The cooperation continues between the American Academy of Family Physicians (AAFP), the Advisory Committee on Immunization Practices (ACIP), and the American College of Obstetricians and Gynecologists (ACOG) with the publication of the Recommended Adult Immunization Schedule, United States, 2003–2004(Figures 1 and 2). This is the second year that a family physician has led the Working Group on Adult Immunizations at the ACIP (Richard D. Clover, M.D., led the group last year). The schedule, although similar to last year's schedule, has some changes in formatting, footnotes, and clarity of language. Annual updates to the schedule are planned.
One part of the schedule lists immunizations indicated by age, particularly influenza, tetanus and diphtheria toxoids, and pneumococcal polysaccharide vaccine. Fortunately, supplies for all of these vaccines are good this year. A new live, attenuated influenza vaccine (LAIV; FluMist) for healthy persons five to 49 years of age should be available in addition to the older inactivated vaccine. A major advantage of LAIV is that it is administered intranasally. LAIV contains cold-adapted viruses that do not replicate well in the lower airways. However, exacerbations of asthma were noted after vaccination in some age groups; thus, the safety of LAIV has not been established in persons with asthma. LAIV is contraindicated in immunodeficient persons. The efficacy is good in children 60 to 84 months of age and in adults 18 to 49 years of age; hence, LAIV is licensed for healthy persons five to 49 years of age; safety and efficacy have not been established in the elderly. Although the optimal influenza vaccination season is October and November, inactivated influenza vaccine or LAIV can be given December through March for those who were not vaccinated during the fall. Vaccine information statements for influenza vaccines as well as all other routine vaccines can be downloaded free fromhttp://www.cdc.gov/nip.
Several developments relate to pneumococcal polysaccharide vaccine. The payment by Medicare Part B for pneumococcal polysaccharide vaccine increased from $13.10 to $18.62, effective October 1, 2003. Billing information for Medicare for influenza and pneumococcal vaccinations can be obtained athttp://www.cms.hhs.gov/preventiveservices/2.asp. Influenza vaccination during the fall is a prime opportunity to assess pneumococcal polysaccharide vaccination status and check for indications for it. The Centers for Medicare and Medicaid Services now allow the use of standing orders in hospitals to give influenza and pneumococcal polysaccharide vaccines.
FIGURE 1. Recommended Adult Immunization Schedule by Age Group, United States, 2003–2004
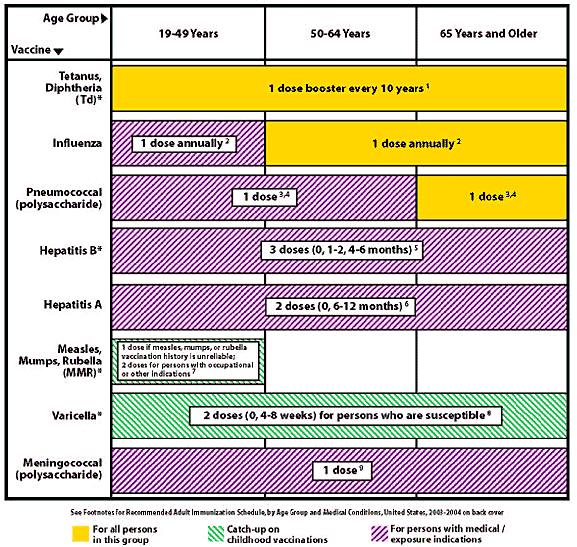
1. Tetanus and diphtheria (Td)—Adults, including pregnant women with uncertain histories of a complete primary vaccination series, should receive a primary series of Td. A primary series for adults is 3 doses: the first 2 doses given at least 4 weeks apart and the third dose, 6 to 12 months after the second. Administer 1 dose if the person had received the primary series and the last vaccination was 10 years ago or longer. Consult MMWR 1991;40(RR-10):1–21 for administering Td as prophylaxis in wound management. The American College of Physicians (ACP) Task Force on Adult Immunization supports a second option for Td use in adults: a single Td booster at 50 years of age for persons who have completed the full pediatric series, including the teenage/young adult booster. Guide for Adult Immunization. 3d ed. ACP 1994:20.
