Stress fractures of the tarsal navicular bone are being recognized with increasing frequency in physically active persons. Diagnosis is commonly delayed, and outcome often suffers because physicians lack familiarity with the condition. Navicular stress fractures typically present in a running athlete who has gradually increasing pain in the dorsal midfoot with occasional radiation of pain down the medial arch. Because initial plain films are often normal, the next diagnostic test of choice is triple-phase bone scan, which is positive early in the process and localizes the lesion well. After a positive bone scan, a computed tomographic scan should be obtained to provide anatomic detail and guide therapy. Nondisplaced, noncomminuted fractures respond well to six weeks of non–weight-bearing cast immobilization. Displacement, comminution, and delayed or nonunion fractures are indications for surgical open reduction internal fixation.
The navicular bone of the foot is a flattened, concave, boat-shaped bone wedged between the head of the talus and the three cuneiforms. Some common variants have an additional facet articulation with the cuboid bone. Medially the navicular tuberosity provides an insertion site for the tibialis posterior tendon (Figure 1). The location and unique impingement during foot strike of the navicular bone predispose it to well-localized stress and remodeling.1 During foot strike, the navicular bone becomes impinged with maximal effort between the proximal talus and the distal cuneiforms. Biomechanical analysis of navicular motion during stride reveals that most of this impingement force is focused at the central one third of the navicular bone.2–4
FIGURE 1.
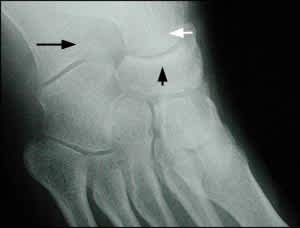
Radiograph of the foot, anteroposterior view, showing the calcaneus (black arrow), talus (white arrow), and navicular bone (arrowhead).
This anatomic impingement is even more significant in light of the vascular anatomy of the navicular bone. A microangiopathic study5 of cadaveric feet showed that while the navicular bone is supplied by both the anterior and posterior tibial arteries, the branches enter at the small “waist” of cortical bone and branch out to supply the medial and lateral thirds.2 This design leaves the central one third, the area of greatest stress, as an area of relative avascularity (Figure 2).
FIGURE 2.
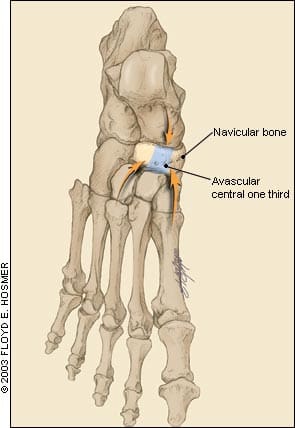
Dorsal view of the tarsal navicular bone. The avascular central one-third is also the fulcrum of the impingement forces from the first and third metacarpal bones, as well as the talus.
Tarsal navicular stress fractures were first described in 1958 in a study of racing grey-hounds.6 The fractures were always seen in the right hind foot and were initially termed “broken hock.” The counterclockwise racing of the greyhounds on a banked track may have predisposed their uphill foot to increased stress. The lesion was first described in humans in a 1970 study.7 Even then, the difficulty of locating the lesion on plain radiographs was noted. Because of the vertical nature of the fracture, it was understood that diagnosis “may require special views and laminography for detection.”7
Studies8–10 in the 1980s projected a navicular fracture incidence of 0.7 to 2.4 percent of all stress fractures. Recent studies1,11,12 reveal an incidence of 14 to 35 percent of all stress fractures. A study11 of elite-level athletes showed that track athletes accounted for 59 percent of all tarsal navicular stress fractures.
Vague symptomatology and elusive radiographic localization typically lead to a delay in diagnosis averaging four months from initial symptom onset.5,13 Early diagnosis of these lesions and proper management usually yields a favorable outcome5; however, delayed diagnosis may result in inadequate treatment and either delayed union or nonunion healing of the fracture.13,14 In a landmark study,5 the most common treatment of navicular stress fractures was found to be limitation of activity, which had a dismal 26 percent cure rate.
Mechanism of Injury
The anatomic predisposition to localization of stress in the avascular central one third of the navicular bone combined with the repetitive foot strike of weight-bearing exercises that involve antagonistic muscular load are thought to eventually result in bone failure.15 The premonitory symptoms of navicular “bone strain” are generally undetectable by radiographs and computed tomographic (CT) scans. Until a diagnosis is made, there is increased stress and bony resorption focused at the central one third of the navicular bone. A bone scan performed at this phase will be positive. If stressful activity continues, the resorptive changes continue to progress until a fracture line becomes evident on CT scan and plain radiographs.1,16
Several authors have attempted to identify persons who are at increased risk of navicular stress fracture. One study17 used force-plate analysis and proposed calcaneal pitch angle, talometatarsal angle, and pronation velocity as potential risk factors for navicular stress fractures. Other studies5,18–20 have shown that the following factors predispose a person to navicular stress fractures: pes cavus, wide-heeled shoes, short first metatarsals, metatarsus adductus, metatarsus hyperostosis, medial narrowing of the talonavicular joint, talar beaking, limited subtalar motion, and limited ankle dorsiflexion. However, no statistically significant risk factors have been demonstrated, and no consensus exists as to persons at risk. As with all overuse injuries, training errors, improper equipment, improper technique, environment, and anatomic variants may all increase the risk for injury.
