Almost one half of pregnancies in the United States are unintended. Primary reasons for the high rate of unplanned pregnancy include dissatisfaction with or underuse of effective contraceptive methods and poor compliance with contraceptive methods that require daily adherence. Several effective forms of contraception have become available in the United States within the past four years. The combined hormonal vaginal ring is inserted into the vagina for three weeks and then removed; after one ring-free week, a new ring is inserted. The contraceptive patch works in much the same way as oral contraceptive pills but requires only once-weekly application by the patient. A new intrauterine system that releases levonorgestrel provides the same contraception as traditional intrauterine devices but is associated with less menorrhagia and dysmenorrhea. The intrauterine system is highly effective and carries minimal risk of pelvic inflammatory disease. In providing counseling about contraception, the physician should consider the woman's preference and determine the likelihood of adherence to the regimen. In case of contraceptive failure, emergency contraception is effective.
Approximately 49 percent of pregnancies in the United States are unintended.1 One cost analysis2 found that compared with pregnancy and abortion, contraception saves an estimated $9,000 to $14,000 per woman of childbearing age over a five-year period. Although some methods of contraception have side effects, morbidity and mortality rates are significantly higher for pregnancy and childbirth than for the use of any contraceptive method alone.3
Oral contraceptive pills (OCPs), male condoms, and female sterilization are the contraceptive methods most commonly used in the United States.4 However, approximately 3 million U.S. women at risk for unintended pregnancy are using no form of contraception.5 Frequently cited reasons for discontinuing a method when contraception is still desired include side effects, difficulty of use, safety concerns, and lack of access to health care.5 Furthermore, personal beliefs and preferences influence a woman's willingness to use a contraceptive method correctly.
Family physicians provide contraceptive counseling to a significant number of patients each year. Knowledge of the wide variety of contraceptive methods and barriers to the use of some of these methods is important in helping each patient make an appropriate choice.
Traditional Methods of Contraception
The benefits, side effects, and potential complications of traditional methods of contraception are reviewed in detail elsewhere.6–8 A summary of these methods is provided in Table 1.8–11
It is important to note certain changes that have occurred in the past several years. For example, no contraceptive sponges currently are available in the United States, although a new vaginal sponge (Protectaid) is under investigation. Likewise, the levonorgestrel implant system marketed as Norplant is no longer being manufactured; however, a single-rod implant (Implanon), presently used outside the United States, may become available in the future.
TABLE 1 Traditional Contraceptive Methods
| Method | Side effects | Noncontraceptive benefits | Potential complications |
|---|---|---|---|
| Oral contraceptive pills | Nausea, headaches, dizziness, spotting, weight gain, breast tenderness, chloasma | Decreases menstrual pain, premenstrual syndrome, and blood loss; protects against symptomatic PID,8 some cancers (ovarian, endometrial), some benign tumors (leiomyomata, benign breast masses), and ovarian cysts; reduces acne. | Cardiovascular complications (stroke, heart attack, blood clots, high blood pressure), depression, hepatic adenomas; possible increased risk of breast and cervical cancers |
| IUD | Menstrual cramping, spotting, increased bleeding | Copper-containing IUDs may provide protection against endometrial cancer9; progesterone-releasing IUDs decrease menstrual blood loss and pain. | PID (following insertion), uterine perforation, anemia |
| Male condom | Decreased sensation, allergy to latex, loss of spontaneity | Protects against STDs, including HIV infection; delays premature ejaculation. | Anaphylactic reaction to latex |
| Female condom | Aesthetically unappealing; for some women, awkward to use | Protects against STDs. | None known |
| Hormonal implant | Tenderness at implant site, menstrual changes, hair loss, weight gain | Does not disturb lactation; may decrease menstrual cramps, pain, and blood loss. | Infection at implant site, complicated removal, depression |
| Progestin-only injections | Menstrual changes, weight gain, headaches, adverse effects on cholesterol levels (i.e., decreases high-density lipoprotein cholesterol level) | Does not disturb lactation; reduces risk of seizures; may have protective effects against PID, as well as ovarian and endometrial cancers. | Depression, allergic reactions, pathologic weight gain; possible bone loss |
| Female sterilization | Pain at surgical site; psychologic reactions, including regret that procedure was performed | Reduces risk of ovarian cancer; may protect against PID.10 | Infection, anesthetic complications; if pregnancy occurs after tubal ligation, high risk of ectopic pregnancy |
| Abstinence | Psychologic reactions | Prevents STDs, including HIV infection. | None known |
| Barrier contraceptives: diaphragm, cervical cap, vaginal sponge | Pelvic pressure, vaginal irritation, allergy; vaginal discharge if left in too long | Provides modest protection against some STDs. | Vaginal and urinary tract infections, toxic shock syndrome |
| Spermicides | Vaginal irritation, allergy | Provides modest protection against some STDs; potential increased risk of HIV infection with use of nonoxynol–911 | Vaginal and urinary tract infections |
| Lactational amenorrhea | Mastitis from staphylococcal infection | Breastfeeding has nutritional benefits for infant. | Increased risk of HIV transmission to infant if mother is HIV positive |
PID = pelvic inflammatory disease; IUD = intrauterine device; STD = sexually transmitted disease; HIV = human immunodeficiency virus.