2. Influenza vaccination—Medical indications: chronic disorders of the cardiovascular or pulmonary systems including asthma; chronic metabolic diseases including diabetes mellitus, renal dysfunction, hemoglobinopathies, or immunosuppression (including immunosuppression caused by medications or by human immunodeficiency virus [HIV]), requiring regular medical follow-up or hospitalization during the preceding year; women who will be in the second or third trimester of pregnancy during the influenza season. Occupational indications: health care workers. Other indications: residents of nursing homes and other long-term care facilities; persons likely to transmit influenza to persons at high risk (in-home caregivers to persons with medical indications, household contacts and out-of-home caregivers of children birth to 23 months of age, or children with asthma or other indicator conditions for influenza vaccination, household members and care-givers of elderly and adults with high-risk conditions); and anyone who wishes to be vaccinated. For healthy persons 5 to 49 years of age without high-risk conditions, the inactivated vaccine or the intranasally administered influenza vaccine (FluMist) may be given. MMWR 2003;52(RR-8):1–36; MMWR 2003;53(RR13):1–8.
3. Pneumococcal polysaccharide vaccination—Medical indications: chronic disorders of the pulmonary system (excluding asthma), cardiovascular diseases, diabetes mellitus, chronic liver diseases including liver disease as a result of alcohol abuse (e.g., cirrhosis), chronic renal failure or nephrotic syndrome, functional or anatomic asplenia (e.g., sickle cell disease or splenectomy), immunosuppressive conditions (e.g., congenital immunodeficiency, HIV infection, leukemia, lymphoma, multiple myeloma, Hodgkin's disease, generalized malignancy, organ or bone marrow transplantation), chemotherapy with alkylating agents, antimetabolites, or long-term systemic corticosteroids. Geographic/ other indications: Alaskan Natives and certain American Indian populations. Other indications: residents of nursing homes and other long-term care facilities. MMWR 1997;46(RR-8):1–24.
4. Revaccination with pneumococcal polysaccharide vaccine—One-time revaccination after 5 years for persons with chronic renal failure or nephrotic syndrome, functional or anatomic asplenia (e.g., sickle cell disease or splenectomy), immunosuppressive conditions (e.g., congenital immunodeficiency, HIV infection, leukemia, lymphoma, multiple myeloma, Hodgkin's disease, generalized malignancy, organ or bone marrow transplantation), chemotherapy with alkylating agents, antimetabolites, or long-term systemic corticosteroids. In persons 65 years and older, one-time revaccination if they were vaccinated 5 or more years previously and were younger than 65 years at the time of primary vaccination. MMWR 1997;46(RR-8):1–24.
5. Hepatitis B vaccination—Medical indications: hemodialysis patients, patients who receive clotting-factor concentrates. Occupational indications: health care workers and public safety workers who have exposure to blood in the workplace, persons in training in schools of medicine, dentistry, nursing, laboratory technology, and other allied health professions. Behavior indications: injecting drug users, persons with more than one sex partner in the previous 6 months, persons with a recently acquired sexually transmitted disease (STD), all clients in STD clinics, men who have sex with men. Other indications: household contacts and sex partners of persons with chronic hepatitis B virus (HBV) infection, clients and staff of institutions for the developmentally disabled, international travelers who will be in countries with high or intermediate prevalence of chronic HBV infection for more than 6 months, inmates of correctional facilities. MMWR 1991;40(RR-13):1–19. (www.cdc.gov/travel/diseases/hbv.htm).
