Glaucoma is the second most common cause of legal blindness in the United States. Open-angle glaucoma is an asymptomatic, progressive optic neuropathy characterized by enlarging optic disc cupping and visual field loss. Patients at increased risk for open-angle glaucoma include blacks older than 40 years, whites older than 65 years, and persons with a family history of glaucoma or a personal history of diabetes or severe myopia. Elevated intraocular pressure is a strong, modifiable risk factor for open-angle glaucoma, but it is not diagnostic. Some patients with glaucoma have normal intraocular pressure (i.e., normal-pressure glaucoma), and many patients with elevated intraocular pressure do not have glaucoma (i.e., glaucoma suspects). Routine measurement of intraocular pressure by primary care physicians to screen patients for glaucoma is not recommended. Open-angle glaucoma usually is discovered during an adult eye evaluation performed for other indications. Final diagnosis and treatment occur in collaboration with ophthalmologists and optometrists. Formal visual field testing (perimetry) is a mainstay of glaucoma diagnosis and management. Eye drops, commonly nonspecific beta-blocker or prostaglandin analog drops, generally are the first-line treatment to reduce intraocular pressure. Laser treatment and surgery usually are reserved for patients in whom medical treatment has failed. Without treatment, open-angle glaucoma can end in irreversible vision loss.
Glaucoma is a leading cause of blindness and vision impairment. It affects approximately 2.5 million persons in the United States, including 3 percent of persons older than 55 years, although about one half are unaware that they have the disease.1,2 Glaucoma is the second most common cause of legal blindness in the United States and the leading cause of legal blindness among blacks.2 About 120,000 Americans are blind as a result of glaucoma, at a cost of about $1.5 billion per year in benefits, lost tax revenues, and health expenses.
Pathophysiologically, glaucoma is a progressive optic nerve disease often associated with elevated intraocular pressure and characterized by optic disc cupping and visual field loss. Vision loss from glaucoma is asymptomatic and irreversible.1 Aqueous is a clear fluid that fills the anterior and posterior chambers of the eye. Aqueous is produced by the ciliary body, passes through the pupil, and drains through the trabecular meshwork (Figure 1). Impaired outflow of aqueous humor causes elevated intraocular pressure. In open-angle glaucoma, impaired outflow results from dysfunction of the drainage system.3 In angle-closure glaucoma, impaired outflow results from occlusion of the anterior chamber angle itself, impairing access of the aqueous to the drainage system3 (Figure 1).
Open-Angle Glaucoma
Open-angle glaucoma is a progressive optic neuropathy characterized by acquired loss of retinal ganglion cells and atrophy of the optic nerve.2 It accounts for more than 90 percent of cases of glaucoma in the United States.4 Elevated intraocular pressure is the major risk factor for open-angle glaucoma, but it is not a diagnostic factor.2,5–7 The rate at which patients with elevated intraocular pressure develop glaucomatous optic nerve damage is approximately 1 percent per year.8 The pathophysiology of open-angle glaucoma includes a progressive decrease in the number of retinal ganglion cells when nerve fibers at the point where the optic nerve exits the eye become pinched and die. This condition leads to thinning of the neural rim and progressive enlargement of the optic nerve cup. The loss of nerve fibers causes a permanently decreased visual field.9
FIGURE 1.
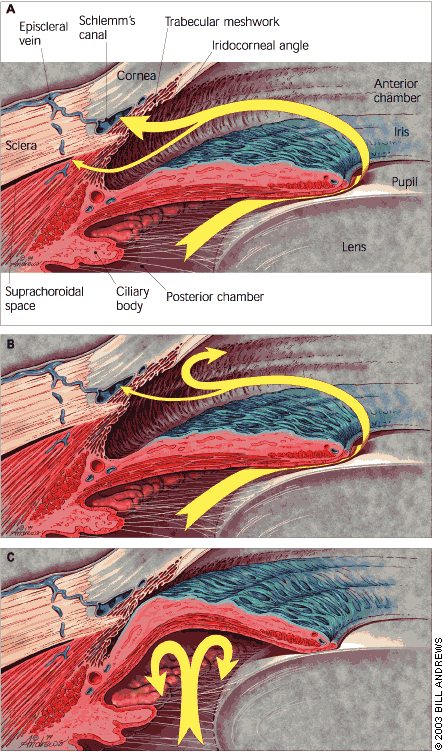
Normal and abnormal aqueous humor outflow. (A) Normal outflow through trabecular meshwork (large arrow) and uveoscleral routes (small arrow) and related anatomy. Most aqueous flow is through the trabecular meshwork. Each pathway is drained by the eye's venous circulation. (B) In primary open-angle glaucoma, aqueous outflow by these pathways is diminished. (C) In angle-closure glaucoma, the iris is abnormally positioned so as to block aqueous outflow through the anterior chamber (iridocorneal) angle.
