This is a corrected version of the article that appeared in print.
Am Fam Physician. 2003;67(10):2165-2172
Infertility affects 15 percent of couples, and 50 percent of male infertility is potentially correctable. Evaluation of the subfertile man requires a complete medical history, physical examination, and laboratory studies. The main purpose of the male evaluation is to identify and treat correctable causes of subfertility. In addition, many men seek an explanation for their condition, which can be discovered during their evaluation. Furthermore, the male fertility evaluation can uncover significant medical and genetic pathology that could affect the patient's health or that of his offspring. Although pregnancies can be achieved without any evaluation other than a semen analysis, this test alone is insufficient to adequately evaluate the male patient. Treatment of correctable male-factor pathology is cost effective, does not increase the risk of multiple births, and can spare the woman invasive procedures and potential complications associated with assisted reproductive technologies. Appropriate evaluation and treatment of the subfertile man are critical in delivering suitable care to the infertile couple.
Infertility, defined as the inability to conceive after one year of unprotected intercourse, affects 15 percent of couples. It is not unreasonable, however, to begin a fertility evaluation sooner than one year if the woman's age is a factor and delaying the evaluation could further diminish the couple's chance of conceiving. Male subfertility is one of the most rapidly growing fields in medicine, with dramatic advances in diagnosis and treatment.
Although infertility (or subfertility) is often attributed to female causes, fertility is a two-person phenomenon. Successful conception depends on many complicated events, including satisfactory sexual and ejaculatory function, appropriate timing, and a complex set of interactions between the male and the female reproductive tracts. Male and female factors coexist in about one third of cases, while one third of cases are secondary to male factors only.1 Therefore, evaluation of both partners is critical, and the woman's gynecologic evaluation should proceed simultaneously with the man's.
Causes of Male Subfertility
The most common identifiable cause of male subfertility is a varicocele, a condition of palpably distended veins of the pampiniform plexus of the spermatic cord.1,2 The term “subclinical varicocele” refers to a lesion too small to be detected by physical examination. The concept of a subclinical varicocele arose from the observation in early reports that the detrimental effect of small varicoceles equaled that of larger varicoceles. However, more recent studies suggest that larger varicoceles have a greater impact on fertility.3 As a result, most subspecialists who deal with male subfertility do not regard subclinical varicoceles as clinically significant.
Another common correctable cause of male subfertility is obstruction, which may occur after a vasectomy.1,4 Less common correctable causes include ejaculatory dysfunction, infection, medications, and hormonal deficiency (Table 1).1 When the sum of these correctable causes is calculated, it becomes apparent that more than one half of cases of male subfertility are potentially correctable.
In addition, specific corrective treatments such as vasectomy reversal and varicocele ligation are more cost effective than empiric treatment with assisted reproductive technologies.5–7 Furthermore, correction of underlying male factors can allow for natural conception, does not carry an increased risk of multiple births, and spares the woman invasive procedures and the potential complications of these therapies.8
Recent advances, particularly in molecular genetics, have improved our understanding of some forms of male subfertility. A significant proportion of male subfertility currently is unexplained. About 13 percent of men with nonobstructive azoospermia (i.e., no sperm in the semen because of low or absent sperm production) have been shown to have Y-chromosome microdeletions, and about 70 percent of men with congenital bilateral absence of the vas deferens are carriers of cystic fibrosis mutations.9–13
The most significant advance in the treatment of severe male infertility is in vitro fertilization with intracytoplasmic sperm injection (ICSI).14 With this technique, a single sperm is injected directly into the oocyte, circumventing severely decreased sperm quantity or quality. Only one viable sperm per egg is required for ICSI, and a precise diagnosis is not required to achieve conception.15
