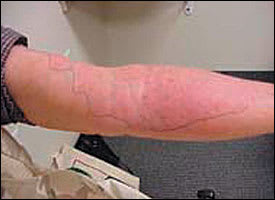
Inked margins of cellulitis.
Most cases of superficial cellulitis improve within one day, but patients who exhibit thickening of the dermis usually take several days of parenteral antibiotics before significant improvement occurs. Antibiotics should be maintained for at least three days after the resolution of acute inflammation.5 Adjunctive therapy includes the following: cool compresses; appropriate analgesics for pain; tetanus immunization; and immobilization and elevation of the affected extremity.6
A parenteral second- or third-generation cephalosporin (with or without an aminoglycoside) should be considered in patients who have diabetes, immunocompromised patients, those with unresponsive infections, or in young children.5 The patient may also require a plain radiograph of the area or surgical debridement to evaluate for gas gangrene, osteomyelitis, or necrotizing fasciitis.6
Recurrent episodes of cellulitis or undergoing surgery, such as mastectomy with lymph node dissection, can compromise venous or lymphatic circulation and cause dermal fibrosis, lymphedema, epidermal thickening, and repeated episodes of cellulitis. These patients may benefit from prophylaxis with erythromycin, penicillin, or clindamycin (Cleocin).5,7
Periorbital cellulitis is caused by the same organisms that cause other forms of cellulitis and is treated with warm soaks, oral antibiotics, and close follow-up.8 Children with periorbital or orbital cellulitis often have underlying sinusitis.9 If the child is febrile and appears toxic, blood cultures should be performed and lumbar puncture considered. Haemophilus influenzae type b (Hib) in young children was a significant concern until the widespread use of the Hib vaccine and coverage with a parenteral third-generation cephalosporin was used routinely. Recently, some researchers have recommended no longer routinely covering for H. influenzae.8–10
Orbital cellulitis occurs when the infection passes the orbital septum and is manifested by proptosis, orbital pain, restricted eye movement, visual disturbances, and concomitant sinusitis. Complications include abscess formation, persistent blindness, limited eye movement, diplopia, and, rarely, meningitis.11 This ocular emergency requires intravenous antibiotics, otorhinolaryngology, and ophthalmologic consultation.12
Perianal cellulitis is caused by group A beta-hemolytic streptococcal infection and occurs most often in children. A study13 of children with perianal cellulitis found a mean age of onset of 4.25 years. Ninety percent of patients presented with dermatitis, 78 percent with itching, 52 percent with rectal pain, and 35 percent with blood-streaked stools. Despite 10 days of oral antibiotics (primarily penicillin or erythromycin), the recurrence rate was high at 39 percent. If there is recurrence, the presence of an abscess should be considered, with needle aspiration of the site for bacteriology being more accurate than a skin swab.14
Erysipelas
Erysipelas (Figure 3), also known as St. Anthony's fire, usually presents as an intensely erythematous infection with clearly demarcated raised margins, and often with associated lymphatic streaking (Table 1). Common sites are the legs and face.15 Most cases do not have an inciting wound or skin lesion and are preceded by influenza-like symptoms. The incidence of erysipelas is rising, especially in young children, the elderly, persons with diabetes, alcoholic persons, and patients with compromised immune systems or lymphedema.6
FIGURE 3.
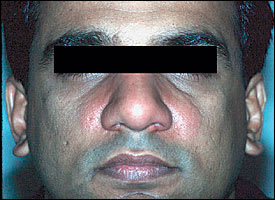
Erysipelas.
Erysipelas is caused almost exclusively by beta-hemolytic streptococcus and thus can be treated with standard dosages of oral or intravenous penicillin. However, most physicians treat this infection the same as cellulitis, which is outlined earlier. Adjunctive treatment and complications are the same as for cellulitis.