However, more than two thirds of patients with elevated intraocular pressure (i.e., pressure greater than 21 mm Hg) do not lose visual field or develop optic nerve cupping.1,2 These patients, who do not have glaucoma, are referred to as “glaucoma suspects.”
Conversely, about 15 percent of patients with otherwise characteristic glaucomatous nerve damage have a consistently normal intraocular pressure (i.e., 21 mm Hg or less).1,2 These patients have normal-pressure glaucoma.
The only known causative risk factors for open-angle glaucoma are elevated intraocular pressure and insults to the eye, including trauma, uveitis, and steroid therapy. While steroid therapy of any kind may contribute to elevated intraocular pressure,4 topical eye and periocular steroids seem most likely to increase pressure.3 Associated risk factors, in addition to age, include black race, which increases the prevalence of glaucoma by a factor of four, and a positive first-degree family history, which increases the prevalence by a factor of seven.1,2,4,10
SYMPTOMS
Rarely do patients with glaucoma have symptoms.4,11 After loss of more than 40 percent of the nerve fibers, patients may notice a gradual loss of peripheral vision, or “tunnel vision.”2,4,9,10 Open-angle glaucoma usually is an incidental finding during an adult eye evaluation performed for other indications.2
PHYSICAL FINDINGS
If glaucoma is suspected because of a patient's complaint or risk factors, the family physician should perform direct ophthalmoscopy on both eyes, concentrating on the optic disc, before possible referral (Figure 2). Indeed, some physicians recommend that direct ophthalmoscopy be part of every adult complete physical examination. Optic disc findings frequently are noted before visual field deficits appear.2,12 Diagnostic findings include a symmetrically enlarged cup-to-disc ratio greater than 0.53 (Figures 3 and 4), cup-to-disc ratio asymmetry between the two eyes of 0.2 or more,2,4 or a highly asymmetric cup in one eye.10
Open-angle glaucoma generally is a bilateral disease, although it often is asymmetric.2,4 Damage in one eye significantly increases the risk of subsequent damage in the other eye.2 Progressive optic nerve cupping is a manifestation of progressive optic nerve death12 and uncontrolled glaucoma. Definitive perimetric (visual field) evidence (Figure 5), detailed ophthalmoscopic evidence, or both confirm the diagnosis of open-angle glaucoma.10
FIGURE 2.
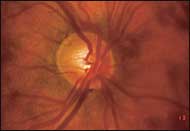
Normal optic disc. Note the distinct optic disc margins, the well-demarcated cup, and the healthy pink color of the neuroretinal rim.
CLINICAL INDICATIONS FOR REFERRAL
Primary care physicians should refer patients with risk factors or findings suggestive of glaucoma to eye specialists for a comprehensive eye examination, including perimetry (Table 1).2,10,13 Perimetry, a computer-based test that provides a printout of the visual fields, is a mainstay of glaucoma diagnosis and management5,10 (Figure 5). Because open-angle glaucoma is a chronic disease, these printouts are tracked over the long term.
FIGURE 3.
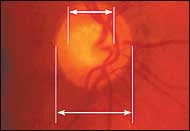
The cup-to-disc ratio of this optic nerve is approximately 0.6. Clinical correlation with the patient's history and examination is required to decide if this optic nerve is abnormal.
FIGURE 4.
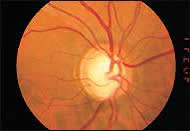
Glaucomatous optic nerve cupping. The cup in this optic nerve is enlarged to 0.8, and there is typical thinning of the inferior neuroretinal rim, forming a “notch.”
TABLE 1 Glaucoma: Clinical Indications for Referral to an Ophthalmologist or Optometrist
| Purpose of referral | Clinical indications |
|---|---|
| Screening a high-risk patient | Blacks older than 40 years |
| Whites older than 65 years | |
| Family history of glaucoma | |
| Personal history of diabetes or severe myopia | |
| Repeat screening at undetermined intervals | |
| Evaluating a patient with findings suggestive of glaucoma | Elevated pressure on tonometry, if performed |
| Suspicious optic disc cupping (cup-to-disc ratio greater than 0.5) | |
| Optic disc ratio difference between discs of 0.2 or more | |
| Highly asymmetric cup in one eye | |
| Apparently abnormal visual fields by confrontation |
FIGURE 5.