When using sperm from men with known or presumed genetic infertility, it must be assumed that any male offspring also will be infertile. Y-chromosome microdeletions from the father are inherited by the sons when ICSI is used.16,17 Counseling about these potential genetic issues is a critical part of the male fertility evaluation. Because ICSI bypasses normal natural selection, concerns have been raised about the offspring. There does not appear to be an increased risk of major malformations in children born from ICSI compared with the general population.18
Evaluation
The main goals of evaluating the subfertile man are to identify correctable causes of infertility and to help him and his partner conceive by the most natural, least invasive means possible. In addition, the evaluation may uncover significant underlying medical or genetic pathology. Subfertility may be related to an underlying malignancy, such as a testicular or pituitary tumor. Other medical problems that significantly affect the health of the man or his offspring may be discovered.19,20 If the only evaluation is a semen analysis, underlying pathology can be missed. Finally, many men seek an explanation for their condition, of which they may be deprived without a complete evaluation.21
History and Physical Examination
A careful history can offer clues to the underlying cause of infertility and provide an assessment of the man's fertility potential. The duration of the infertility, previous evaluation and treatment, previous pregnancies (for either partner), and any difficulty establishing these pregnancies should be documented. Inadequate frequency or timing of intercourse, sexual dysfunction, and lubricant use can impede pregnancy. The optimal frequency of intercourse is every day or every other day around the expected time of ovulation.22 [Evidence level B, uncontrolled clinical trial] Because nearly all commercially available lubricants are spermatotoxic, their use is discouraged.1
Most men of reproductive age do not have a significant medical history, but some specific risk factors may be identified. For example, diabetes mellitus can cause erectile and ejaculatory dysfunction. Previous disorders of the testes, such as cryptorchidism or spermatic cord torsion, or a history of inguinal, scrotal, or retroperitoneal surgery, are associated with subfertility. Use of prescription or illicit drugs and exposure to environmental toxins also can impair fertility (Table 2).1,23,24 In the review of systems, anosmia may suggest an underlying hypothalamic etiology (such as Kallmann's syndrome) or a pituitary etiology, while frequent respiratory infections are a feature of Young's syndrome (e.g., chronic sinusitis, bronchiectasis, obstructive azoospermia) and Kartagener's syndrome (e.g., primary ciliary dyskinesia/immotile cilia, chronic sinusitis, bronchiectasis, situs inversus). Headaches, visual field disturbances, or galactorrhea should prompt an investigation for a tumor of the central nervous system1 (Table 3).
A thorough examination can identify underlying causes of subfertility. Abnormal distribution of hair and fat can suggest an underlying endocrinopathy, such as hypogonadotropic hypogonadism. The position and size of the urethral meatus should be noted because severe hypospadias can impair sperm deposition near the cervix. [ corrected] Normal testes are 20 cm3 or more, or at least 4 cm in greatest dimension. Those smaller than 20 cm3 are suggestive of decreased sperm production and may occur in hypogonadal men as well.1 The presence of the vasa deferentia and epididymides as well as any induration or engorgement suggestive of obstruction should be noted.
Varicoceles are found most commonly on the left side, but up to 20 percent may be bilateral. Diagnosis should be made in a warm room by palpation of the spermatic cord with the patient in the standing position. Varicoceles are graded 1+ (palpable with Valsalva's maneuver only), 2+ (palpable), and 3+ (visible through the scrotal skin). An isolated right-sided varicocele or a lesion on either side that does not disappear when the patient assumes the supine position should prompt imaging of the retroperitoneum to evaluate for inferior vena caval or renal vein obstruction. Digital rectal examination is performed to examine the prostate gland, seminal vesicles, and possible cysts that can cause ejaculatory duct obstruction. Table 3 presents clinical clues to and possible diagnoses of male infertility.