A combination OCP that contains the new progestogen drospirenone (Yasmin) has been available for several years. Drospirenone has some antimineralocorticoid activity and has been shown to decrease the water retention, negative affect, and appetite changes that commonly are associated with menstrual cycle changes.12 [Strength of Recommendation (SOR) B, descriptive studies and lower quality clinical trials] Serum potassium levels should be monitored when women use this OCP in conjunction with other medicines that also raise potassium levels, because hyperkalemia is a potential side effect.
Finally, studies on the length of time that active pills are given have provided more options and flexibility for OCP users.13 The U.S. Food and Drug Administration (FDA) recently approved the use of levonorgestrelethinyl estradiol (Seasonale) in an extended OCP regimen consisting of 84 days of active pills and seven days of nonhormonal pills.
Newer Methods of Contraception
Traditional methods of contraception have some drawbacks (Table 1).8–11 Consequently, patient dissatisfaction and inconsistent or incorrect use may result in unintended pregnancy. For example, the success of OCPs, the most widely used reversible contraceptive method, is highly dependent on daily adherence. In one study,14 approximately 47 percent of OCP users failed to take one or more pills per cycle, and 22 percent failed to take two or more pills.
In designing the newer contraceptives, attention has been given to improving the side effect profiles of older methods and to developing delivery systems that do not require daily patient adherence.
VAGINAL CONTRACEPTIVE RING
The combined hormonal vaginal ring (NuvaRing) has a unique delivery system. The ring works in a similar manner as OCPs, but daily action by the patient is not required. The ring (Figure 1), which releases ethinyl estradiol at a rate of 15 mcg per day and etonogestrel at a rate of 120 mcg per day, is placed in the vagina for three weeks. If for some reason the ring is out of the vagina for more than three hours, back-up contraception should be used until the ring has been back in place for seven days. After three weeks, the ring is removed for one week, and a new ring is inserted. Withdrawal bleeding occurs during the ring-free week.
In a one-year study,15 the overall failure rate for the vaginal ring was 0.65 pregnancies per 100 women-years. The majority of women in the study considered insertion and removal of the vaginal ring to be easy, and 90 percent used the device correctly. Adverse effects that led to discontinuation of vaginal ring use most often were related to foreign body sensation, coital problems, and expulsion of the device. However, only 3.6 percent of women in the study stopped using the device for these reasons.
FIGURE 1.

Vaginal ring (NuvaRing). The circular ring is flexible and easily inserted into the vagina. Unlike the diaphragm, the vaginal ring does not have to be in a specific position, because absorption of the hormones can occur anywhere in the vagina.
Photograph used with permission of Organon Inc., West Orange, N.J.
In terms of cycle control, the vaginal ring is associated with a lower incidence of breakthrough bleeding than levonorgestrel–ethinyl estradiol OCPs, and with a higher rate of normal withdrawal bleeding.16
COMBINED HORMONAL INJECTION
The combined hormonal injection, which contains 25 mg of medroxyprogesterone acetate and 5 mg of estradiol cypionate, is another method of contraception that does not require daily adherence. Unlike progestin-only preparations, this injection is given monthly. The combined hormonal injection is available outside the United States. Although approved by the FDA, the combined hormonal injection currently is not being manufactured in this country, and its future availability is unknown.
Clinical trials have shown this contraceptive method to be highly effective, with life-table failure rates of zero to 0.2 pregnancies per 100 women-years.17 Because of the added estrogen, bleeding patterns usually are regular and comparable with the bleeding patterns occurring with combination OCPs.18
CONTRACEPTIVE PATCH
In 2002, the FDA approved the use of a combination contraceptive patch (Ortho Evra) that releases 20 mcg of ethinyl estradiol and 150 mcg of norelgestromin per day. It inhibits ovulation in much the same way as OCPs. The patch is applied weekly for three weeks, followed by a patch-free week during which withdrawal bleeding occurs. Recommended application sites include the upper arm, buttocks, lower abdomen, and upper torso (excluding the breasts).