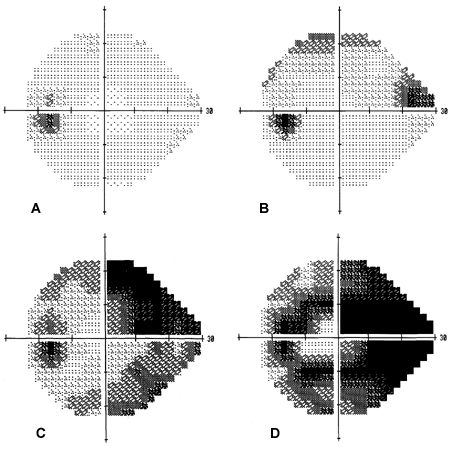
Computerized visual field analysis demonstrating progressive visual field loss in the left eye of a patient with uncontrolled glaucoma. (A) The first visual field is normal and shows the location of the blind spot created by the optic nerve. (B) The first visual field abnormality in this patient is loss of visual field in the superior and nasal portion of the visual field. (C) As damage progresses, visual field loss extends to involve both the superior and inferior portions of vision. (D) Finally, in advanced disease, extensive damage to the entire visual field occurs, sparing the very central portion of vision.
A painful red eye suggests acute angle-closure glaucoma, a medical emergency. Acute angle-closure glaucoma differs from open-angle glaucoma. In acute angle-closure glaucoma, the peripheral iris occludes the anterior chamber angle, blocking aqueous outflow. The intraocular pressure rises rapidly. Permanent vision loss from ocular ischemia may occur within hours as the intraocular pressure approaches the central retinal artery systolic blood pressure.3,10,14–16 Acute angle-closure glaucoma may present with constitutional symptoms, such as headache, nausea, vomiting, or malaise, that can obscure the diagnosis.10,11,17–19
A unilateral red eye associated with vomiting is considered acute angle-closure glaucoma until proved otherwise.15,20 All patients with suspected angle-closure glaucoma should be referred immediately for ophthalmologic consultation for confirmation of the diagnosis and evaluation for definitive operative treatment.3,4,10,16,17,21 Medical therapy for angle-closure glaucoma is not an appropriate substitute for laser iridotomy or iridectomy.17
SCREENING
Routine measurement of intraocular pressure by primary care physicians to screen patients for glaucoma is not recommended.13 [Evidence level C, consensus/expert guidelines] One half of patients with open-angle glaucoma have normal pressure at a single measurement.2 Intraocular pressure, the optic nerve, and perimetry provide complementary clues but are not easily combined in population-based screening programs.2
The U.S. Preventive Services Task Force recommends that eye specialists, at undetermined intervals, screen the following persons for glaucoma: blacks older than 40 years, whites older than 65 years, and patients with a family history of glaucoma or a personal history of diabetes or severe myopia (Table 1).2,10,13
According to the American Academy of Ophthalmology, screening for glaucoma as part of an eye professional's comprehensive adult eye evaluation is the most effective way of diagnosing glaucoma.2
MEDICAL TREATMENT
Intraocular pressure is the only treatable risk factor for open-angle glaucoma.2,3 However, outcome data have been lacking that unequivocally link lowering of intraocular pressure with preserving vision.22 A recent randomized trial23 showed that topical ocular hypotensive medication was effective in delaying or preventing the onset of open-angle glaucoma in patients with elevated intraocular pressure. Two recent trials24,25 showed that lowering intraocular pressure decreased glaucoma progression. [Evidence level A, randomized controlled trials (RCTs)] The overall goal of glaucoma therapy is to preserve vision as documented by perimetry without untoward effects from therapy, not simply to lower the intraocular pressure.2,10
NONPHARMACOLOGIC MANAGEMENT
Regular aerobic exercise can help lower intraocular pressure.6 When patients with glaucoma understand the possibility of asymptomatic and irreversible vision loss, their compliance with treatment is likely to improve.2
PHARMACOLOGIC MANAGEMENT
The appropriateness of medical versus surgical versus laser treatment as optimal first-line therapy for open-angle glaucoma is the subject of current long-term RCTs.2,4