Clinical Presentation
Commonly occurring in track and field athletes (Table 1),1,5,7,10,13,17,18,21–23 navicular stress fractures present as vague, aching pain in the dorsal midfoot that may radiate along the medial arch. The pain typically increases with activity such as running and jumping. With continued participation, the pain occurs sooner during activity and lasts longer into postactivity rest periods.1,5,17,24 Symptoms are rarely bilateral.
TABLE 1 Activity-Specific Incidence of Navicular Stress Fractures
Various factors contribute to the common delay in diagnosis of navicular stress fractures. Often, athletes can continue activity until pain increases too much by altering their gait and decreasing use of the forefoot.18 Pain also resolves rapidly with rest, making it possible for athletes to resume participation after a week of respite from activity.
Physical Examination
Patients who present with navicular stress fractures typically have a normal range of motion and strength to manual muscle testing, and the neurovascular examination is normal. There is no ecchymosis or deformity and usually no swelling. The talonavicular joint can be localized by inverting-everting the forefoot. The nickel-sized area at the central region of the proximal dorsal navicular bone, referred to as the “N” spot, is tender in 81 percent of patients with navicular stress fractures.5 Patients generally exhibit increased pain with hopping, toe hopping, and standing on their toes in the equinus position.18
Radiologic Tests
PLAIN RADIOGRAPHS
When suspicion justifies diagnostic studies, the initial step is typically plain radiographs. Unfortunately, only 33 percent of plain radiographs have sensitivity for navicular stress fractures,1,25 because the majority of fractures are incomplete.1 In addition, because bony resorption requires 10 days to three weeks to allow visualization of these fractures on plain radiographs, even complete fractures are often not seen on initial films.26 However, plain films are useful if positive, and they also assist in ruling out other etiologies.27
BONE SCAN
If plain films are negative or inconclusive, triple-phase bone scan is the next recommended diagnostic procedure. Bone scan, unlike plain radiography, is positive at an early stage and is almost 100 percent sensitive for navicular stress fracture.1 The entire navicular bone demonstrates uptake in all phases in a positive test24 (Figure 3); delayed-phase images may take up to two years after union to return to normal.15 The high negative predictive value of bone scanning essentially excludes the diagnosis with a negative test; however,the positive predictive value is lower. A bone scan may be positive with negative follow-up studies (e.g., CT scan, magnetic resonance imaging [MRI]). This phenomenon is thought to represent “bone strain” or subclinical stress reaction, and it inevitably proceeds to actual fracture if physical activity is continued at the same intensity level.28 Positive bone scans must always be correlated with further imaging (i.e., CT scan) because of lack of specificity, poor demonstration of comminution and displacement, and lack of resolution of the anatomic characteristics of these fractures.24
FIGURE 3.
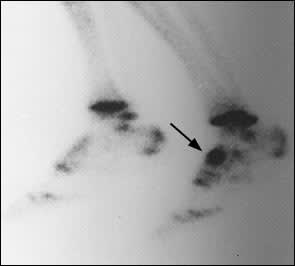
Triple-phase bone scan of the lower extremities revealing intense uptake in the entire left navicular bone (arrow), the classic sign of a navicular stress fracture.
CT SCANNING
CT scanning is the gold standard for optimal evaluation of a fracture once bone scan has demonstrated increased uptake in the navicular bone.24 Unlike bone scanning, the anatomic resolution of CT scans is excellent. The best images are obtained with 1.5-mm slices using a bone algorithm through the plane of the talonavicular joint.29 False-negative CT reports, estimated at 7 percent, are typically caused by confusion of an actual fracture with nutrient arteries.13
CT scans most commonly demonstrate a partial fracture coursing from the proximal dorsal central one third of the navicular bone and extending toward the distal plantar pole of the bone1 (Figure 4). Fragmentation is seen in approximately 14 percent of navicular fractures.13 Sclerosis is typically seen at the proximal articular rim; this finding is believed to represent the normal stress of weight bearing, not early evidence of nonunion.29
FIGURE 4.