Impetigo
Impetigo is most commonly seen in children aged two to five years and is classified as bullous or nonbullous (Table 1). The nonbullous type predominates and presents with an erosion (sore), cluster of erosions, or small vesicles or pustules that have an adherent or oozing honey-yellow crust. The predilection for the very young can be remembered by the common lay misnomer, “infant tigo.” Impetigo usually appears in areas where there is a break in the skin, such as a wound, herpes simplex infection, or maceration associated with angular cheilitis (Figure 4), but Staphylococcus aureus can directly invade the skin and cause a de novo infection.6
FIGURE 4.
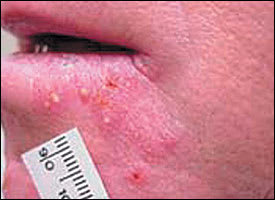
Nonbullous impetigo.
The bullous form of impetigo presents as a large thin-walled bulla (2 to 5 cm) containing serous yellow fluid. It often ruptures leaving a complete or partially denuded area with a ring or arc of remaining bulla (Figure 5). More than one area may be involved and a mix of bullous and nonbullous findings can exist. Nonbullous impetigo was previously thought to be a group A streptococcal process and bullous impetigo was primarily thought to be caused by S. aureus. Studies16,17 now indicate that both forms of impetigo are primarily caused by S. aureus with Streptococcus usually being involved in the nonbullous form. If the infection is a toxin-producing, phage group II, type 71 Staphylococcus (the same toxin seen in Staphylococcus scalded skin syndrome, a medical emergency where large sheets of the upper epidermis slough off), large bullae will form as the toxin produces intradermal cleavage.18 Otherwise, smaller bullae develop and the honey-crusted lesions predominate.
FIGURE 5.
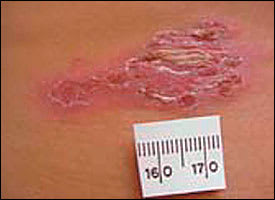
Ruptured bullous impetigo.
A study19 published in 1990 concluded that topical mupirocin (Bactroban) ointment is as effective as oral erythromycin in treating impetigo. However, because the lesions of bullous impetigo can be large and both forms of impetigo can have satellite lesions, an oral antibiotic with activity against S. aureus and group A beta-hemolytic streptococcal infection is warranted in nonlocalized cases. Because of developing resistance, erythromycin is no longer the drug of choice.17 Azithromycin (Zithromax) for five days and cephalexin (Keflex) for 10 days have been shown to be effective and well-tolerated.20 Dicloxacillin (Pathocil), oxacillin (Prostaphlin), first-generation cephalosporins, or amoxicillin-clavulanate are also acceptable alternatives.5 Broad-spectrum fluoroquinolones have also been shown to be effective, and several have been approved by the U.S. Food and Drug Administration for treating skin and soft tissue infections.21 These medications have excellent skin penetration and good bioavailability, but no generic forms are currently available, and they are only approved for use in adults.
As with other diseases involving Streptococci, there is a small chance of developing glomerulonephritis, especially in children aged two to six years. Presenting signs and symptoms of glomerulonephritis include edema and hypertension; about one third of patients have smoky or tea-colored urine. Streptococcal glomerulonephritis usually resolves spontaneously although acute symptoms and problems may occur.22
Impetigo can be spread by direct person-to-person contact, so appropriate hygiene is warranted. Nasal carriage of S. aureus has been implicated as a source of recurrent disease and can be reduced by the topical application of mupirocin twice daily for five days.23
Folliculitis
Hair follicles can become inflamed by physical injury, chemical irritation, or infection that leads to folliculitis (Table 1). Classification is by the depth of involvement of the hair follicle. The most common form is superficial folliculitis that manifests as a tender or painless pustule that heals without scarring.24 The hair shaft will frequently be seen in the center of the pustule. Multiple or single lesions can appear on any skin bearing hair including the head, neck, trunk, buttocks, and extremities. Associated systemic symptoms or fever rarely exist. S. aureus is the most likely pathogen; however, commensal organisms such as yeast and fungi occasionally appear, especially in immunocompromised patients. These lesions typically resolve spontaneously. Topical therapy with erythromycin, clindamycin, mupirocin, or benzoyl peroxide can be administered to accelerate the healing process.25
Staphylococci will occasionally invade the deeper portion of the follicle, causing swelling and erythema with or without a pustule at the skin surface. These lesions are painful and may scar. This inflammation of the entire follicle or the deeper portion of the hair follicle (isthmus and below) is called deep folliculitis. Oral antibiotics are usually used in the treatment and include first-generation cephalosporins, penicillinase-resistant penicillins, macrolides, and fluoroquinolones.