When initiating treatment, the eye specialist presumes that pretreatment intraocular pressure contributed to the optic nerve damage and may cause additional future damage.2 Consequently, the eye specialist establishes a pressure at which further optic nerve damage is likely to stop—the target intraocular pressure—generally a level 20 to 40 percent below the pretreatment pressure. The specific number depends on the level of pressure and the degree of optic nerve and visual field damage at diagnosis.2,26 The more advanced the damage, the lower the target pressure.2,26 The target pressure may even be below normal intraocular pressure in a patient without glaucoma. The initial target pressure is an estimate subject to change and a means toward the ultimate goal of preserving vision.2
Eye drops generally are the initial treatment of choice1,2,5,26 (Table 2).27 They reduce intraocular pressure by decreasing aqueous production or increasing aqueous outflow.5
To decrease aqueous production, a nonspecific beta-blocker eye drop such as levobunolol (Betagan) was, until recently, the first drug class to be chosen. Levobunolol is the least expensive agent and often can be used once daily. If the patient is already taking a systemic beta blocker, the physician should consider using another class of eye drops, because systemic beta-blocker therapy may reduce the ocular hypotensive effects of beta-blocker eye drops.28 [Evidence level A, RCT]
Physicians should avoid using beta-blocker eye drops in patients with reactive airway disease, cardiac conduction defects, or heart failure.3,4 Systemic absorption of these eye drops can cause bradycardia,29 bronchospasm, and even fatalities from bronchospasm in patients with asthma and heart conditions.27
Betaxolol (Betoptic) is a relatively selective beta-blocker eye drop.3,10 Betaxolol may have a lower risk of systemic side effects, especially pulmonary, than the nonspecific beta blockers.3
TABLE 2 Agents for Management of Open-Angle Glaucoma
| Drug action/class | Agent | Strength (%) | Unit size | Dosing interval | Pregnancy category* | Cost (generic)† |
|---|---|---|---|---|---|---|
| Decrease aqueous production: beta blockers, nonspecific | Levobunolol (Betagan Liquifilm) | 0.250 | 10 mL | Twice a day | C | $ 48.50 (27.00) |
| Levobunolol | 0.500 | 5 mL | Once or twice a day | C | 29.50 (9.50) | |
| Carteolol (Ocupress) | 1.000 | 10 mL | Twice a day | C | 56.00 (37.00) | |
| Metipranolol (Optipranolol) | 0.300 | 10 mL | Twice a day | C | 30.00 (27.00) | |
| Timolol maleate (Timoptic) | 0.250 | 10 mL | Twice a day | C | 36.00 (11.00) | |
| Timolol maleate | 0.500 | 10 mL | Twice a day | C | 43.00 (12.00) | |
| Timolol maleate (XE: solution or gel) | 0.250 | 5 mL | Once a day | C | 29.00 (26.00) | |
| Timolol maleate (XE: solution or gel) | 0.500 | 5 mL | Once a day | C | 34.50 (31.00) | |
| Decrease aqueous production: beta blockers, relatively specific | Betaxolol (Betoptic) | 0.500 | 10 mL | Twice a day | C | 58.00 (NA) |
| Decrease aqueous production: carbonic anhydrase inhibitors | Dorzolamide (Trusopt) | 2.000 | 10 mL | Three times a day | C | 54.00 (NA) |
| Brinzolamide (Azopt) | 1.000 | 10 mL | Three times a day | C | 53.00 (NA) | |
| Decrease aqueous production, may increase outflow: alpha agonists | Apraclonidine (Iopidine) | 0.500 | 5 mL | Three times a day | C | 57.50 (NA) |
| Apraclonidine | 1.000 | 24 0.1-mL unit-dose packets | Three times a day | C | 229.00 (NA) | |
| Brimonidine tartrate (Alphagan) | 0.200 | 10 mL | Three times a day | B | 72.50 (NA) | |
| Brimonidine tartrate (purite preserved; Alphagan P) | 0.150 | 15 mL | Three times a day | B | 95.00 (NA) | |
| Increase aqueous outflow: prostaglandin analogs | Latanoprost (Xalatan) | 0.005 | 2.5 mL | Once a day | C | 50.00 (NA) |
| Bimatoprost (Lumigan) | 0.030 | 5 mL | Once a day | C | 100.00 (NA) | |
| Travoprost (Travatan) | 0.004 | 2.5 mL | Once a day | C | 45.50 (NA) | |
| Unoprostone (Rescula) | 0.150 | 5 mL | Twice a day | C | 47.00 (NA) | |
| Increase aqueous outflow: parasympathomimetic | Pilocarpine (Pilocar) | 1.000 | 15 mL | Three to four times a day | C | 5.00 (3.50) |
| Pilocarpine (Pilocar) | 2.000 | 15 mL | Three to four times a day | C | 7.00 (4.50) | |
| Increase aqueous outflow: sympathomimetic | Dipivefrin (Propine) | 0.100 | 10 mL | Twice a day | B | 45.50 (10.00) |
| Combination medications | Dorzolamide and timolol maleate (Cosopt) | 2/0.5 | 10 mL | Twice a day | C | 92.00 (NA) |
NA = not applicable.