| Clinical clue | Possible diagnosis |
|---|---|
| History | |
| Infrequent intercourse, lubricant use | Coital factors causing infertility |
| Irregular or infrequent menses | Ovulatory dysfunction/female factors |
| Difficulty obtaining erection | Erectile dysfunction |
| Decreased libido | Hypogonadism |
| Blood or pain with ejaculation | Prostatitis/leukocytospermia |
| Small-volume or absent ejaculate | Retrograde ejaculation |
| Anabolic steroid use | Pituitary suppression/secondary hypogonadism |
| Cryptorchidism | Oligospermia or testicular failure |
| History of testicular torsion | Oligospermia or testicular failure |
| Previous chemotherapy | Oligospermia or testicular failure |
| Previous retroperitoneal surgery | Ejaculatory dysfunction |
| Previous inguinal or scrotal surgery | Excurrent* duct obstruction |
| Headache, visual changes | Pituitary tumor |
| Anosmia | Kallmann's syndrome |
| Frequent respiratory infections | Young's or Kartagener's syndrome |
| Patient reports scrotal mass | Testicular tumor or varicocele |
| Physical examination | |
| Decreased virilization or abnormal hair distribution, gynecomastia | Hypogonadism, primary or secondary |
| Palpably dilated veins of spermatic cord | Varicocele |
| Small testes | Testicular failure |
| Impalpable vas deferens | Congenital absence of the vas deferens |
| Inguinal scars | Unreported inguinal surgery, possible excurrent duct obstruction |
| Prostate nodule | Prostate cancer |
| Other prostate mass on rectal examination | Ejaculatory duct abnormality |
| Laboratory testing | |
| Low semen volume | Ejaculatory dysfunction, incomplete specimen collection |
| Azoospermia | Obstruction vs. testicular failure (see Figure 2) |
| Leukocytospermia | Prostatitis |
| Oligospermia, asthenospermia (low motility) | Possible varicocele |
| Sperm in post-ejaculatory urine | Retrograde ejaculation |
| Elevated FSH level | Testicular failure |
| Low testosterone, low FSH level | Hypogonadotropic hypogonadism |
Laboratory Evaluation
The semen analysis is the foundation of the laboratory evaluation. At least two samples, preferably taken at least two or three weeks apart, should be analyzed after two to three days of sexual abstinence. The sample should be collected by masturbation in a clean container and analyzed within one hour of collection. The sample can be collected at home if it is kept at body temperature and brought to the laboratory in sufficient time.25
Leukocytospermia, which is defined as more than 1 million white blood cells per mL of semen, requires specific testing. It is not possible to definitively identify these cells by microscopic appearance alone.27 In the presence of significant leukocytospermia, empiric antibiotic therapy is reasonable. Doxycycline (Vibramycin), in a dosage of 100 mg twice a day for two weeks, is an effective regimen. A repeat semen analysis should be performed at the completion of therapy.
The semen analysis does not test fertility, but rather fertility potential. The chance of initiating a pregnancy correlates with the total number of moving sperm.28 Pregnancies can be established with subnormal parameters, illustrating the importance of the female partner's fertility potential and the fact that an abnormal semen analysis cannot be equated with subfertility. The morphology is a measurement of the percentage of the normal-shaped sperm. The Kruger or strict morphology score has been correlated with decreased success with fertilization in vitro. The significance of this finding in estimating the chance for natural conception is less clear. As with any other single semen parameter, it cannot be used in an absolute way to predict fertility.29
A semen analysis does not assess sperm function. Specialized testing is available to evaluate this factor, and the test of choice varies among treatment centers. Most tests attempt to examine some component of sperm-oocyte interaction or fertilization. Interpretation of these test results can be difficult, and the question of whether the results influence clinical management is controversial. These tests generally are performed only in specialized fertility laboratories.
Male subfertility caused by correctable endocrinopathies is rare. As such, hormone testing for all subfertile men is not necessary. When sperm concentration is less than 10 million per mL, measurement of the serum testosterone and follicle-stimulating hormone (FSH) levels is indicated.30 The levels of serum testosterone and FSH are adequate to assess the pituitary-testicular axis in the majority of cases. If the total testosterone level is normal, no further endocrine testing is needed. If the total testosterone level is low, the serum luteinizing hormone and prolactin levels can be checked to evaluate for a pituitary cause.
When testosterone is merely borderline or only slightly low, supplementation should be avoided unless the man is significantly symptomatic (i.e., erectile dysfunction, markedly decreased energy level, lack of libido). Testosterone supplementation will actually lower the sperm concentration in such men because it can cause pituitary suppression of gonadotropins. In the setting of subfertility, the FSH level may be elevated, suggesting end-organ (testicular) failure. A low level may indicate an underlying FSH deficiency, such as occurs with hypogonadotropic hypogonadism (Table 3).