Gram-negative folliculitis usually involves the face and affects patients with a history of long-term antibiotic therapy for acne. Pathogens include Klebsiella, Enterobacter, and Proteus species. It can be treated as severe acne with isotretinoin (Accutane), but use of isotretinoin is associated with major side effects, including birth defects.26
“Hot tub” folliculitis is caused by Pseudomonas aeruginosa contamination of under-treated water in a hot tub or whirlpool. Multiple pustular or papular perifollicular lesions appear on the trunk and sometimes extremities within six to 72 hours after exposure (Figure 6), and mild fever and malaise may occur. Lesions in the immunocompetent patient typically resolve spontaneously within a period of seven to 10 days.25 Treatment is directed at prevention by appropriately cleaning the whirlpool or hot tub and maintaining appropriate chlorine levels (bromine and copper solutions are less common alternatives) in the water.
FIGURE 6.
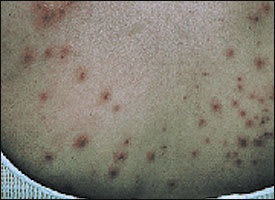
Folliculitis caused by contamination of undertreated water in a hot tub or whirlpool.
Furuncles and Carbuncles
Furuncles and carbuncles occur as a follicular infection progresses deeper and extends out from the follicle (Table 1). Commonly known as an abscess or boil, a furuncle is a tender, erythematous, firm or fluctuant mass of walled-off purulent material, arising from the hair follicle. These lesions may occur anywhere on the body, but have a predilection for areas exposed to friction. Furuncles rarely appear before puberty. The pathogen is usually S. aureus. Typically, the furuncle will develop into a fluctuant mass and eventually open to the skin surface, allowing the purulent contents to drain, either spontaneously or following incision of the furuncle.
Carbuncles are an aggregate of infected hair follicles that form broad, swollen, erythematous, deep, and painful masses that usually open and drain through multiple tracts. Constitutional symptoms, including fever and malaise, are commonly associated with these lesions but are rarely found with furuncles. With both of these lesions, gentle incision and drainage is indicated when lesions “point” (fluctuant or boggy with a thin shiny appearance of the overlying skin); caution should be taken to not incise deeper than the pseudo capsule that has been built at the site of infection. Loculations should be broken with a hemostat. The wound may be packed (usually with iodoform gauze) to encourage further drainage. In severe cases, parenteral antibiotics such as cloxacillin (Tegopen), or a first-generation cephalosporin such as cefazolin (Ancef), are required.27 The physician should be aware of the potential for gas-containing abscesses or necrotizing fasciitis, which require immediate surgical debridement.
Final Comment
The majority of bacterial skin infections are caused by the gram-positive bacteria Staphylococcus and Streptococcus species. Antibiotics are used empirically with consideration for resistance patterns. Current antibiotic recommendations include penicillinase-resistant penicillins, first-generation cephalosporins, azithromycin, clarithromycin, amoxicillin-clavulanic acid, or a second-generation fluoroquinolone in the skeletally mature patient. Gram-negative coverage with a second-, third-, or fourth-generation cephalosporin is usually indicated in children under three years and in patients with diabetes or who are immunocompromised.