6. Hepatitis A vaccination—For the combined HepA-HepB vaccine, use 3 doses at 0, 1, and 6 months. Medical indications: persons with clotting-factor disorders or chronic liver disease. Behavior indications: men who have sex with men, users of injecting and noninjecting illegal drugs. Occupational indications: persons working with hepatitis A virus (HAV)-infected primates or with HAV in a research laboratory setting. Other indications: persons traveling to or working in countries that have high or intermediate endemicity of hepatitis A. MMWR 1999;48(RR-12):1–37. (www.cdc.gov/travel/diseases/hav.htm)
7. Measles, Mumps, Rubella vaccination (MMR)—Measles component: Adults born before 1957 may be considered immune to measles. Adults born in or after 1957 should receive at least one dose of MMR unless they have a medical contraindication, documentation of at least one dose, or other acceptable evidence of immunity. A second dose of MMR is recommended for adults who:
were recently exposed to measles or are in an outbreak settingwere previously vaccinated with killed measles vaccinewere vaccinated with an unknown vaccine between 1963 and 1967are students in post-secondary educational institutionswork in health care facilitiesplan to travel internationally
Mumps component: 1 dose of MMR should be adequate for protection. Rubella component: Give 1 dose of MMR to women whose rubella vaccination history is unreliable, and counsel women to avoid becoming pregnant for 4 weeks after vaccination. In women of child-bearing age, regardless of birth year, routinely determine rubella immunity and counsel women about congenital rubella syndrome. Do not vaccinate pregnant women or those planning to become pregnant in the next 4 weeks. If pregnant and susceptible, vaccinate as early in the postpartum period as possible. MMWR 1998;47(RR-8):1–57; MMWR 2001;50:1117.
8. Varicella vaccination—Recommended for all persons who do not have reliable clinical history of varicella infection or serologic evidence of varicella zoster virus (VZV) infection who may be at high risk for exposure or transmission. This includes health care workers and family contacts of immunocompromised persons, those who live or work in environments where transmission is likely (e.g., teachers of young children, day care employees, and residents and staff members in institutional settings), persons who live or work in environments where VZV transmission can occur (e.g., college students, inmates and staff members of correctional institutions, and military personnel), adolescents and adults living in households with children, women who are not pregnant but who may become pregnant in the future, international travelers who are not immune to infection. NOTE: Greater than 95 percent of U.S.-born adults are immune to VZV. Do not vaccinate pregnant women or those planning to become pregnant in the next 4 weeks. If pregnant and susceptible, vaccinate as early in the postpartum period as possible. MMWR 1996;45(RR-11):1–36; MMWR 1999;48(RR-6):1–5.
9. Meningococcal vaccine (quadrivalent polysaccharide for serogroups A, C, Y, and W-135)—Consider vaccination for persons with medical indications: adults with terminal complement component deficiencies, with anatomic or functional asplenia. Other indications: travelers to countries in which disease is hyperendemic or epidemic (“meningitis belt” of sub-Saharan Africa, Mecca, Saudi Arabia for Hajj). Revaccination at 3 to 5 years may be indicated for persons at high risk for infection (e.g., persons residing in areas in which disease is epidemic). Counsel college freshmen, especially those who live in dormitories, about meningococcal disease and the vaccine so that they can make an educated decision about receiving the vaccination. MMWR 2000;49(RR-7):1–20. NOTE: The AAFP recommends that colleges should take the lead on providing education on meningococcal infection and vaccination and offer it to those who are interested. Physicians need not initiate discussion of the meningococcal quadrivalent polysaccharide vaccine as part of routine medical care.
*—Covered by the National Vaccine Injury Compensation Program. For information on how to file a claim, call 800-338-2382. Please also visitwww.hrsa.gov/osp/vicp. To file a claim for vaccine injury, contact: U.S. Court of Federal Claims, 717 Madison Place, N.W., Washington, D.C. 20005, 202-219-9657.
This schedule indicates the recommended age groups for routine administration of currently licensed vaccines for persons 19 years and older. Licensed combination vaccines may be used whenever any components of the combination are indicated and the vaccine's other components are not contraindicated. Providers should consult the manufacturers' package inserts for detailed recommendations.
Report all clinically significant postvaccination reactions to the Vaccine Adverse Event Reporting System (VAERS). Reporting forms and instructions on filing a VAERS report are available by calling 800–822–7967 or from the VAERS Web site atvaers.hhs.gov.
For additional information about the vaccines listed above and contraindications for immunization, visit the National Immunization Program Web site atwww.cdc.gov/nip or call the National Immunization Hotline at 800-232-2522 (English) or 800-232-0233 (Spanish).