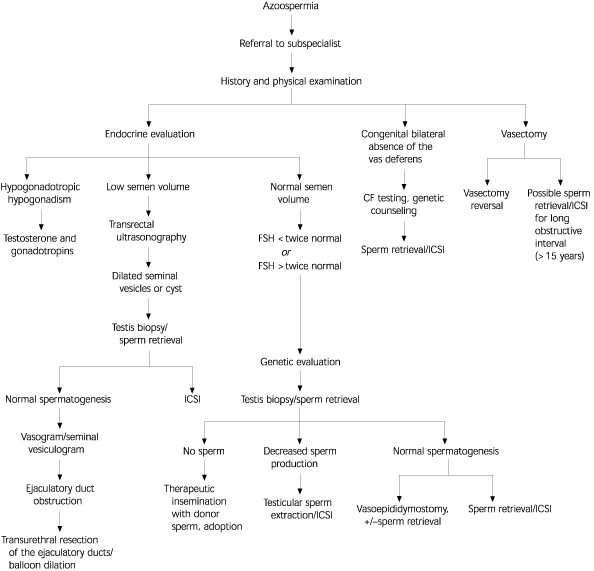
More specialized testing may be required based on the outcome of this initial evaluation (Figure 1). Most specialized testing requires referral to a center with clinical and laboratory expertise in the field of reproductive medicine. For example, men with low ejaculate volume (less than 1 mL) should have a post-ejaculatory urine sample analysis to rule out retrograde ejaculation. Azoospermic men can undergo testicular biopsy to evaluate the level of sperm production and differentiate between testicular failure and obstruction (i.e., normal sperm production). In patients with azoospermia, low semen volume (less than 1 mL), and a normal FSH level, transrectal ultrasonography is indicated to evaluate for possible ejaculatory duct obstruction31 (Figure 2).32
| Variable | Value |
|---|---|
| Volume | ≥2 mL |
| pH | ≥7.2 |
| Sperm concentration | ≥20 × 06 per mL |
| Motility | ≥50 percent (grade A and B) or 25 percent or more with progressive motility within 60 minutes of ejaculation |
| Morphology | > 50 percent normal* |
| White blood cells | < 1 × 106 per mL |
Genetic testing and counseling are indicated in specific instances. In patients with azoospermia or severe oligospermia, the karyotype should be determined because of the increased incidence of karyotypic abnormalities in this population.10 Men with congenital absence of the vas deferens, either unilateral or bilateral, may be carriers of cystic fibrosis. These men and their partners should undergo testing for cystic fibrosis mutations.11,12 These men also should have abdominal ultrasonography to check for renal agenesis.
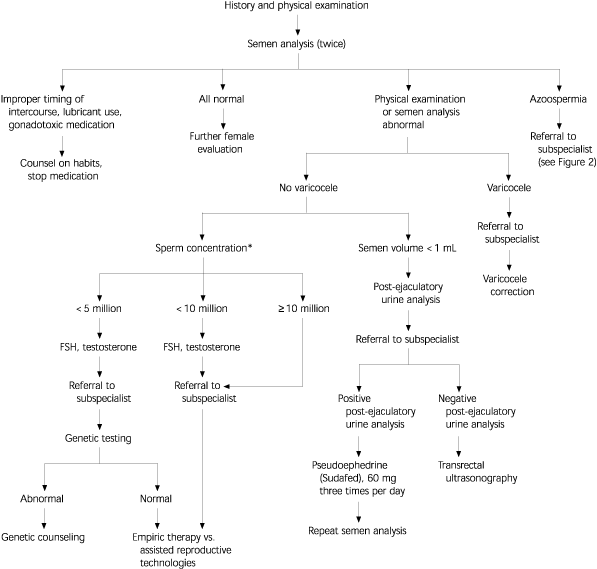
The outcome of the initial evaluation can help guide treatment. If correctable causes are found, specific corrective treatment is offered. If no correctable problem exists, the couple may wish to pursue treatment with assisted reproductive technologies such as intrauterine insemination.33 An alternative to assisted reproductive technologies is empiric treatment with clomiphene citrate (Serophene), although few convincing data show benefit.34 Some reproductive subspecialists advocate abandoning the male evaluation, with the exception of the semen analysis. Whether this is an efficient approach to conception is debatable, but denying the man an evaluation, including an opportunity to learn the cause of his problem and the chance for specific corrective therapy, seems inappropriate.20,21