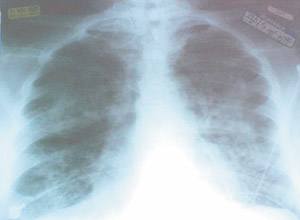
A 74-year-old man with a three-month history of worsening anemia was admitted for a transfusion of two units of packed red blood cells. The patient tolerated the first unit without difficulty. Before the second unit finished infusing, he developed shortness of breath, chills, back pain, and tachycardia. The transfusion was stopped, and he was treated with diphenhydramine, 50 mg intravenously, and furosemide, 20 mg intravenously. There was no improvement in his respiratory distress, despite significant diuresis. The patient had no previous history of blood transfusions or allergic reactions. Physical examination showed a well-developed elderly man in moderate distress. He was afebrile and normotensive. Supplemental oxygen at 10 L per minute by face mask was needed to keep his oxygen saturation above 95 percent. No stridor or other upper airway abnormalities were noted. Pulmonary examination was significant for bilateral rales, more pronounced at the bases. Skin examination was unremarkable, with no flushing or urticarial lesions, and the patient did not complain of pruritus. Electrocardiography showed sinus tachycardia but was otherwise unremarkable. A chest radiograph was obtained, which was interpreted as bilateral patchy alveolar filling and small pleural effusions (see accompanying figure).
Question
Based on the patient's history, physical examination, and chest films, which one of the following is the correct diagnosis?
A. Anaphylactic transfusion reaction.
B. Acute circulatory overload.
C. Bacterial contamination of blood product.
D. Transfusion-related acute lung injury.
E. Congestive heart failure.
Discussion
The answer is D: transfusion-related acute lung injury (TRALI). This syndrome can include dyspnea, bilateral pulmonary edema, hypotension, and fever. TRALI is the third leading cause of transfusion-related mortality and has been estimated to occur in one out of every 5,000 transfusions.1 Despite this incidence, it is probably underreported, because it may be mistaken for other causes of pulmonary edema.
Clinically, the presentation may be similar to adult respiratory distress syndrome (ARDS). Chest radiographs classically demonstrate bilateral interstitial and alveolar infiltrates. Although the initial lung injury with TRALI may be severe and even require mechanical ventilation, it typically improves rapidly, unlike the permanent damage that often occurs with ARDS. Still, there is an estimated 5 percent mortality rate associated with TRALI.
The exact etiology of TRALI remains unclear, but there is an association with anti-granulocyte antibodies in donor plasma. These antibodies are estimated to exist in as many as 20 percent of blood donors, but most patients who receive blood products containing these antibodies do not develop TRALI. Conditions that possibly predispose to TRALI include infection, recent surgery, and transfusion of large volumes of blood products.
An important distinction for TRALI is the recognition that the pulmonary edema is not cardiogenic and that the affected patient does not have circulatory overload. Patients with circulatory overload will respond favorably to diuretics, whereas treating TRALI patients with overzealous diuresis can lead to hypotension and further complications.
Anaphylactic transfusion reactions (most notably from ABO mismatch) also may present with respiratory distress. With anaphylaxis, however, upper airway findings such as stridor or laryngeal edema occur often, and most patients will experience pruritus and flushing, which may be followed by urticaria.
Patients who receive blood products affected by bacterial contamination often present with fever, hypotension, and vascular collapse. Respiratory distress and pulmonary edema are not seen as frequently.
Pulmonary edema can worsen suddenly in patients with chronic heart failure, but a transfusion-related reaction was more likely for this patient.
This patient was diagnosed as having a TRALI reaction, and no further diuresis was attempted. He responded well to supportive therapy with noninvasive positive pressure ventilation and was discharged 72 hours later without further complications. Follow-up studies revealed antigranulocyte antibodies in the donor's blood. The donor was identified and advised against further blood donation.