*—Information on pregnancy categories from Drug facts and comparisons 2002. 56th ed. St. Louis: Facts & Comparisons, 2002:1875. Regardless of the designated pregnancy category or presumed safety, no drug should be administered during pregnancy unless it is clearly needed and potential benefits outweigh potential hazards to the fetus.
A—Controlled studies show no risk. Adequate, well-controlled studies in pregnant women have failed to demonstrate risk to the fetus.
B—No evidence of risk in humans. Either animal findings show risk, but human findings do not; or if no adequate human studies have been done, animal findings are negative.
C—Risk cannot be ruled out. Human studies are lacking, and animal studies are either positive for fetal risk or lacking. However, potential benefits may justify the potential risks.
D—Positive evidence of risk. Investigational or post-marketing data show risk to the fetus. Nevertheless, potential benefits may outweigh the potential risks. If needed in a life-threatening situation or serious disease, the drug may be acceptable if safer drugs cannot be used or are ineffective.
X—Contraindicated in pregnancy. Studies in animal or human, or investigational or post-marketing reports have shown fetal risk that clearly outweighs any possible benefit to the patient.
†—Estimated cost to the pharmacist based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 2001. Cost to the patient will be higher, depending on prescription filling fee. Costs rounded to the nearest half dollar.
When one particular drug does not help the patient to reach the target pressure level, physicians can change to a different class or add a second medication from a different class. A multiple drug regimen may be necessary to adequately control glaucoma. The topical carbonic anhydrase inhibitors, dorzolamide (Tru-sopt) and brinzolamide (Azopt), often are used as adjunctive therapy30 but rarely as initial therapy. A dorzolamide and timolol maleate combination (Cosopt) is available to decrease aqueous production by two different mechanisms in a single agent if monotherapy fails.26,31 [Reference31—Evidence level A, RCT]
Apraclonidine (Iopidine) and brimonidine tartrate (Alphagan) are alpha agonists, effective as adjunctive or, occasionally, primary therapy.29 Both may increase aqueous outflow in addition to decreasing aqueous production. Local ocular allergy often limits the usefulness of apraclonidine.4
Several classes of eye drops increase aqueous outflow. Latanoprost (Xalatan), a topical prostaglandin analog, is now the most frequently prescribed glaucoma medication in the world. The U.S. Food and Drug Administration recently approved latanoprost as a once-daily eyedrop for the initial treatment of elevated intraocular pressure associated with open-angle glaucoma or ocular hypertension because it proved beneficial in randomized studies.32 It is used alone or in addition to other agents. Latanoprost is highly effective with a much lower risk of systemic side effects than beta blockers but also is more expensive.33,34 [Reference33—Evidence level A, meta-analysis. Reference34—Evidence level A, RCT] Because it is instilled once daily, latanoprost is especially appropriate in non-compliant patients. Latanoprost can increase eyelash growth, and iris and eyelash pigmentation.4,9,34 Most other prostaglandin analog drugs similar to latanoprost also are used once daily. Pilocarpine (Pilocar), a parasympathomimetic miotic, was once a mainstay of treatment. Ocular side effects and multiple dosing limit its use.4 Dipivefrin (Propine), a sympathomimetic drug,5,6 is rarely used these days.
Patients using eye drops should be taught application techniques to decrease systemic absorption and systemic side effects.2 (See patient information handout, p. 1950.)
SURGICAL MANAGEMENT
Laser trabeculoplasty or surgical trabeculectomy is indicated when the target intraocular pressure cannot be reached medically,2 when optic nerve damage progresses despite achieving intraocular pressure goals with maximal medical therapy, or when the patient is unable to comply with or tolerate medical therapy.1 Laser trabeculoplasty and trabeculectomy are outpatient surgical procedures. Laser trabeculoplasty is also an effective first-line therapy when cost, inconvenience, or the high risk of side effects makes medical therapy unacceptable to patients. Trabeculectomy carries important visual risks and usually is reserved for use in patients in whom medical and laser-treatment options have failed. Long-term RCTs are underway to evaluate surgical and laser procedures as routine primary treatment instead of medication.2,4