The overall failure rate for the contraceptive patch has been reported to be only 0.88 pregnancies per 100 women-years, with a method failure rate of 0.7 pregnancies per 100 women-years. However, this form of contraception may be less effective in women weighing more than 90 kg (198 lb).19
In a study20 comparing the contraceptive patch and OCPs, spotting occurred at a higher rate in the first two months of patch use. However, spotting rates for the two contraceptive methods were similar in subsequent cycles. A key finding in this study was that compliance was significantly greater with patch use than with OCP use. Although adverse event rates were similar with both contraceptive methods, application site reactions were unique to the patch, and breast discomfort was more common in the first two months of patch use. Finally, the study showed that patch adhesion appears to be reliable, with only 2.8 percent of the patches partially detaching and 1.8 percent completely detaching.
LEVONORGESTREL INTRAUTERINE SYSTEM
The FDA recently approved the use of an intrauterine system (Mirena) that releases 20 mcg of levonorgestrel per day and provides effective contraception for at least five years (Figure 2). This intrauterine system has been shown to be as effective as copper-containing intrauterine devices (IUDs).21 [SOR A, systematic review of randomized controlled trials (RCTs)] Pregnancy rates are comparable with those occurring with surgical sterilization.22
Although copper-containing IUDs can increase bleeding and dysmenorrhea, the levonorgestrel system actually lessens these symptoms.21 The typical bleeding profile with the new intrauterine system is irregular bleeding or spotting for the first six months of use, followed by very light menses, with 20 percent of women having amenorrhea at one year of use. The amenorrhea results from the local action of levonorgestrel, which creates an atrophic endometrium. Because estradiol levels are maintained, osteopenia is not associated with this contraceptive method.
Because of the safety profile of the new intrauterine system and the high rates of oligomenorrhea and amenorrhea, the system currently is being evaluated for use in conditions such as dysfunctional uterine bleeding.9
Only minimal side effects have been found with the intrauterine system.23 As with copper-containing IUDs, there is a risk of expulsion and ectopic pregnancy. Side effects unique to the levonorgestrel system have been related to the hormonal component and include a slight increase in the rates of headaches and acne.21
Recent data indicate that copper-containing IUDs are associated with lower rates of pelvic inflammatory disease (PID) and infertility than was previously thought.24,25 Evidence suggests that the risk of PID is even lower with the levonorgestrel system.23 [Strength of recommendation: A, RCT]
The new intrauterine system and the standard copper-containing IUD are inserted using different devices. Hence, attention to technique is important. The levonorgestrel system costs significantly more than the standard IUD. Note that the copper-containing IUD has been shown to be the most cost-effective method of birth control at two years.2
FIGURE 2.
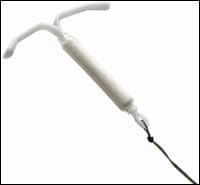
Intrauterine system (Mirena). This system differs from the traditional copper-containing intrauterine device in appearance and mechanism of action. The arms fold up rather than down, and the insertion technique is slightly different.
Photograph used with permission of Berlex Laboratories, Wayne, N.J.
Contraceptive Counseling
A brief overview of current contraceptive options is provided in Table 2.8,11,26,27 When counseling a patient about contraceptive methods, the family physician should be aware that the only effective contraceptive for any patient is the one that the patient is willing to use consistently and correctly. Ultimately, the patient is the one who must decide on the method of contraception; this decision often will be based on the patient's personal beliefs, preferences, and specific needs at the time. If needs change, a patient may want to switch to a different method of contraception. The physician's role is to educate the patient about the advantages and disadvantages of each contraceptive method that is medically appropriate for that patient and then allow the patient to choose the most desirable method.