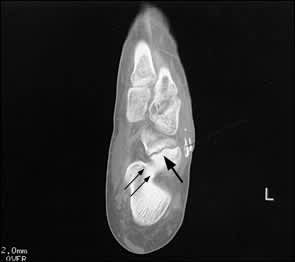
Computed tomographic scan of the left foot. This axial view reveals complete fracture of the navicular bone (arrow), located more laterally than is typical. Incidentally noted is a calcaneonavicular tarsal coalition (double arrow).
A proposed CT-based classification system separates fractures into three groups: dorsal cortical break (type I), fracture propagation into the navicular body (type II), and fracture propagation into another cortex (type III). Type III lesions have the longest average healing time with conservative care. These lesions may warrant more aggressive treatment and orthopedic referral for possible open reduction internal fixation.30
MRI
MRI is a reasonable choice for imaging navicular stress fractures, particularly if bone scanning is not available, because it is extremely sensitive and provides good spatial resolution.31 Because of its cost and limited availability, MRI is frequently overlooked as a modality; however, in cases of high pre-test probability, MRI offers the attractive profile of almost 100 percent sensitivity as well as good anatomic resolution of the fracture. Bony edema on T2-weighted images, an early finding, can add useful information regarding the acuity of the injury and delineate associated injury32 (Figure 5). MRI does not add significant information if bone scan and CT scan are available. If cost is not an issue, and time is of the essence, an MRI can give information similar to that of bone scan plus CT scan.
FIGURE 5.
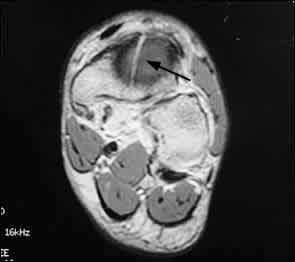
Magnetic resonance image of the foot, revealing a sagittally oriented fracture through the central region of the navicular (arrow). Note extensive surrounding edema.
Treatment and Return to Activity
Therapeutic intervention in navicular stress fractures has been clearly defined1,13,18,19 (Table 2).13 In 86 percent of cases of nondisplaced navicular stress fracture, six weeks in a well-molded non–weight-bearing cast results in healing.1 If clinical healing has occurred and there is no tenderness at the “N” spot after six weeks in the cast, functional rehabilitation may begin1 (Table 31). The patient should be reassessed every two weeks and, if there is no increase in tenderness at the “N” spot, can typically return to full activity six weeks after removal of the cast. If the navicular bone remains tender, the patient should be kept in the non–weight-bearing cast for two more weeks and clinically reexamined after that time.1,5,13,19,24
Once rehabilitation has begun, any foot pain must be reevaluated carefully. General discomfort caused by immobilization, along with stiffness of the talocrural, subtalar, and midtarsal joints, is to be expected and responds to physical therapy.1 However, if the discomfort localizes over the navicular bone, further immobilization or surgical intervention may be indicated.
TABLE 3 Functional Rehabilitation of Tarsal Navicular Stress Fracture
| Weeks 1 to 2: | Normal activities of daily living, swimming, and water running are permitted. |
| Weeks 3 to 4: | If there is no increase in tenderness at the “N” spot, jogging on grass for five minutes on alternate days is permitted. After one week, increase to 10 minutes on alternate days. |
| Weeks 5 to 6: | If “N” spot is not tender, running at 50 percent of maximum speed with walk recovery on alternate days is permitted. This speed can be increased to 75 percent over another two weeks. |
| After 6 weeks: | If “N” spot is not tender, patient can gradually return to full training activity, as tolerated. |
Information from Khan KM, Brukner PD, Kearney C, Fuller PJ, Bradshaw CJ, Kiss ZS. Tarsal navicular stress fracture in athletes. Sports Med 1994;17:65–76.
Radiologic follow-up is not useful in most circumstances. Plain films are not sensitive enough to detect subtle changes in most cases and delay the clinical picture significantly.1,25 Bone scans can remain abnormal for up to two years after clinical healing.15 CT scanning can reveal significant loss of bony mineralization, sclerosis, notching of the proximal articular surface, and subchondral cysts even in a normal healing fracture; it is, therefore, unreliable as a guide to follow-up.1,5,13,15,24,25
Analysis of the patient's gait with orthotic evaluation is prudent to help compensate for any underlying biomechanical abnormality and prevent recurrence.1,15,19,25 It is important to address any training abnormalities, nutritional concerns, flexibility issues, or other factors that may place the athlete at risk of recurrence.
OPERATIVE TREATMENT
Indications for operative intervention include the rare displaced fracture, fragmentation, and failure of conservative therapy leading to delayed union or nonunion.14,18,24 Some physicians advocate more aggressive surgical treatment for type III complete fractures.30 Surgical intervention also may be indicated in athletes who need quick healing to allow them to return to play. The average time for athletes to return to play after surgical intervention compared with conservative management using a non–weight-bearing cast is 3.8 months and 5.6 months, respectively.1 Typically, surgical intervention consists of screw fixation, with possible bone graft inlay.