Approved by the Advisory Committee on Immunization Practices (ACIP), and accepted by the American College of Obstetricians and Gynecologists (ACOG) and the American Academy of Family Physicians (AAFP).
The other part of the schedule lists immunizations indicated and contraindicated by medical conditions. Because the influenza mortality rate is primarily determined by the number of high-risk conditions, influenza vaccination of high-risk persons and their close contacts is particularly important.
FIGURE 2. Recommended Immunizations for Adults by Medical Conditions, United States, 2003–2004
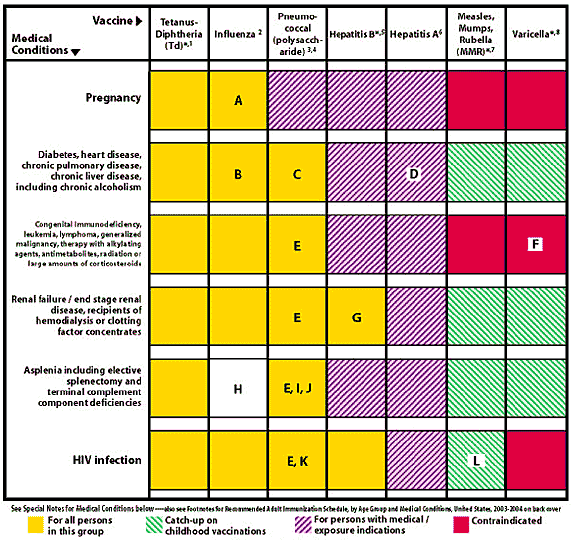
A. In women without chronic diseases or conditions, vaccinate if pregnancy will be at second or third trimester during influenza season. In women with chronic diseases or conditions, vaccinate at any time during the pregnancy.
B. Although chronic liver disease and alcoholism are not indicator conditions for influenza vaccination, give 1 dose annually if the patient is 50 years or older, has other indications for influenza vaccine, or if the patient requests vaccination.
C. Asthma is an indicator condition for influenza but not for pneumococcal vaccination.
D. In all persons with chronic liver disease.
E. In persons younger than 65 years, revaccinate once after 5 years or more have elapsed since initial vaccination.
F. Persons with impaired humoral immunity but intact cellular immunity may be vaccinated. MMWR 1999;48(RR-06):1–5.
G. Hemodialysis patients: Use special formulation of vaccine (40 mcg per mL) or two 1.0 mL 20 mcg doses given at one site. Vaccinate early in the course of renal disease. Assess antibody titers to hepatitis B surface antigen (anti-HBs) levels annually. Administer additional doses if anti-HBs levels decline to less than 10 milli-international units (mIU) per mL.
H. There are no data specifically on risk of severe or complicated influenza infections among persons with asplenia. However, influenza is a risk factor for secondary bacterial infections that may cause severe disease in persons with asplenia.
I. Administer meningococcal vaccine and consider Hib vaccine.
J. Elective splenectomy: vaccinate at least 2 weeks before surgery.
K. Vaccinate as close to diagnosis as possible when CD4+ cell counts are highest.
L. Withhold MMR or other measles-containing vaccines from HIV-infected persons with evidence of severe immunosuppression. MMWR 1998;47(RR-8):21–2; MMWR 2002;51(RR-02):22–4.
A key development this year was publication of the revised edition of the Standards for Adult Immunization Practices,1 which discusses ways to ensure optimal immunization of adults against vaccine-preventable diseases.
Information on immunizations by family physician leaders for family physicians can be found athttp://www.immunizationed.org, which includes free handheld personal digital assistant software in Palm and Windows formats and links to recent articles on immunization.2 Educational materials for case-based learning for residents and students can be found athttp://www.atpm.org/Immunization/TIME/body_time.html. Materials for offices about adult immunization can be found athttp://www.partnersforimmunization.org,http://www.immunize.org,http://www.cdc.gov/nip,http://www.nfid.org/ncai,http://www.immunizationinfo.org, andhttps://www.aafp.org.