TABLE 2 Overview of Contraceptive Options Currently Available in the United States
| Unintended pregnancies within first year of use (%)26 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Method | Typical use* | Perfect use † | Women continuing use at one year (%)26 | Frequency of use | Protection against STDs8 | Use while breastfeeding8 | Cost‡ | |
| None | 85 | 85 | — | — | — | — | — | |
| Spermicide | 29 | 15 | 42 | Each time | No § | Yes | $ 0.35 to 12 | |
| Withdrawal | 27 | 4 | 43 | Each time | No | Yes | None | |
| Periodic abstinence | ||||||||
| Calendar method | 25 | 9 | ∼ 50 | Each time | No | Yes | None | |
| Ovulation method | 25 | 3 | ∼ 50 | Each time | No | Yes | None | |
| Symptothermal method‖ | 25 | 2 | ∼ 50 | Each time | No | Yes | None | |
| Postovulation method | 25 | 1 | ∼ 50 | Each time | No | Yes | None | |
| Cervical cap: parous women∥ | 32 | 26 | 46 | Each time | No | Yes | 30 to 40 | |
| Cervical cap: nulliparous women∥ | 16 | 9 | 57 | Each time | No | Yes | 30 to 40 | |
| Diaphragm∥ | 16 | 6 | 57 | Each time | Yes# | Yes | 30 to 40 | |
| Female condom | 21 | 5 | 49 | Each time | Yes | Yes | 2 to 3 each | |
| Male condom | 15 | 2 | 53 | Each time | Yes | Yes | 0.50 to 2 each | |
| OCPs | 8 | 0.3 | 68 | Taken daily | No | ** † † | 20 to 50 per cycle | |
| Contraceptive patch | Unknown | 0.3 | 68 | Applied weekly | No | ** | 36 per month | |
| Vaginal ring | Unknown | 0.3 | 68 | Inserted every 4 weeks | No | ** | 43 per month | |
| Progestin-only OCPs | 3 | 0.3 | 56 | Given every 12 weeks | No | Yes | 30 to 35 every 3 months‡ ‡ | |
| Copper-containing IUD | 0.8 | 0.6 | 78 | Inserted every 10 years | No | Yes | 250 to 300 every 10 years§ § | |
| Levonorgestrel intrauterine system | 0.1 | 0.1 | 81 | Inserted every 5 years | No | Yes | 300 to 400 every 5 years§ § | |
| Female sterilization | 0.5 | 0.5 | 100 | Done once | No | Yes | 1,200 to 2,500 ∥ ∥ | |
| Male sterilization | 0.15 | 0.10 | 100 | Done once | No | Yes | 250 to 1,000 | |
STD = sexually transmitted disease; OCP = oral contraceptive pill; IUD = intrauterine device; HIV = human immunodeficiency virus.
*—Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
†—Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience accidental pregnancy during the first year if they do not stop use for any other reason.
‡—Cost information for spermicide, cervical cap, diaphragm, progestin-only injections, female and male sterilization, condoms, OCPs, IUD, and levonorgestrel intrauterine system from reference8; cost information for contraceptive patch, vaginal ring, and combined hormonal injection based on average prices in a survey conducted in the authors' area (prices may vary according to region).
§—Possible decrease in human papillomavirus transmission but no protection against HIV infection, gonorrhea, and chlamydial infection; potential increase in HIV transmission with use of nonoxynol-9.11
‖—Cervical mucus (ovulation) method supplemented by calendar method in preovulatory phase and by basal body temperature method in postovulatory phase.
∥—Method used with spermicide.
#—May protect against gonorrhea, chlamydial infection, cervical dysplasia, and pelvic inflammatory disease; protection against HIV infection cannot be assumed.
**—At less than six weeks postpartum, all estrogen-containing contraceptives should be avoided; although use between six weeks and six months postpartum is not contraindicated, there is concern that estrogen-containing contraceptives may decrease the quantity of breast milk.27
‡ ‡—Can breastfeed if taking progestin-only OCPs.
‡ ‡—Excludes cost of office visit for injection (cost may vary according to region).
§ §—Excludes cost of office visit for insertion (cost may vary according to region).
‖‖—Estimate for sterilization by minilaporatomy.
Adapted from Hatcher RA. A pocket guide to managing contraception. 5th ed. Tiger, Ga.: Bridging the Gap Foundation, 2000–2003:36,44–141, with additional information from references 8,11,26, and 27.
Many patients who use contraception have medical conditions; therefore, it is important to discuss the safety of various contraceptive methods, because some are contraindicated in certain circumstances. The World Health Organization27 has established medical eligibility criteria for contraceptive use. If a patient's first choice for contraception is contraindicated, the physician should help the patient select an appropriate alternative.
One important consideration is the risk of contracting sexually transmitted diseases (STDs). Many of the contraceptives that have the greatest efficacy in preventing pregnancy provide no protection against STDs, and methods (specifically condoms) that protect against STDs have higher contraceptive failure rates in typical users. In patients at risk for STDs, greater emphasis should be placed on use of a barrier method of contraception, either alone or in combination with another contraceptive method.28
Patients also should be made aware of options for emergency contraception. This topic has been reviewed previously29,30; however, two products (Plan B and Preven) are now available in the United States. The use of emergency contraception has not been shown to decrease compliance with standard contraceptive methods.31

